Polyimide
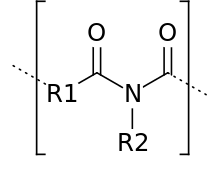
Polyimide (sometimes abbreviated PI) is a polymer of imide monomers. Polyimides have been in mass production since 1955. With their high heat-resistance, polyimides enjoy diverse applications in roles demanding rugged organic materials, e.g. high temperature fuel cells, displays, and various military roles. A classic polyimide is Kapton, which is produced by condensation of pyromellitic dianhydride and 4,4'-oxydianiline.[1]
Classification
According to the composition of their main chain, polyimides can be:
- Aliphatic (linear polyimides),
- Semi-aromatic,
- Aromatic: these are the most used polyimides because of their thermostability.
According to the type of interactions between the main chains, polyimides can be:
- Thermoplastic: very often called pseudothermoplastic.
- Thermosetting: commercially available as uncured resins, polyimide solutions, stock shapes, thin sheets, laminates and machined parts.
Synthesis
Several methods are possible to prepare polyimides, among them:
- The reaction between a dianhydride and a diamine (the most used method).
- The reaction between a dianhydride and a diisocyanate.
Dianhydrides used as precursors to these materials include pyromellitic dianhydride, benzoquinonetetracarboxylic dianhydride and naphthalene tetracarboxylic dianhydride. Common diamine building blocks include 4,4'-diaminodiphenyl ether ("DAPE"), meta-phenylenediamine ("MDA"), and 3,3-diaminodiphenylmethane.[1] Hundreds of diamines and dianhydrides have been examined to tune the physical and especially the processing properties of these materials. These materials tend to be insoluble and have high softening temperatures, arising from charge-transfer interactions between the planar subunits.[2]
Properties
Thermosetting polyimides are known for thermal stability, good chemical resistance, excellent mechanical properties, and characteristic orange/yellow color. Polyimides compounded with graphite or glass fiber reinforcements have flexural strengths of up to 50,000 psi (340 MPa) and flexural moduli of 3,000,000 psi (21,000 MPa). Thermoset polymer matrix polyimides exhibit very low creep and high tensile strength. These properties are maintained during continuous use to temperatures of up to 232 °C (450 °F) and for short excursions, as high as 400 °C (752 °F).[3] Molded polyimide parts and laminates have very good heat resistance. Normal operating temperatures for such parts and laminates range from cryogenic to those exceeding 260 °C (500 °F). Polyimides are also inherently resistant to flame combustion and do not usually need to be mixed with flame retardants. Most carry a UL rating of VTM-0. Polyimide laminates have a flexural strength half life at 249 °C (480 °F) of 400 hours.
Typical polyimide parts are not affected by commonly used solvents and oils — including hydrocarbons, esters, ethers, alcohols and freons. They also resist weak acids but are not recommended for use in environments that contain alkalis or inorganic acids. Some polyimides, such as CP1 and CORIN XLS, are solvent-soluble and exhibit high optical clarity. The solubility properties lend them towards spray and low temperature cure applications.
Applications
Polyimide materials are lightweight, flexible, resistant to heat and chemicals. Therefore, they are used in the electronics industry for flexible cables, as an insulating film on magnet wire and for medical tubing. For example, in a laptop computer, the cable that connects the main logic board to the display (which must flex every time the laptop is opened or closed) is often a polyimide base with copper conductors. Examples of polyimide films include Apical, Kapton, UPILEX, VTEC PI, Norton TH and Kaptrex.
The semiconductor industry uses polyimide as a high-temperature adhesive; it is also used as a mechanical stress buffer. Some polyimide can be used like a photoresist; both "positive" and "negative" types of photoresist-like polyimide exist in the market.
An additional use of polyimide resin is as an insulating and passivation[4] layer in the manufacture of digital semiconductor and MEMS chips. The polyimide layers have good mechanical elongation and tensile strength, which also helps the adhesion between the polyimide layers or between polyimide layer and deposited metal layer. The minimum interaction between the gold film and the polyimide film, coupled with high temperature stability of the polyimide film, results in a system that provides reliable insulation when subjected to various types of environmental stresses.[5][6]
Polyimide powder can be used to produce parts and shapes by sintering technologies (hot compression molding, direct forming, and isostatic pressing). Because of their high mechanical stability even at elevated temperatures they are used as bushings, bearings, sockets or constructive parts in demanding applications. To improve tribological properties compounds with solid lubricants like graphite, PTFE or molybdenum sulfide are common. Polyimide parts and shapes include P84 NT, VTEC PI, Meldin,[7] Vespel and Plavis.
In coal-fired power plants, waste incinerators or cement plants, polyimide fibres are used to filter hot gases. In this application, a polyimide needle felt separates dust and particulate matter from the exhaust gas.
Polyimide is also the most common material used for the reverse osmotic film in purification of water, or the concentration of dilute materials from water, such as maple syrup production.[8][9]
Multi-layer insulation used on spacecraft is usually made of polyimide coated with thin layers of aluminum. The gold-like material often seen on the outside of spacecraft is actually single aluminized polyimide, with the single layer of aluminum facing in.[10] The yellowish-brown polyimide gives the surface its gold-like color.
The IKAROS solar sailing spacecraft uses polyimide resin sails to operate without rocket engines.[11]
See also
References
- 1 2 Walter W. Wright and Michael Hallden-Abberton "Polyimides" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a21_253
- ↑ Liaw, Der-Jang; Wang, Kung-Li; Huang, Ying-Chi; Lee, Kueir-Rarn; Lai, Juin-Yih; Ha, Chang-Sik “Advanced polyimide materials: Syntheses, physical properties and applications” Progress in Polymer Science (2012), 37(7), 907-974. doi:10.1016/j.progpolymsci.2012.02.005
- ↑ P2SI 900HT Tech Sheet
- ↑ The effect of polyimide passivation on the electromigration of Cu multilayer interconnections
- ↑ Digital Isolation Offers Compact, Low-Cost Solutions to Challenging Design Problems
- ↑ iCoupler Products with isoPower Technology: Signal and Power Transfer Across Isolation Barrier Using Microtransformers
- ↑ http://www.seals.saint-gobain.com/meldin-material-grades.aspx
- ↑ http://www.wisegeek.net/what-is-a-reverse-osmosis-water-softener.htm | What is a reverse osmosis water softener?
- ↑ http://www.freepatentsonline.com/4532041.html patent 4532041; Asymmetric polyimide reverse osmosis membrane
- ↑ "Thermal Control Overview" (PDF). Sheldahl Multi Layer Insulation. Retrieved 28 December 2015.
- ↑ Courtland, Rachel (10 May 2010). "Maiden voyage for first true space sail". The New Scientist. Retrieved 11 June 2010.
Further reading
- Modern Plastic Mid-October Encyclopedia Issue, Polyimide, thermoset, page 146.
- Varun Ratta: POLYIMIDES: Chemistry & structure-property relationships – literature review (Chapter 1).