Personal genomics
| Part of a series on |
| Genetic genealogy |
|---|
| Concepts |
| Related topics |
Personal genomics is the branch of genomics concerned with the sequencing, analysis and interpretation of the genome of an individual. The genotyping stage employs different techniques, including single-nucleotide polymorphism (SNP) analysis chips (typically 0.02% of the genome), or partial or full genome sequencing. Once the genotypes are known, the individual's variations can be compared with the published literature to determine likelihood of trait expression, ancestry inference and disease risk.
Automated high-throughput sequencers have increased the speed and reduced the cost of sequencing, making it possible to offer genetic testing to consumers today for as little as $1,000. The emerging market of direct-to-consumer genome sequencing services has brought new questions about both the medical efficacy and the ethical dilemmas associated with widespread knowledge of individual genetic information.
In personalized medicine
Personalized medicine is a medical method that targets treatment structures and medicinal decisions based on a patient's predicted response or risk of disease.[1] The National Cancer Institute or NCI, an arm of the National Institutes of Health, lists a patient's genes, proteins, and environment as the primarily factors analyzed to prevent, diagnose, and treat disease through personalized medicine.[1]
There are various subcategories of the concept of personalized medicine such as predictive medicine, precision medicine and stratified medicine. Although these terms are used interchangeably to describe this practice, each carries individual nuances. Predictive medicine describes the field of medicine that utilizes information, often obtained through personal genomics techniques, to both predict the possibility of disease, and institute preventative measures for a particular individual.[2] Precision medicine is a term very similar to personalized medicine in that it focuses on a patient's genes, environment, and lifestyle; however, it is utilized by National Research Council to avoid any confusion or misinterpretations associated with the broader term. Stratified medicine is a version of personalized medicine which focuses on dividing patients into subgroups based on specific responses to treatment, and identifying effective treatments for the particular group.[3]
Examples of the use of personalized medicine include oncogenomics and pharmacogenomics. Oncogenomics is a field of study focused on the characterization of cancer–related genes. With cancer, specific information about a tumor is used to help create a personalized diagnosis and treatment plan.[4] Pharmacogenomics is the study of how a person's genome affects their response to drugs.[5] This field is relatively new but growing fast due in part to an increase in funding for the NIH Pharmacogenomics Research Network. Since 2001, there has been an almost 550% increase in the number of research papers in PubMed related to the search terms pharmacogenomics and pharmacogenetics.[6] This field allows researchers to better understand how genetic differences will influence the body's response to a drug and inform which medicine is most appropriate for the patient. These treatment plans will be able to prevent or at least minimize the adverse drug reactions which are a, "significant cause of hospitalizations and deaths in the United States." Overall, researchers believe pharmacogenomics will allow physicians to better tailor medicine to the needs of the individual patient.[5] As of November 2016, the FDA has approved 204 drugs with pharmacogenetics information in its labeling. These labels may describe genotype-specific dosing instructions and risk for adverse events amongst other information.[7]
Disease risk may be calculated based on genetic markers and genome-wide association studies for common medical conditions, which are multifactorial and include environmental components in the assessment. Diseases which are individually rare (less than 200,000 people affected in the USA) are nevertheless collectively common (affecting roughly 8-10% of the US population[8]). Over 2500 of these diseases (including a few more common ones) have predictive genetics of sufficiently high clinical impact that they are recommended as medical genetic tests available for single genes (and in whole genome sequencing) and growing at about 200 new genetic diseases per year.[9]
Cost of sequencing an individual's genome
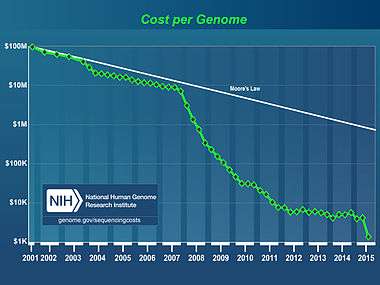
The cost of sequencing a human genome is dropping rapidly, due to the continual development of new, faster, cheaper DNA sequencing technologies such as "next-generation DNA sequencing".
