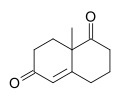
Wieland–Miescher ketone
 | |
| Names | |
|---|---|
| IUPAC name
8a-Methyl-3,4,7,8-tetrahydro-2H-naphthalene-1,6-dione | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.039.497 |
| PubChem CID |
|
| |
| |
| Properties | |
| C11H14O2 | |
| Molar mass | 178.23 g/mol |
| Melting point | 47 to 50 °C (117 to 122 °F; 320 to 323 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The Wieland–Miescher ketone [2] is a racemic bicyclic diketone (enedione) and is a versatile synthon which has so far been employed in the total synthesis of more than 50 natural products, predominantly sesquiterpenoids, diterpenes and steroids possessing possible biological properties including anticancer, antimicrobial, antiviral, antineurodegenerative and immunomodulatory activities. The reagent is named after two chemists from Ciba Geigy, Karl Miescher and Peter Wieland (not to be confused with Heinrich Otto Wieland). Examples of syntheses performed using the optically active enantiomer of this diketone as a starting material are that of ancistrofuran [3] and the Danishefsky total synthesis of Taxol.[4]
Most advances in total synthesis methods starting from Wieland–Miescher ketone were fueled by the search for alternative methods for the industrial synthesis of contraceptive and other medicinally relevant steroids, an area of research that flourished in the 1960s and 1970s.[5] Wieland–Miescher ketone contains the AB-ring structure of steroids and is for this reason an attractive starting material for the steroid skeleton, an approach used in one successful synthesis of adrenosterone.[6]
The original Wieland–Miescher ketone is racemic and prepared in a Robinson annulation of 2-methyl-1,3-cyclohexanedione and methyl vinyl ketone. The intermediate alcohol is not isolated.[7] The required 2-methyl-1,3-cyclohexanedione can be prepared from resorcinol[8] by hydrogenation over Raney nickel to dihydroresorcinol as the enolate followed by alkylation with methyl iodide.
An enantioselective synthesis employs L-proline as an organocatalyst:[9]
This reaction appeared in 1971 in the patent literature by Z. G. Hajos and D. R. Parrish. In this patent, the isolation and characterization of the above pictured optically active intermediate bicyclic ketol (in parentheses) has also been described, because they worked at ambient temperature in anhydrous dimethylformamide (DMF) solvent. Working in DMSO solvent does not allow isolation of the bicyclic ketol intermediate, it leads directly to the optically active bicyclic dione.[10] The reaction is called the Hajos-Parrish reaction or the Hajos-Parrish-Eder-Sauer-Wiechert reaction.[11]
This reaction has also been performed in a one-step procedure, leading to 49% yield and 76% Enantiomeric excess (ee):[12]
Other proline-based catalysts have been investigated [13]
References
- ↑ (±)-8a-Methyl-3,4,8,8a-tetrahydro-1,6(2H,7H)-naphthalenedione at Sigma-Aldrich
- ↑ Wieland, P.; Miescher, K. Über die Herstellung mehrkerniger Ketone., Helv. Chim. Acta 1950, 33, 2215. doi:10.1002/hlca.19500330730
- ↑ Ciceri, Paola, Demnitz, F.W. Joachim, Souza, Márcia C.F. de, Lehmanna, Maik. A Common Approach to the Synthesis of Monocyclofarnesyl Sesquiterpenes. J. Braz. Chem. Soc. 1998, 9, 409-414. ISSN 0103-5053. (Article)
- ↑ Samuel J. Danishefsky, John J. Masters, Wendy B. Young, J. T. Link, Lawrence B. Snyder, Thomas V. Magee, David K. Jung, Richard C. A. Isaacs, William G. Bornmann, Cheryl A. Alaimo, Craig A. Coburn, and Martin J. Di Grandi (1996). "Total Synthesis of Baccatin III and Taxol". J. Am. Chem. Soc. 118 (12): 2843-2859. doi:10.1021/ja952692a
- ↑ Wiechert, R. The Role of Birth Control in the Survival of the Human Race. Angew. Chem. Int. Ed. 1977, 16, 506-513.
- ↑ Dzierba, C. D.; Zandi, K. S.; Moellers, T.; Shea, K. J. An Ascending Synthesis of Adrenalcorticosteroids. The Total Synthesis of (+)-Adrenosterone. J. Am. Chem. Soc. 1996, 118, 4711-4712.
- ↑ Wieland-Miescher synthesis: a) water, acetic acid, hydroquinone (stabilizes EVK) b)L-proline, DMSO d) hydrogen, Raney nickel, sodium hydroxide e) methyl iodide, dioxane. specific rotation 68°.
- ↑ A. B. Mekler, S. Ramachandran, S. Swaminathan, and Melvin S. Newman Organic Syntheses, Coll. Vol. 5, p.743 (1973); Vol. 41, p.56 (1961). (Article)
- ↑ 1,6(2H, 7H)-Naphthalenedione, 3,4,8,8a-tetrahydro-8a-methyl-, (S)- Paul Buchschacher, A. Fürst, and J. Gutzwiller Organic Syntheses, Coll. Vol. 7, p.368 (1990); Vol. 63, p.37 (1985). (Article)
- ↑ Z. G. Hajos, D. R. Parrish, German Patent DE 2102623 (29 July 1971) and USP 3,975,440 (Aug.17, 1976) Example 21.
- ↑ 1 H-Indene-1,5(6 H)-dione, 2,3,7,7a-tetrahydro-7a-methyl-, (S)- Zoltan G. Hajos and David R. Parrish Organic Syntheses, Coll. Vol. 7, p.363 (1990); Vol. 63, p.26 (1985) Article Identical reaction with 2-methyl-1,3-cyclopentanedione (5 membered ring instead of a 6 membered ring)
- ↑ A proline-catalyzed asymmetric Robinson annulation reaction Tetrahedron Letters, Volume 41, Issue 36, September 2000, Pages 6951-6954 Tommy Bui and Carlos F. Barbas doi:10.1016/S0040-4039(00)01180-1
- ↑ Org. Synth. 2011, 88, 330-341 Link

