Transition metal dinitrogen complex

Metal dinitrogen complexes are coordination compounds that contain the dinitrogen (N2) as a ligand. In the area of coordination chemistry, the atomic and diatomic forms of nitrogen are distinguished, although otherwise "nitrogen" refers to N2.
Metal complexes of N2 have been studied since 1965 when the first complex was reported by Allen and Senoff.[1] This complex, [Ru(NH3)5(N2)]2+ was synthesised from hydrazine hydrate and ruthenium trichloride and consists of a 16e− [Ru(NH3)5]2+ centre attached to one end of N2.[2][3] Interest in such complexes arises because N2 comprises the majority of the atmosphere and because many useful compounds contain nitrogen atoms. Biological nitrogen fixation probably occurs via the binding of N2 to a metal center in the enzyme nitrogenase, followed by a series of steps that involve electron transfer and protonation. The hydrogenation of N2 is only weakly exothermic, hence the industrial hydrogenation of nitrogen via the Haber-Bosch Process employs high pressures and high temperatures.[4]
Bonding modes
In terms of its bonding to transition metals, N2 is related to CO and acetylene as all three species have triple bonds. A variety of bonding modes have been characterized.
End-on
As a ligand, N2 usually binds to metals as an "end-on" ligand, as illustrated by Allen and Senoff's complex. Such complexes are usually analogous to related CO derivatives. A good example of this relationship are the complexes IrCl(CO)(PPh3)2 and IrCl(N2)(PPh3)2.[5] Few complexes contain more than one N2 ligand, and no example features three (in contrast metal hexacarbonyls are common). The dinitrogen ligand in W(N2)2(Ph2PCH2CH2PPh2)2 can be reduced to produce ammonia.[6]
An end-on complex also containing a hydride ligand is [FeH(N2)(dmpe)2)]+. This cation undergoes deprotonation to form a Fe(N2)(dmpe)2 which reacts with hydrogen chloride to give ammonium.
Bridging, end-on
N2 also serves as a bridging ligand, as illustrated by {[Ru(NH3)5]2(μ-N2)}4+.
A study in 2006 of iron-dinitrogen complexes showed that the N–N bond is significantly weakened upon complexation with iron atoms with a low coordination number. The complex involved bidentate chelating ligands attached to the iron atoms in the Fe–N–N–Fe core, in which N
2 acts as a bridging ligand between the iron atoms. Increasing the coordination number of iron by modifying the chelating ligands and adding another ligand per iron atom showed an increase in the strength of the N–N bond in the resulting complex. It is thus suspected that Fe in a low-coordination environment is a key factor to the fixation of nitrogen by the nitrogenase enzyme, since its Fe–Mo cofactor also features Fe with low coordination numbers.[7]
Side-on, bridging
In a second mode of bridging, bimetallic complexes are known wherein the N-N vector is perpendicular to the M-M vector. One example is [(η5-C5Me4H)2Zr]2(μ2,η2,η2-N2).[8] The dimetallic complex {(C5HMe4)2Zr}2{μ-η2-N2} reacts with H2.[9](Figure 12)
|
A related ditantalum tetrahydride complex also reduce the N2[10].
Reactivity
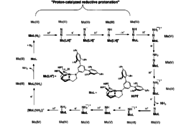
Some metal-dintrogen complexes catalyze the hydrogenation of N2 to ammonia. One such catalyst is the Mo(III) triamidoaminocomplex Mo[(HIPTN)3N].[11] This system utilizes reductants such as Cp*2Cr and protonating reagents such as 2,6-lutidinium salts.[12] The reducing equivalents and protons are added stepwise. The cycle begins with N2 coordinated to Mo in an end-on fashion. The oxidation state of molybdenum remains unchanged until the last step when it is oxidized from Mo(III) to Mo(IV). For the rest of the cycle, dinitrogen is bound to Mo via a double or a triple bond accompanied by the change in oxidation state of Mo from +4 to +5 and the release of 2 equivalents of ammonia gas. Another homogeneous catalytic system based on molybdenum[13](Figure 9) The precatalyst is a dinitrogen-bridged dimolybdenum complex bearing PNP(2,6-bis(di-tert-butyl phosphinomethyl) pyridine) ligand. Adding acid to the precatalyst leads to the splitting of dinitrogen bridged dimolybdenum complex and generates an inactive molybdenum hydride cation and an active molybdenum nitrogen adduct(MoPNP(N2)). The latter MoPNP(N2) can then undergo a full catalytic cycle with proton and electron addition to form ammonia. However this catalytic system needs a huge excess of cobaltocene (72 equiv) and [LutH]OTf (96 equiv).
