Silicone

Silicones, also known as polysiloxanes, are polymers that include any inert, synthetic compound made up of repeating units of siloxane, which is a chain of alternating silicon atoms and oxygen atoms, frequently combined with carbon and/or hydrogen. They are typically heat-resistant and rubber-like, and are used in sealants, adhesives, lubricants, medicine, cooking utensils, and thermal and electrical insulation. Some common forms include silicone oil, silicone grease, silicone rubber, silicone resin, and silicone caulk.[1]
Chemistry
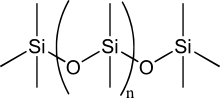
More precisely called polymerized siloxanes or polysiloxanes, silicones consist of an inorganic silicon-oxygen backbone chain (⋯-Si-O-Si-O-Si-O-⋯) with organic side groups attached to the silicon atoms. These silicon atoms are tetravalent. So, silicones are polymers constructed from inorganic-organic monomers. Silicones have in general the chemical formula [R2SiO]n, where R is an organic group such as an alkyl (methyl, ethyl) or phenyl group.
In some cases, organic side groups can be used to link two or more of these -Si-O- backbones together. By varying the -Si-O- chain lengths, side groups, and crosslinking, silicones can be synthesized with a wide variety of properties and compositions. They can vary in consistency from liquid to gel to rubber to hard plastic. The most common siloxane is linear polydimethylsiloxane (PDMS), a silicone oil. The second largest group of silicone materials is based on silicone resins, which are formed by branched and cage-like oligosiloxanes.
Terminology and history
F. S. Kipping and Matt Saunders coined the word silicone in 1901 to describe polydiphenylsiloxane by analogy of its formula, Ph2SiO (Ph stands for phenyl, C6H5), with the formula of the ketone benzophenone, Ph2CO (his term was originally silicoketone). Kipping was well aware that polydiphenylsiloxane is polymeric whereas benzophenone is monomeric and noted that Ph2SiO and Ph2CO had very different chemistry.[2][3] The discovery of the structural differences between Kippings' molecules and the ketones means that silicone is no longer the correct term (though it remains in common usage) and that the term siloxanes is correct according to the nomenclature of modern chemistry.[4]
Silicone is sometimes mistakenly referred to as silicon. The chemical element silicon is a crystalline metalloid widely used in computers and other electronic equipment. Although silicones contain silicon atoms, they also include carbon, hydrogen, oxygen, and perhaps other kinds of atoms as well, and have physical and chemical properties that are very different from elemental silicon.
A true silicone group with a double bond between oxygen and silicon does not commonly exist in nature; chemists find that the silicon atom usually forms single bonds with each of two oxygen atoms, rather than a double bond to a single atom. Polysiloxanes are among the many substances commonly known as "silicones".
Molecules containing silicon-oxygen double bonds do exist and are called silanones but they are very reactive. Despite this, silanones are important as intermediates in gas-phase processes such as chemical vapor deposition in microelectronics production, and in the formation of ceramics by combustion.[5]
Synthesis
Most common are materials based on polydimethylsiloxane, which is derived by hydrolysis of dimethyldichlorosilane. This dichloride reacts with water as follows:
- n Si(CH3)2Cl2 + n H2O → [Si(CH3)2O]n + 2n HCl
The polymerization typically produces linear chains capped with Si-Cl or Si-OH (silanol) groups. Under different conditions the polymer is a cyclic, not a chain.[1]
For consumer applications such as caulks silyl acetates are used instead of silyl chlorides. The hydrolysis of the acetates produce the less dangerous acetic acid (the acid found in vinegar) as the reaction product of a much slower curing process. This chemistry is used in many consumer applications, such as silicone caulk and adhesives.
Branches or cross-links in the polymer chain can be introduced by using organosilicon precursors with fewer alkyl groups, such as methyltrichlorosilane and methyltrimethoxysilane. Ideally, each molecule of such a compound becomes a branch point. This process can be used to produce hard silicone resins. Similarly, precursors with three methyl groups can be used to limit molecular weight, since each such molecule has only one reactive site and so forms the end of a siloxane chain.
Combustion
When silicone is burned in air or oxygen, it forms solid silica (silicon dioxide)(SiO2) as a white powder, char, and various gases. The readily dispersed powder is sometimes called silica fume.