The National Human Genome Research Institute, an arm of the U.S. National Institutes of Health, has reported that the cost to sequence a whole human-sized genome has dropped from about $14 million in 2006 to below $1,500 by late 2015.[11]
There are 6 billion base pairs in the diploid human genome. Statistical analysis reveals that a coverage of approximately ten times is required to get coverage of both alleles in 90% human genome from 25 base pair reads with shotgun sequencing.[12] This means a total of 60 billion base pairs that must be sequenced. An Applied Biosystems SOLiD, Illumina or Helicos[13] sequencing machine can sequence 2 to 10 billion base pairs in each $8,000 to $18,000 run. The cost must also take into account personnel costs, data processing costs, legal, communications and other costs. One way to assess this is via commercial offerings. The first such whole diploid genome sequencing (6 billion bp, 3 billion from each parent) was from Knome and their price dropped from $350,000 in 2008 to $99,000 in 2009.[14][15] This inspects 3000-fold more bases of the genome than SNP chip-based genotyping, identifying both novel and known sequence variants, some relevant to personal health or ancestry.[16] In June 2009, Illumina announced the launch of its own Personal Full Genome Sequencing Service at a depth of 30X for $48,000 per genome.[17] In 2010, they cut the price to $19,500.[18]
In 2009, Complete Genomics of Mountain View announced that it would provide full genome sequencing for $5,000, from June 2009.[19] This will only be available to institutions, not individuals.[20] Prices are expected to drop further over the next few years through economies of scale and increased competition.[21][22] As of 2014, full exome sequencing was offered by Gentle for less than $2,000, including personal counseling along with the results.[23]
The decreasing cost in general of genomic mapping has permitted genealogical sites to offer it as a service,[24] to the extent that one may submit one's genome to crowd sourced scientific endeavours such as OpenSNP[25] or DNA.land at the New York Genome Center, as examples of citizen science.[26] The Corpas family, encouraged by the low prices in genome sequencing, was the first example of citizen science crowd sourced analysis of personal genomes.[27]
Ethical issues
Genetic discrimination is discriminating on the basis of information obtained from an individual's genome. Genetic non-discrimination laws have been enacted in some US states[28] and at the federal level, by the Genetic Information Nondiscrimination Act (GINA). The GINA legislation prevents discrimination by health insurers and employers, but does not apply to life insurance or long-term care insurance. The passage of the Affordable Care Act in 2010 strengthened the GINA protections by prohibiting health insurance companies from denying coverage because of patient's "pre-existing conditions" and removing insurance issuers ability to adjust premium costs based on certain factors such as genetic diseases.[29] Given the ethical concerns about pre-symptomatic genetic testing of minors,[30][31][32][33] it is likely that personal genomics will first be applied to adults who can provide consent to undergo such testing, although genome sequencing is already proving valuable for children if any symptoms are present.[34]
There are also concerns regarding human genome research in developing countries. The tools for conducting whole genome analyses are generally found in high-income nations, necessitating partnerships between developed and developing countries in order to study the patients afflicted with certain diseases. The relevant tools for sharing access to the collected data are not equally accessible across low-income nations and without an established standard for this type of research, concerns over fairness to local researchers remain unsettled.[35]
Other issues
Genetic privacy
In the United States, biomedical research containing human subjects is governed by a baseline standard of ethics known as The Common Rule, which aims to protect a subject's privacy by requiring "identifiers" such as name or address to be removed from collected data.[36] A 2012 report by the Presidential Commission for the Study of Bioethical Issues stated, however, that "what constitutes 'identifiable' and 'de-identified' data is fluid and that evolving technologies and the increasing accessibility of data could allow de-identified data to become re-identified."[36] In fact, research has already shown that it is "possible to discover a study participant's identity by cross-referencing research data about him and his DNA sequence … [with] genetic genealogy and public-records databases." [37] This has led to calls for policy-makers to establish consistent guidelines and best practices for the accessibility and usage of individual genomic data collected by researchers.[38]
There is also controversy regarding the concerns with companies testing individual DNA. There are issues such as "leaking" information, the right to privacy and what responsibility the company has to ensure this does not happen. Regulation rules are not clearly laid out. What is still not determined is who legally owns the genome information: the company or the individual whose genome has been read. There have been published examples of personal genome information being exploited.[39] Additional privacy concerns, related to, e.g., genetic discrimination, loss of anonymity, and psychological impacts, have been increasingly pointed out by the academic community[39] as well as government agencies.[36]
Personalized genome utility
Full genome sequencing holds large promise in the world of healthcare in the potential of precise and personalized medical treatments. This use of genetic information to select appropriate drugs is known as pharmacogenomics. This technology may allow treatments to be catered to the individual and the certain genetic predispositions they may have (such as personalized chemotherapy).
