Tris(bipyridine)ruthenium(II) chloride
ruthenium(II)_chloride.png) | |
ruthenium(II)-chloride-powder.jpg) | |
| Names | |
|---|---|
| Other names
Ru-bpy Ruthenium-tris(2,2’-bipyridyl) dichloride | |
| Identifiers | |
| |
| ECHA InfoCard | 100.034.772 |
| RTECS number | VM2730000 |
| Properties | |
| C30H24N6Cl2Ru·6H2O | |
| Molar mass | 640.53 g/mol (anhydrous) 748.62 g/mol (hexahydrate) |
| Appearance | red solid |
| Density | solid |
| Melting point | >300 °C |
| slightly soluble in water; soluble in acetone | |
| Structure | |
| Octahedral | |
| 0 D | |
| Hazards | |
| Main hazards | mildly toxic |
| R-phrases (outdated) | none |
| S-phrases (outdated) | S22 S24/25 |
| Related compounds | |
| Related compounds |
Ruthenium trichloride |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Tris(bipyridine)ruthenium(II) chloride is the coordination compound with the formula [Ru(bpy)3]Cl2. This red crystalline salt is obtained as the hexahydrate, although all of the properties of interest are in the cation [Ru(bpy)3]2+, which has received much attention because of its distinctive optical properties. The chlorides can be replaced with other anions, such as PF6−.
Synthesis and structure
-cation-3D-balls.png)
This salt is prepared by treating an aqueous solution of ruthenium trichloride with 2,2'-bipyridine. In this conversion, Ru(III) is reduced to Ru(II), and hypophosphorous acid is typically added as a reducing agent.[1] [Ru(bpy)3]2+ is an octahedral coordination complex. The complex is chiral, with D3 symmetry. It has been resolved into its enantiomers, which are kinetically stable. In excited state the molecule loses symmetry and attains C2 symmetry, as the transferred electron doesn't distribute symmetrically to the 3 bipyridyl ligands in the lowest lying triplet excited state.[2]
Photochemistry of [Ru(bpy)3]2+
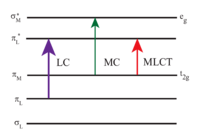
[Ru(bpy)3]2+ absorbs UV light and visible light. In aqueous medium the molecule strongly absorbs at 452 ± 3 nm corresponding to MLCT transition (extinction coefficient of 14,600 M−1cm−1), 285 nm corresponding to ligand centered π*← π transition and a weak transition around 350 nm (d-d transition).[3] Solutions of the resulting excited state have a comparatively long lifetime of 890 nanoseconds in acetonitrile[4] and 650 ns in water.[4] The excited state relaxes to the ground state by emission of a photon at the wavelength of 620 nm. The long lifetime of the excited state is attributed to the fact that it is triplet, whereas the ground state is a singlet state and in part due to the fact that the structure of the molecule allows for charge separation. Singlet-triplet transitions are forbidden and therefore often slow.
The triplet excited state has both oxidizing and reducing properties. This unusual situation arises because the excited state can be described as an Ru3+ complex containing a bpy− ligand. Thus, the photochemical properties of [Ru(bpy)3]2+ are reminiscent of the photosynthetic assembly, which also involves separation of an electron and a hole.[5]
[Ru(bpy)3]2+ has been examined as a photosensitiser for both the oxidation and reduction of water. Upon absorbing a photon, [Ru(bpy)3]2+ converts to the aforementioned triplet state, denoted [Ru(bpy)3]2+*. This species transfers an electron, located on one bpy ligand, to a sacrificial oxidant such as peroxodisulfate (S2O82−). The resulting [Ru(bpy)3]3+ is a powerful oxidant and oxidizes water into O2 and protons via a metal oxide catalyst.[6] Alternatively, the reducing power of [Ru(bpy)3]2+* can be harnessed to reduce methylviologen, a recyclable carrier of electrons, which in turn reduces protons at a platinum catalyst. For this process to be catalytic, a sacrificial reductant, such as EDTA4− or triethanolamine is provided to return the Ru(III) back to Ru(II).
Derivatives of [Ru(bpy)3]2+ are numerous.[7][8] Such complexes are widely discussed for applications in biodiagnostics, photovoltaics and organic light-emitting diode, but no derivative has been commercialized. Application of [Ru(bpy)3]2+ and its derivatives to fabrication of optical chemical sensors is arguably one of the most successful areas so far.[9]
[Ru(bpy)3]2+ and photoredox catalysis
Photoredox catalysis using combination of [Ru(bpy)3]2+ catalyst and visible light has been considered as a tool for preparative organic chemistry since the 1970s.[10] However, only a few research groups dealt with this topic until the beginning of the 21st century. Since 2008, development of this bond-forming strategy for organic synthesis has gained considerable momentum due to the seminal studies by MacMillan,[11] Yoon,[12] and Stephenson[13] groups. Depending on the choice of suitable reductive or oxidative quencher, the [Ru(bpy)3]2+ catalyst can be used to trigger photoreduction or photooxidation, respectively. Current status of this field has been recently summarized in several review articles.[10][14][15][16] It can be anticipated that photoredox reactivity of complexes based on metals other than Ru (e.g. Ir, Re, and bimetallic photocatalysts) will also be intensively explored. Additionally, transformations triggered by purely organic photoredox catalysts start to attract attention as highlighted in the recent report by Zeitler group.[17]
Safety
Metal bipyridine as well as related phenanthroline complexes are generally bioactive, as they can act as intercalating agents.