Properties
Silicones exhibit many useful characteristics, including:[1]
- Low thermal conductivity
- Low chemical reactivity
- Low toxicity
- Thermal stability (constancy of properties over a wide temperature range of −100 to 250 °C).
- The ability to repel water and form watertight seals.
- Does not stick to many substrates, but adheres very well to others, e.g. glass.
- Does not support microbiological growth.
- Resistance to oxygen, ozone, and ultraviolet (UV) light. This property has led to widespread use of silicones in the construction industry (e.g. coatings, fire protection, glazing seals) and the automotive industry (external gaskets, external trim).
- Electrical insulation properties. Because silicone can be formulated to be electrically insulative or conductive, it is suitable for a wide range of electrical applications.
- High gas permeability: at room temperature (25 °C), the permeability of silicone rubber for such gases as oxygen is approximately 400 times that of butyl rubber, making silicone useful for medical applications in which increased aeration is desired. Conversely, silicone rubbers cannot be used where gas-tight seals are necessary.
Uses
Silicones are used in many products. Ullmann's Encyclopedia of Industrial Chemistry lists the following major categories of application: Electrical (e.g., insulation), electronics (e.g., coatings), household (e.g., sealants for cooking apparatus), automobile (e.g., gaskets), aeroplane (e.g., seals), office machines (e.g., keyboard pads), medicine/dentistry (e.g., teeth impression molds), textiles/paper (e.g., coatings). For these applications, an estimated 400,000 tons of silicones were produced in 1991. Specific examples, both large and small are presented below.[1]
Automotive
In the automotive field, silicone grease is typically used as a lubricant for brake components since it is stable at high temperatures, is not water-soluble, and is far less likely than other lubricants to foul. It is also used as DOT 5 brake fluid.
Automotive spark plug wires are insulated by multiple layers of silicone to prevent sparks from jumping to adjacent wires, causing misfires. Silicone tubing is sometimes used in automotive intake systems (especially for engines with forced induction).
Sheet silicone is used to manufacture gaskets used in automotive engines, transmissions, and other applications.
Automotive body manufacturing plants and paint shops avoid silicones, as they may cause "fish eyes", small, circular craters in the finish.
Additionally, silicone compounds such as silicone rubber are used as coatings and sealants for airbags; the high strength of silicone rubber makes it an optimal adhesive/sealant for high impact airbags. Recent technological advancements allow convenient use of silicone in combination with thermoplastics to provide improvements in scratch and mar resistance and lowered coefficient of friction.
Coatings
Silicone films can be applied to such silica-based substrates as glass to form a covalently bonded hydrophobic coating.
Many fabrics can be coated or impregnated with silicone to form a strong, waterproof composite such as silnylon.
Cookware

- As a low-taint, non-toxic material, silicone can be used where contact with food is required. Silicone is becoming an important product in the cookware industry, particularly bakeware and kitchen utensils.
- Silicone is used as an insulator in heat-resistant potholders and similar items; however, it is more conductive of heat than similar less dense fiber-based products. Silicone oven mitts are able to withstand temperatures up to 260 °C (500 °F), allowing reaching into boiling water.
- Molds for chocolate, ice, cookies, muffins and various other foods.
- Non-stick bakeware and reusable mats used on baking sheets.
- Other products such as steamers, egg boilers or poachers, cookware lids, pot holders, trivets, and kitchen mats.
Defoaming
Silicones are used as active compound in defoamers due to their low water solubility and good spreading properties.
Dry cleaning
Liquid silicone can be used as a dry cleaning solvent, providing an alternative to the traditional chlorine-containing perchloroethylene (perc) solvent. Use of silicones in dry cleaning reduces the environmental impact of a typically high-polluting industry.
Electronics
Electronic components are sometimes encased in silicone to increase stability against mechanical and electrical shock, radiation and vibration, a process called "potting".
Silicones are used where durability and high performance are demanded of components under hard conditions, such as in space (satellite technology). They are selected over polyurethane or epoxy encapsulation when a wide operating temperature range is required (−65 to 315 °C). Silicones also have the advantage of little exothermic heat rise during cure, low toxicity, good electrical properties and high purity.
The use of silicones in electronics is not without problems, however. Silicones are relatively expensive and can be attacked by solvents. Silicone easily migrates as either a liquid or vapor onto other components.
