Oligodendrocyte progenitor cell
| Oligodendrocyte progenitor cell | |
|---|---|
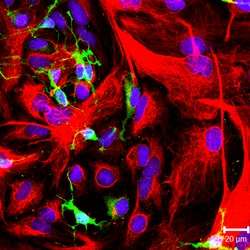 NG-2 positive oligodendrocyte progenitor cells (green color) and GFAP-positive astrocytes (red color) in culture. | |
| Details | |
| Identifiers | |
| Latin | Cellula oligodendrocytoprogenetrix |
| Code | TH H2.00.06.2.01007 |
| NeuroLex ID | Oligodendrocyte precursor cell |
Oligodendrocyte progenitor cells (OPCs), also known as oligodendrocyte precursor cells, NG2-glia or polydendrocytes, are a subtype of glial cells in the central nervous system.[1] They are precursors to oligodendrocytes and may also be able to differentiate into neurons and astrocytes. Differentiated oligodendrocytes support axons and provide electrical insulation in the form of a myelin sheath, enabling faster action potential propagation and high fidelity transmission without a need for an increase in axonal diameter.[2] The loss or lack of OPCs, and consequent lack of differentiated oligodendrocytes, is associated with a loss of myelination and subsequent impairment of neurological functions.[3]
Description
Oligodendrocyte progenitor cells are a subtype of glial cells in the central nervous system, characterized by expression of the proteoglycans PDGFRA, and CSPG4.[1] OPCs are smaller than neurons, of comparable size to other glia, and can either have a bipolar or complex multipolar morphology with processes reaching up to ~50 um.[4]
OPCs encompass approximately 3-4% of cells in the grey matter and 7-8% in white matter, making them the fourth largest group of glia after astrocytes, microglia and oligodendrocytes.[5]
OPCs are particularly prevalent in the hippocampus and in all layers of the neocortex.[6][7] In white matter, OPCs are found along unmyelinated axons [8] as well as along myelinated axons, engulfing nodes of Ranvier.[9][10] Recently, OPCs have been shown to reside in close contact with NG2-expressing pericytes in cerebral white matter, as well.[11]
OPCs have a remarkable homogenic distribution throughout the brain. This is achieved through active self-repulsion, causing the cells to be generally equally spaced from one another.[4][12] OPCs constantly survey their surroundings through actively extending and retracting growth cones. Death or differentiation of an OPC is rapidly followed by migration or local proliferation of a neighbouring cell.
OPCs receive synaptic contacts onto their processes from both glutamatergic[13] and GABAergic neurons.[1][14] OPCs receive preferred somatic contacts from fast-spiking GABAergic neurons, while non-fast spiking interneurons have a preference for contacting the processes.[15] These inhibitory connections (in mice) occur mainly during a specific period in development, from postnatal day 8 till postnatal day 13.
Function
OPCs were long held to function purely as progenitors to oligodendrocytes, hence the name. Later, additional functions were suggested.
The primary function is to serve as precursor for oligodendrocytes as well as some protoplasmic astrocytes in grey matter.[16] Postnatally, OPCs remain lineage-restricted and generally only differentiate into oligodendrocytes.
Whereas some studies suggested that OPCs can generate cortical neurons, other studies rejected these findings.[17] The question is unresolved, as studies continue to find that certain populations of OPCs can form neurons.[18]
OPCs synthesize the neuromodulatory factors prostaglandin D2 synthase (PTGDS) and neuronal pentraxin 2 (Nptx2).[19] This is mediated by the protein NG2, whose intracellular domain can be cleaved by the γ-secretase [20][21] and translocated to the nucleus.
The two N-terminal LNS (laminin/neurexin/sex hormone-binding globulin-domain) domains of the NG2 ectodomain can modulate signalling via AMPA and NMDA receptors of neuronal synapses within the cortex, including neuronal LTP. The NG2 ectodomain is released into the ECM from the full-length NG2 protein by constitutive and activity-dependent activity of the ADAM10 protease (α-secretase activity), showing that NG2 can modulate the neuronal glutamatergic system.[20][21]
Development
OPCs originate in the neuroepithelium of the spine and migrate to other areas of the brain.[22] Several waves of OPC production and migration lead to the generation of OLGs (oligodendrocytes).[23] OPCs are highly proliferative, migratory and bipolar.[24] The first wave of OPC production originates in the ganglionic eminence.
As development progresses a second and third wave of OPCs originate from the lateral and caudal ganglionic eminences and generate the majority of adult OLGs.[25] OPCs then migrate across most of the developing brain and spinal cord and eventually myelinate the entire central nervous system (CNS).[26] They differentiate into the less mobile, pro-oligodendrocytes that further differentiate into OLGs, a process characterized by the emergence of the expression of myelin basic protein (MBP), proteolipid protein (PLP), or myelin-associated glycoprotein (MAG).[24] Following terminal differentiation in vivo, mature OLGs wrap around and myelinate axons. In vitro, OLGs create an extensive network of myelin-like sheets. The process of differentiation can be observed both through morphological changes and cell surface markers specific to the discrete stage of differentiation, though the signals for differentiation are unknown.[27] The various waves of OPCs could myelinate distinct regions of the brain, which suggests that distinct functional subpopulations of OPCs perform different functions.[28]
OPC's are found in white and in matter. However, the higher of OPCs is higher in white matter than in gray matter because of a higher rate of proliferation in the former. White matter OPCs proliferate and contribute to adult oligodendrogenesis, while gray matter OPCs are slowly proliferative or quiescent and mostly remain in an immature state.[29] White and gray matter OPCs have different resting membrane potentials and ion channel expression. Gray matter lacks voltage-gated sodium channels while white matter does not and produces action potentials. Cells that produce action potentials can receive signals from other neurons.[30] These differences in OPC function depend on their locations.