At the same time, full sequencing of the genome can identify polymorphisms that are so rare that no conclusions may be drawn about their impact, creating uncertainty in the analysis of individual genomes, particularly in the context of clinical care. Czech medical geneticist Eva Machácková writes: "In some cases it is difficult to distinguish if the detected sequence variant is a causal mutation or a neutral (polymorphic) variation without any effect on phenotype. The interpretation of rare sequence variants of unknown significance detected in disease-causing genes becomes an increasingly important problem." [40] In fact, research has shown the average person is a carrier of 54 genetic mutations that are classified as lethal, yet present no negative effects in their health.[41]
Doctors are currently conducting tests for which some are not correctly trained to interpret the results. Many are unaware of how SNPs respond to one another. This results in presenting the client with potentially misleading and worrisome results which could strain the already overloaded health care system. This may antagonize the individual to make uneducated decisions such as unhealthy lifestyle choices and family planning modifications. Moreover, negative results which may potentially be inaccurate, theoretically decrease the quality of life and mental health of the individual (such as increased depression and extensive anxiety).
Direct-to-consumer genetics
There are also three potential problems associated with the validity of personal genome kits. The first issue is the test's validity. Handling errors of the sample increases the likelihood for errors which could affect the test results and interpretation. The second affects the clinical validity, which could affect the test's ability to detect or predict associated disorders. The third problem is the clinical utility of personal genome kits and associated risks, and the benefits of introducing them into clinical practices.[42]
People need to be educated on interpreting their results and what they should be rationally taking from the experience. It is not only the average person who needs to be educated in the dimensions of their own genomic sequence but also professionals, including physicians and science journalists, who must be provided with the knowledge required to inform and educate their patients and the public.[43][44][45] Examples of such efforts include the Personal Genetics Education Project (pgEd), the Smithsonian collaboration with NHGRI, and the MedSeq, BabySeq and MilSeq projects of Genomes to People, an initiative of Harvard Medical School and Brigham and Women's Hospital.
Popular culture
The 1997 science fiction film GATTACA presents a near-future society where personal genomics is readily available to anyone, and explores its societal impact.
See also
- Comparison of DNA sequencing services
- Human genome map
- Human Genome Project
- Single-nucleotide polymorphism
- Population genomics
- Full Genome Sequencing
- Bioinformatics
- Genomics
- Personalized medicine
- Systems biology
- Transcriptomics
- Omics
- Population groups in biomedicine
- Genomic counseling
- Personal medicine
References
- 1 2 "NCI Dictionary of Cancer Terms". National Cancer Institute. Retrieved 5 December 2016.
- ↑ "Predictive medicine - Latest research and news | Nature". www.nature.com. Retrieved 5 December 2016.
- ↑ MRC, Medical Research Council, (2016-03-03). "Stratified medicine". www.mrc.ac.uk. Retrieved 5 December 2016.
- ↑ Strausberg, Robert L.; Simpson, Andrew J. G.; Old, Lloyd J.; Riggins, Gregory J. (27 May 2004). "Oncogenomics and the development of new cancer therapies". Nature. 429 (6990): 469–474. Bibcode:2004Natur.429..469S. PMID 15164073. doi:10.1038/nature02627.
- 1 2 Reference, Genetics Home. "What is pharmacogenomics?". Genetics Home Reference. Retrieved 5 December 2016.
- ↑ Johnson, Julie A (5 December 2016). "Pharmacogenetics in clinical practice: how far have we come and where are we going?". Pharmacogenomics. 14 (7): 835–843. PMC 3697735
 . PMID 23651030. doi:10.2217/pgs.13.52.
. PMID 23651030. doi:10.2217/pgs.13.52. - ↑ Research, Center for Drug Evaluation and. "Genomics - Table of Pharmacogenomic Biomarkers in Drug Labeling". www.fda.gov. Retrieved 5 December 2016.