Silicone contamination of electrical switch contacts can lead to failures by causing an increase in contact resistance, often late in the life of the contact, well after any testing is completed.[6][7] Use of silicone-based spray products in electronic devices during maintenance or repairs can cause later failures.
Firestops
| Wikimedia Commons has media related to Silicone foam. |
Silicone foam has been used in North American buildings in an attempt to firestop openings within fire-resistance-rated wall and floor assemblies to prevent the spread of flames and smoke from one room to another. When properly installed, silicone-foam firestops can be fabricated for building code compliance. Advantages include flexibility and high dielectric strength. Disadvantages include combustibility (hard to extinguish) and significant smoke development.
Silicone-foam firestops have been the subject of controversy and press attention due to smoke development from pyrolysis of combustible components within the foam, hydrogen gas escape, shrinkage, and cracking. These problems have led to reportable events among licensees (operators of nuclear power plants) of the Nuclear Regulatory Commission (NRC).
Silicone firestops are also used in aircraft.
 Silicone "foamfixer" pump used to apply silicone foam firestop materials.
Silicone "foamfixer" pump used to apply silicone foam firestop materials. Self-leveling silicone firestop system used around copper pipe through-penetrations in a two-hour fire-resistance rated concrete floor assembly.
Self-leveling silicone firestop system used around copper pipe through-penetrations in a two-hour fire-resistance rated concrete floor assembly.
Lubricants
Silicone greases are used for many purposes, such as bicycle chains, airsoft gun parts, and a wide range of other mechanisms. Typically, a dry-set lubricant is delivered with a solvent carrier to penetrate the mechanism. The solvent then evaporates, leaving a clear film that lubricates but does not attract dirt and grit as much as an oil-based or other traditional "wet" lubricant.
Silicone personal lubricants are also available for use in medical procedures or sexual activity. See below.
Medicine
Silicone is used in microfluidics, seals, gaskets, shrouds, and other applications requiring high biocompatibility. Additionally, the gel form is used in bandages and dressings, breast implants, testicle implants, pectoral implants, contact lenses, and a variety of other medical uses.
Scar treatment sheets are often made of medical grade silicone due to its durability and biocompatibility. Polydimethylsiloxane is often used for this purpose, since its specific crosslinking results in a flexible and soft silicone with high durability and tack.
Polydimethylsiloxane (PDMS) has been used as the hydrophobic block of amphiphilic synthetic block copolymers used to form the vesicle membrane of polymersomes.
Moldmaking
Two-part silicone systems are used as rubber molds to cast resins, foams, rubber, and low-temperature alloys. A silicone mold generally requires little or no mold-release or surface preparation, as most materials do not adhere to silicone. For experimental uses, ordinary one-part silicone can be used to make molds or to mold into shapes. If needed, common vegetable cooking oils or petroleum jelly can be used on mating surfaces as a mold-release agent.[8]
Cooking molds used as bakeware do not require coating with cooking oil, allowing the baked food to be more easily removed from the mold after cooking.
Ophthalmology
Silicone has many applications like silicone oil used to replace vitreous following vitrectomy, silicone intraocular lenses following cataract extraction, silicone tubes to keep nasolacrimal passage open following dacrycystorhinostomy, canalicular stents for canalicular stenosis, punctal plugs for punctal occlusion in dry eyes, silicone rubber and bands as an external tamponade in tractional retinal detachment, and anteriorly located break in rhegmatogenous retinal detachment.
Personal care
Silicones are ingredients widely used in skin care, color cosmetic and hair care applications. Some silicones, notably the amine functionalized amodimethicones, are excellent conditioners, providing improved compatibility, feel, and softness, and lessening frizz. The phenyltrimethicones, in another silicone family, are used in reflection-enhancing and color-correcting hair products, where they increase shine and glossiness (and possibly effect subtle color changes). Phenyltrimethicones, unlike the conditioning amodimethicones, have refractive indices (typically 1.46) close to that of human hair (1.54). However, if included in the same formulation, amodimethicone and phenyltrimethicone interact and dilute each other, making it difficult to achieve both high shine and excellent conditioning in the same product.[9]
Silicone rubber is commonly used in baby bottle nipples (teats) for its cleanliness, aesthetic appearance, and low extractable content.