References
- ↑ Senoff, Caesar V. (1990). "The discovery of [Ru(NH3)5N2]2+: A case of serendipity and the scientific method". Journal of Chemical Education. 67 (5): 368. doi:10.1021/ed067p368.
- ↑ A. D. Allen; C. V. Senoff (1965). "Nitrogenopentammineruthenium(II) complexes". Journal of the Chemical Society, Chemical Communications (24): 621. doi:10.1039/C19650000621.
- ↑ Fryzuk, Michael D. (2013). "N2 coordination". Chem. Commun. 49: 4866–4868. doi:10.1039/C3CC42001A.
- ↑ Fryzuk, M. D.: Johnson, S. A (2000). "The continuing story of dinitrogen activation". Coordination Chemistry Reviews. 200–202: 379–409. doi:10.1016/S0010-8545(00)00264-2.
- ↑ Collman, J. P.; Hoffman, N. W.; Hosking, J. W. (2000). "trans-Chloro(nitrogen)bis(triphenylphosphine)iridium (I)". Inorganic Syntheses. 12: 8–11. ISBN 978-0-470-13171-8. doi:10.1002/9780470132432.ch2.
- ↑ Modern Coordination Chemistry: The Legacy of Joseph Chatt" G. J. Leigh (Editor), N. W. Winterton (Editor) Springer Verlag (2002). ISBN 0-85404-469-8
- ↑ Smith, J.; Sadique, A.; Cundari, T.; Rodgers, K.; Lukat-Rodgers, G.; Lachicotte, R.; Flaschenriem, C.; Vela, J.; Holland, P. (2006). "Studies of low-coordinate iron dinitrogen complexes". Journal of the American Chemical Society. 128 (3): 756–769. PMID 16417365. doi:10.1021/ja052707x.
- ↑ Bernskoetter, W. H.; Lobkovsky, E.; Chirik, P. J. (2005). "Kinetics and Mechanism of N2 Hydrogenation in Bis(cyclopentadienyl) Zirconium Complexes and Dinitrogen Functionalization by 1,2-Addition of a Saturated C-H Bond". Journal of the American Chemical Society. 127 (40): 14051–14061. PMID 16201827. doi:10.1021/ja0538841.
- ↑ Pool, Jaime A.; Lobkovsky, Emil; Chirik, Paul J. "Hydrogenation and cleavage of dinitrogen to ammonia with a zirconium complex". Nature. 427 (6974): 527–530. PMID 14765191. doi:10.1038/nature02274.
- ↑ Fryzuk, Michael D. (2008-09-20). "Side-on End-on Bound Dinitrogen: An Activated Bonding Mode That Facilitates Functionalizing Molecular Nitrogen". Accounts of Chemical Research. 42 (1): 127–133. doi:10.1021/ar800061g.
- ↑ Yandulov, Dmitry V.; Schrock, Richard R. (2003-07-04). "Catalytic Reduction of Dinitrogen to Ammonia at a Single Molybdenum Center". Science. 301 (5629): 76–78. ISSN 0036-8075. PMID 12843387. doi:10.1126/science.1085326.
- ↑ Ballmann, Joachim; Munhá, Rui F.; Fryzuk, Michael D. "The hydride route to the preparation of dinitrogen complexes". Chemical Communications. 46 (7): 1013. doi:10.1039/b922853e.
- ↑ Arashiba, Kazuya; Miyake, Yoshihiro; Nishibayashi, Yoshiaki. "A molybdenum complex bearing PNP-type pincer ligands leads to the catalytic reduction of dinitrogen into ammonia". Nature Chemistry. 3 (2): 120–125. doi:10.1038/nchem.906.
2N2-3D-balls.png)

.png)