- ↑ "NIH Office of Rare Disease Research".
- ↑ "Gene Tests".
- ↑ Wetterstrand, Kris (21 May 2012). "DNA Sequencing Costs: Data from the NHGRI Large-Scale Genome Sequencing Program". Large-Scale Genome Sequencing Program. National Human Genome Research Institute. Retrieved 24 May 2012.
- ↑ "The Cost of Sequencing a Human Genome". National Human Genome Research Institute (NHGRI). Retrieved 5 December 2016.
- ↑ "JDW-genome-supp-mat-march-proof.doc" (PDF). Nature. 452: 872–876. doi:10.1038/nature06884. Retrieved 19 October 2011.
- ↑ "True Single Molecule Sequencing (tSMS): Helicos BioSciences". Helicosbio.com. Archived from the original on 4 October 2011. Retrieved 19 October 2011.
- ↑ "Knome Lowers Price of Full Genome From $350,000 to $99,000". The Genetic Genealogist.
- ↑ Karow, Julia (19 May 2009). "Knome Adds Exome Sequencing, Starts Offering Services to Researchers". GenomeWeb. Retrieved 24 February 2010.
- ↑ Harmon, Katherine (28 June 2010). "Genome Sequencing for the Rest of Us". Scientific American. Retrieved 13 August 2010.
- ↑ "Individual genome sequencing – Illumina, Inc.". Everygenome.com. Retrieved 19 October 2011.
- ↑ "Illumina Cutting Personal Genome Sequencing Price by 60% | GPlus.com". Glgroup.com. 4 June 2010. Retrieved 19 October 2011.
- ↑ Karow, Julia. "Complete Genomics to Offer $5,000 Human Genome as a Service Business in Q2 2009 | In Sequence | Sequencing". GenomeWeb. Retrieved 19 October 2011.
- ↑ Lauerman, John (5 February 2009). "Complete Genomics Drives Down Cost of Genome Sequence to $5,000". Bloomberg. Retrieved 19 October 2011.
- ↑
- ↑ "Illumina launches personal genome sequencing service for $48,000 : Genetic Future". Scienceblogs.com. Retrieved 19 October 2011.
- ↑ {http://www.healthcarejournallr.com/the-journal/contents-index/features/563-what-a-tangled-web-we-weave.html}
- ↑ Kaplan, Sarah (17 April 2016). "How do your 20,000 genes determine so many wildly different traits? They multitask.". The Washington Post. Retrieved 27 August 2016.
- ↑ Greshake, Bastian (2014). "openSNP–A Crowdsourced Web Resource for Personal Genomics". PLOS – via EBSCO.
- ↑ Zimmer, Carl. "Game of Genomes, Episode 13: Answers and Questions". STAT. Retrieved 2016-08-27.
- ↑ Corpas M, Valdivia-Granda W, Torres N, Greshake B, Coletta A, Knaus A, Harrison AP, Cariaso M, Moran F, Nielsen F, Swan D, Weiss Solis DY, Krawitz P, Schacherer F, Schols P, Yang H, Borry P, Glusman G, Robinson PN (Nov 2015). "Crowdsourced direct-to-consumer genomic analysis of a family quartet". BMC Genomics. 16 (910). PMC 4636840
 . PMID 26547235. doi:10.1186/s12864-015-1973-7.
. PMID 26547235. doi:10.1186/s12864-015-1973-7. - ↑ "Genetics and Health Insurance State Anti-Discrimination Laws".
- ↑ "Genetic Discrimination". National Human Genome Research Institute (NHGRI). Retrieved 5 December 2016.
- ↑ McCabe LL; McCabe ER (June 2001). "Postgenomic medicine. Presymptomatic testing for prediction and prevention". Clin Perinatol. 28 (2): 425–34. PMID 11499063. doi:10.1016/S0095-5108(05)70094-4.
- ↑ Nelson RM; Botkjin JR; Kodish ED; et al. (June 2001). "Ethical issues with genetic testing in pediatrics". Pediatrics. 107 (6): 1451–55. PMID 11389275. doi:10.1542/peds.107.6.1451.