Silicones are used in shaving products and personal lubricants.[10]
Plumbing and building construction
The strength and reliability of silicone rubber is widely acknowledged in the construction industry. One-part silicone sealants and caulks are in common use to seal gaps, joints and crevices in buildings. One-part silicones cure by absorbing atmospheric moisture, which simplifies installation. In plumbing, silicone grease is typically applied to O-rings in brass taps and valves, preventing lime from sticking to the metal.
Toys and hobbies
Silly Putty and similar materials are composed of silicones dimethyl siloxane, polydimethylsiloxane, and decamethyl cyclopentasiloxane, with other ingredients. This substance is noted for its unusual characteristics, e.g., that it bounces, but breaks when given a sharp blow; it can also flow like a liquid and will form a puddle given enough time.
Silicone "rubber bands" are a long-lasting popular replacement refill for real rubber bands in the 2013 fad "rubber band loom" toys at two to four times the price (in 2014). Silicone bands also come in bracelet sizes that can be custom embossed with a name or message. Large silicone bands are also sold as utility tie-downs.
Formerol is a silicone rubber (marketed as Sugru) used as an arts-and-crafts material, as its plasticity allows it to be moulded by hand like modeling clay. It hardens at room temperature and it is adhesive to various substances including glass and aluminum.[11]
In making aquariums, manufacturers now commonly use 100% silicone sealant to join glass plates. Glass joints made with silicone sealant can withstand great pressure, making obsolete the original aquarium construction method of angle-iron and putty. This same silicone is used to make hinges in aquarium lids or for minor repairs. However, not all commercial silicones are safe for aquarium manufacture, nor is silicone used for the manufacture of acrylic aquariums as silicones do not have long-term adhesion to plastics.[12]
Sex toys and lubricants
Silicone is a material of choice for soft sex toys, due to its softness, durability, cleanability, non-degradation by petroleum-based lubricants, and lack of phthalates, chemicals suspected of having carcinogenic and mutagenic effects on the skin and mucous membranes.[13][14][15]
Production and marketing
The global demand for silicones approached US$12.5 billion in 2008, approximately 4% up from the previous year. It continues similar growth in the following years to reach $13.5 billion by 2010. The annual growth is expected to be boosted by broader applications, introduction of novel products and increasing awareness of using more environmentally friendly materials.[16]
The leading global manufacturers of silicone base materials belong to three regional organizations: the European Silicone Center (CES) in Brussels, Belgium; the Environment Health and Safety Council (SEHSC) in Herndon, Virginia, USA; and the Silicone Industry Association of Japan (SIAJ) in Tokyo, Japan. Dow Corning Silicones, Evonik Industries, Momentive Performance Materials, Milliken and Company (SiVance Specialty Silicones), Shin-Etsu Silicones, Wacker Chemie, Bluestar Silicones, JNC Corporation, Wacker Asahikasei Silicone, and Dow Corning Toray represent the collective membership of these organizations. A fourth organization, the Global Silicone Council (GSC) acts as an umbrella structure over the regional organizations. All four are non-profit, having no commercial role; their primary missions are to promote the safety of silicones from a health, safety, and environmental perspective. As the European chemical industry is preparing to implement the Registration, Evaluation and Authorisation of Chemicals (REACH) legislation, CES is leading the formation of a consortium[17] of silicones, silanes, and siloxanes producers and importers to facilitate data and cost sharing.
Safety and environmental considerations
No "marked harmful effects on organisms in the environment" have been noted for silicones. Because they are widely used, they are pervasive.[18] They biodegrade readily, a process that is accelerated by a variety of catalysts, including clays.[1] Cyclic silicones have been shown to involve the occurrence of silanols during biodegration in mammals.[19]
Around 200 °C in oxygen-containing atmosphere, PDMS releases traces of formaldehyde but less than other common materials such as polyethylene.,[20][21] and by 200 °C (392 °F) Silicones (< 3 µg CH2O/(g·hr) for a high consistency silicone rubber to 48 µg CH2O/(g·hr)) were found to be superior to mineral oil and plastics (~400 µg CH2O/(g·hr)) at about 200 °C (392 °F), by 250 °C (482 °F) copious amounts of formaldehyde have been found to be produced for all silicones (1200 µg CH2O/(g·hr) to 4600 µg CH2O/(g·hr)).[21]
See also
References
- 1 2 3 4 5 Hans-Heinrich Moretto, Manfred Schulze, Gebhard Wagner (2005) "Silicones" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim. doi:10.1002/14356007.a24_057
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 362. ISBN 0-08-037941-9.