References
- ↑ Broomhead J. A.; Young C. G. (1990). "Tris(2,2'-bipyridine)Ruthenium(II) Dichloride Hexahydrate". Inorganic Syntheses. 28: 338–340. doi:10.1002/9780470132593.ch86.
- ↑ Yeh, Alvin T.; Charles V. Shank; James K. McCusker (2000). "Ultrafast Electron Localization Dynamics Following Photo-Induced Charge Transfer". Science. 289 (5481): 935–938. PMID 10937993. doi:10.1126/science.289.5481.935. Retrieved 19 February 2014.
- ↑ Kalyanasundaram, K. (1982). "Photophysics, photochemistry and solar energy conversion with tris(bipyridyl)ruthenium(II) and its analogues". Coordination Chemistry Reviews. 46: 159–244. doi:10.1016/0010-8545(82)85003-0. Retrieved 19 February 2014.
- 1 2 Montalti, Marco; Alberto Cedi; Luca Prodi; M. Teresa Gandolfi (2006). Handbook of Photochemistry 3rd edition. 6000 Broken Sound Prkway NW, Suite 200 Boca Raton, FL: CRC press Taylor & Francis Group. pp. 379–404. ISBN 0-8247-2377-5.
- ↑ A. J. Bard & M. A. Fox (1995). "Artificial Photosynthesis: Solar Splitting of Water to Hydrogen and Oxygen". Acc. Chem. Res. 28 (3): 141–145. doi:10.1021/ar00051a007.
- ↑ M. Hara, C. C. Waraksa, J. T. Lean, B. A. Lewis and T. E. Mallouk (2000). "Photocatalytic Water Oxidation in a Buffered Tris(2,2'-bipyridyl)ruthenium Complex-Colloidal IrO2 System". J. Phys. Chem. A. 104 (22): 5275–5280. doi:10.1021/jp000321x.
- ↑ A. Juris, V. Balzani, F. Barigelletti, S. Campagna, P. Belser and A von Zelewsky (1988). "Ru(II) polypyridine complexes - photophysics, photochemistry, electrochemistry, and chemiluminescence". Coord. Chem. Rev. 84: 85–277. doi:10.1016/0010-8545(88)80032-8.
- ↑ S. Campagna, F. Puntoriero, F. Nastasi, G. Bergamini and V. Balzani (2007). "Photochemistry and photophysics of coordination compounds: ruthenium". Top. Curr. Chem. Topics in Current Chemistry. 280: 117–214. ISBN 978-3-540-73346-1. doi:10.1007/128_2007_133.
- ↑ G. Orellana* and D. Garcia-Fresnadillo (2004). "Environmental and Industrial Optosensing with Tailored Luminescent Ru(II) Polypyridyl Complexes". Springer Ser. Chem. Sens. Biosens. 1: 309–357. doi:10.1007/978-3-662-09111-1_13.
- 1 2 F. Teply (2011). "Photoredox catalysis by [Ru(bpy)3]2+ to trigger transformations of organic molecules. Organic synthesis using visible-light photocatalysis and its 20th century roots". Collect. Czech. Chem. Commun. Free access article. 76 (7): 859–917. doi:10.1135/cccc2011078.
- ↑ D. A. Nicewicz; D. W. C. MacMillan (2008). "Merging photoredox catalysis with organocatalysis: The direct asymmetric alkylation of aldehydes". Science. 322 (5898): 77–80. PMC 2723798
 . PMID 18772399. doi:10.1126/science.1161976.
. PMID 18772399. doi:10.1126/science.1161976. - ↑ M. A. Ischay; M. E. Anzovino; J. Du; T. P. Yoon (2008). "Efficient visible light photocatalysis of [2+2] enone cycloadditions". J. Am. Chem. Soc. 130 (39): 12886–12887. PMID 18767798. doi:10.1021/ja805387f.
- ↑ J. M. R. Narayanam; J. W. Tucker; C. R. J. Stephenson (2009). "Electron-transfer photoredox catalysis: Development of a tin-free reductive dehalogenation reaction". J. Am. Chem. Soc. 131 (25): 8756–8757. doi:10.1021/ja9033582.
- ↑ J. M. R. Narayanam; C. R. J. Stephenson (2011). "Visible light photoredox catalysis: applications in organic synthesis". Chem. Soc. Rev. 40: 102–113. doi:10.1039/b913880n.
- ↑ T. P. Yoon; M. A. Ischay; J. Du (2010). "Visible light photocatalysis as a greener approach to photochemical synthesis". Nat. Chem. 2 (7): 527–532. PMID 20571569. doi:10.1038/nchem.687.
- ↑ K. Zeitler (2009). "Photoredox catalysis with visible light". Angew. Chem. Int. Ed. 48 (52): 9785–9789. doi:10.1002/anie.200904056.
- ↑ M. Neumann; S. Füldner; B. König; K. Zeitler (2011). "Metal-free, cooperative asymmetric organophotoredox catalysis with visible light". Angew. Chem. Int. Ed. 50 (4): 951–954. PMID 20878819. doi:10.1002/anie.201002992.