- ↑ Borry P; Fryns JP; Schotsmans P; Dierickx K (February 2006). "Carrier testing in minors: a systematic review of guidelines and position papers". Eur. J. Hum. Genet. 14 (2): 133–8. PMID 16267502. doi:10.1038/sj.ejhg.5201509.
- ↑ Borry P; Stultiens L; Nys H; Cassiman JJ; et al. (November 2006). "Presymptomatic and predictive genetic testing in minors: a systematic review of guidelines and position papers". Clin. Genet. 70 (5): 374–81. PMID 17026616. doi:10.1111/j.1399-0004.2006.00692.x.
- ↑ Mark Johnson & Kathleen Gallagher (27 February 2011). "One in a Billion. Nic Volker case may be the leading edge of a wave moving across genetic medicine". Milwaukee Journal Sentinel.
- ↑ de Vries, Jantina; Bull, Susan J; Doumbo, Ogobara; Ibrahim, Muntaser; Mercereau-Puijalon, Odile; Kwiatkowski, Dominic; Parker, Michael (18 March 2011). "Ethical issues in human genomics research in developing countries". BMC Medical Ethics. 12: 5. PMC 3076260
 . PMID 21418562. doi:10.1186/1472-6939-12-5.
. PMID 21418562. doi:10.1186/1472-6939-12-5. - 1 2 3 "Privacy and Progress in Whole Genome Sequencing". Presidential Commission for the Study of Bioethical Issues. Retrieved 30 November 2016.
- ↑ Check Hayden, Erika (2013). "Privacy loophole found in genetic databases". Nature. doi:10.1038/nature.2013.12237.
- ↑ Gutmann, Amy; Wagner, James W. (2013-05-01). "Found Your DNA on the Web: Reconciling Privacy and Progress". Hastings Center Report. 43 (3): 15–18. doi:10.1002/hast.162.
- 1 2 De Cristofaro, Emiliano (2012-10-17). "Whole Genome Sequencing: Innovation Dream or Privacy Nightmare?". arXiv:1210.4820
 [cs.CR].
[cs.CR]. - ↑ Machácková, E. (2003-03-01). "Disease-causing mutations versus neutral polymorphism: use of bioinformatics and DNA diagnosis". Casopis Lekaru Ceskych. 142 (3): 150–153. PMID 12756842.
- ↑ Check Hayden, Erika (2016). "The flip side of personal genomics: When a mutation doesn't spell disease". Nature. doi:10.1038/nature.2016.20986.
- ↑ Hunter, David J.; Khoury, Muin J.; Drazen, Jeffrey M. (2008-01-10). "Letting the Genome out of the Bottle — Will We Get Our Wish?". New England Journal of Medicine. 358 (2): 105–107. PMID 18184955. doi:10.1056/NEJMp0708162.
- ↑ Lunshof, Jeantine; Mardis Elaine [Retrieved from http://www.future-science-group.com/_img/pics/Mardis_Forward.pdf "Navigenics - How it works"]. Future Medicine Magazine. Retrieved 30 March 2012/
- ↑ Roberts, J. Scott; Gornick, Michele C. et al [Retrieved from http://www.genomes2people.org/wp-content/uploads/2017/01/Roberts-et-al-PGen-PHG-2017.pdf "Direct-to-Consumer Genetic Testing: User Motivations, Decision Making, and Perceived Utility of Results"]. Public Health Genomics. Retrieved 20 February 2017/
- ↑ Corpas, Manuel (2012-01-06). "A Family Experience of Personal Genomics". Journal of Genetic Counseling. 21 (3): 368–391. PMID 22223063. doi:10.1007/s10897-011-9473-7.
Bibliography
- Dudley & Karczewski (2013). Exploring Personal Genomics. Oxford University Press. ISBN 978-0199644490.
- Sweet K; Michaelis R (May 2011). The Busy Physician's Guide to Genetics, Genomics and Personalized Medicine (1st ed.). Springer Scientific Press. ISBN 978-94-007-1147-1.
- Cadwalladr, Carole (8 June 2013). "What happened when I had my genome sequenced". The Guardian. Retrieved 10 July 2013.
- Manuel Corpas (2016). Perfect DNA. CreateSpace Independent Publishing Platform. ISBN 978-1539783725.