- ↑ Frederick Kipping, L. L. Lloyd (1901). "XLVII.-Organic derivatives of silicon. Triphenylsilicol and alkyloxysilicon chlorides". J. Chem. Soc., Trans. 79: 449–459. doi:10.1039/CT9017900449.
- ↑ James E. Mark; Harry R. Allcock; Robert West (24 March 2005). Inorganic Polymers. Oxford University. p. 155. ISBN 978-0-19-535131-6.
- ↑ V. N. Khabashesku; Z. A. Kerzina; K. N. Kudin; O. M. Nefedov (1998). "Matrix isolation infrared and density functional theoretical studies of organic silanones, (CH3O)2Si=O and (C6H5)2Si=O". J. Organomet. Chem. 566 (1–2): 45–59. doi:10.1016/S0022-328X(98)00726-8.
- ↑ Paul G. Slade (1999). "16.4.1". Electrical Contacts: Principles and Applications. CRC Press. p. 823. ISBN 0-8247-1934-4.
- ↑ W. Witter & R. Leiper (1979). "A Comparison for the Effects of Various Forms of Silicon Contamination on Contact Performance". IEEE Transactions on Components, Hybrids, and Manufacturing Technology. 2: 56. doi:10.1109/TCHMT.1979.1135411.
- ↑ Joe Hildreth. Chapter 8 – Silicone Caulk Molds. Myheap.com. Retrieved on 2013-08-17.
- ↑ Thomas Clausen et al. "Hair Preparations" in Ullmann's Encyclopedia of Industrial Chemistry, 2007, Wiley-VCH, Weinheim. doi:10.1002/14356007.a12_571.pub2
- ↑ Q. Ashton Acton: Silicones—Advances in Research and Application: 2013 Edition, ScholarlyEditions, 2013, ISBN 9781481692397, p. 226.
- ↑ Formerol fact sheet "Formerol fact sheet pdf"
- ↑ "Aquarium Silicone Applications". Aquarium-pond-answers.com. March 2007. Retrieved 2012-02-28.
- ↑ W.M. Kluwe (1986). "Carcinogenic potential of phthalic acid esters and related compounds: structure-activity relationships" (free text). Environmental Health Perspectives. 65: 271–278. JSTOR 3430194. PMC 1474699
 . PMID 3709453. doi:10.2307/3430194.
. PMID 3709453. doi:10.2307/3430194. - ↑ Norbert H. Kleinsasser; Ernst R. Kastenbauer; Herbert Weissacher; Ruth K. Muenzenrieder; Ulrich A. Harréus (2000). "Phthalates demonstrate genotoxicity on human mucosa of the upper aerodigestive tract". Environmental and Molecular Mutagenesis. 35 (1): 9–12. PMID 10692222. doi:10.1002/(SICI)1098-2280(2000)35:1<9::AID-EM2>3.0.CO;2-1.
- ↑ Walter J. Kozumbo; Rosanna Kroll; Robert J. Rubin (1982). "Assessment of the mutagenicity of phthalate esters". Environmental Health Perspectives. 45: 103–109. JSTOR 3429391. PMC 1568987
 . PMID 6754359. doi:10.2307/3429391.
. PMID 6754359. doi:10.2307/3429391. - ↑ "Market Report: World Silicone Market". Acmite Market Intelligence.
- ↑ "REACH consortium". Reach.silicones.eu. Retrieved 2012-02-28.
- ↑ Bienkowski, Brian (30 April 2013). "Chemicals from Personal Care Products Pervasive in Chicago Air". Scientific American. Retrieved 8 April 2015.
- ↑ S. Varaprath, K. L. Salyers, K. P. Plotzke and S. Nanavati "Identification of Metabolites of Octamethylcyclotetrasiloxane (D4) in Rat Urine" Drug Metab Dispos 1999, 27, 1267-1273.
- ↑ Dave Hard. DIELECTRIC FLUIDS FOR TRANSFORMER COOLING History and Types. General Electric
- 1 2 David C. Timpe Jr. Formaldehyde Generation from Silicone Rubber. Arlon
External links
| Look up silicone in Wiktionary, the free dictionary. |
| Wikimedia Commons has media related to Silicones. |
| Wikimedia Commons has media related to Caulking. |
- Science of Silicone Polymers (Silicone Science On-line, Centre Européen des Silicones – CES)