Through maturation, OPCs are produced in the sub-ventricular zone (SVZ). The stem cells in the SVZ generate C cells which produce OPCs that go into the olfactory bulb.[31] The number of oligodendrocytes that are later formed depends on the part of the SVZ they came from. More OLGs are produced from the dorsal part of the SVZ than the ventrolateral part, and more are produced from the posterior than the rostral part.[32][33] This is due to differing environmental factors in these locations. The Wnt in the dorsal part favors OPC specification and the Bmp in the ventral part inhibits it.[34] These molecules help cause the expression of certain transcription factors.
Expression of Olig2 generates motor neurons and OPCs, dependent on the Shh and regulated by the Notch signaling pathway. This regulation limits the number of motor neurons and allows more OLGs to be produced.[35][36] Olig2 is one of the most important transcription factors involved in OLG production. Olig2 inactivation during development reduces OPC production.[37]
Differentiation of OPCs into OLGs involves massive reorganization of cytoskeleton proteins ultimately resulting in increased cell branching and lamella extension, allowing OLGs to myelinate multiple axons.[24] Laminin, a component of the extracellular matrix, plays an important role regulating OLG production. Mice lacking laminin alpha2-subunit produced fewer OPC's in the SVZ.[38] MicroRNA (miRNA) plays a role in the regulation of OLG differentiation and myelin maintenance. Deletion of Dicer1 in miRNA disrupts normal brain myelination. However, miR-7a, and miRNA in OPCs, promotes OPC production during brain development.[39]
Multiple pathways cause OLG branching, but their specific contributions have yet to be resolved and the process by which oligodendrocytes extend and wrap around multiple axons remains poorly understood.[24]
Endogenous remyelination
Spontaneous myelin repair was first observed in cat models.[40] It was later discovered to occur in the human CNS as well, specifically in cases of multiple sclerosis (MS).[41] Spontaneous myelin repair does not result in morphologically normal oligodendrocytes and is associated with thinner myelin compared to axonal diameter than normal myelin.[42] Despite morphological abnormalities, however, remyelination does restore normal conduction.[43] In addition, spontaneous remyelination does not appear to be rare, at least in the case of MS. Studies of MS lesions reported the average extent of remyelination as high as 47%.[44] Comparative studies of cortical lesions reported a greater proportion of remyelination in the cortex as opposed to white matter lesions.[41]
Cell types
Mature OLGs are unlikely to contribute to spontaneous remyelination even if they survive the original demyelinating injury.[45] New OLGs have been observed in areas of myelin damage, although the source of these new cells is unresolved. One possibility is that mature oligodendrocytes from uninjured areas migrate to the injury site and engage in myelination. This is unlikely because the transplantation of mature human OLGs achieve minimal myelin formation in the demyelinated rodent CNS. Another possibility is that mature OLGs de-differentiate into OPCs that then proliferate and remyelinate, Little experimental evidence supports this view.
Source of new oligodendrocytes
Some evidence suggests that the source of these new OLGs is proliferative adult OLG precursor cells. Such cells have been demonstrated to exist in the adult rodent[46] and human CNS.[47] These OLG precursor cells appear to play a major role in remyelination and are, unlike mature OLGs, able to cause extensive remyelination after transplantation into areas of myelin damage.[48] The role of these cells absent local demyelination, however, is under investigation. The fact that oligodendrocyte progenitors exhibit electrophysiological properties related to the expression of a range of glutamate receptors allowing communication with the neuron-axon unit suggests that OPCs are likely to have additional functions.[49]
The observation of OPCs in MS lesions that have not remyelinated suggested the hypothesis that the differentiation of these progenitors has been inhibited. One proposed mechanism involves the accumulation of myelin debris at the axon, suggesting that the inflammatory environment may be conducive to remyelination, as does the release of growth factors by inflammatory cells and activated microglia.[50] Alternatively, the accumulation of glycosaminoglycan hyaluronan at the lesion site may inhibit OPC differentiation. The release of OPC-specific antibodies by chronically demyelinated axons have been implicated as a remyelination inhibitor.[51] Other proposed mechanisms posit that OPC migration is inhibited by either molecules expressed by chronically demyelinated axons or the accumulation of unreactive astrocytes in MS lesions.[51]
Transplantation
OPC transplants contribute to remyelination, but it is difficult to maintain such cells in adequate concentrations at high purity. Finding a source for these cells remains impractical as of 2016. Should adult cells be used for transplantation, a brain biopsy would be required for each patient, adding to the risk of immune rejection. Embryonically derived stem cells have been demonstrated to carry out remyelination under laboratory conditions, but carry ethical implications. Adult central nervous system stem cells have also been shown to generate myelinating OLGs, but are not readily accessible.[52]
Even if a viable source of OPCS were found, identifying and monitoring the outcome of remyelination remains difficult, though multimodal measures of conduction velocity and emerging magnetic resonance imaging techniques offer improved sensitivity versus other imaging methods.[53] In addition, the interaction between transplanted cells and immune cells and the effect of inflammatory immune cells on remyelination have yet to be fully characterized. If the failure of endogenous remyelination is due to an unfavorable differentiation environment, then this will have to be addressed prior to transplantation.
References
- 1 2 3 Nishiyama, A.; Komitova, M.; Suzuki, R.; Zhu, X. (2009). "Polydendrocytes (NG2 cells): Multifunctional cells with lineage plasticity". Nature Reviews Neuroscience. 10 (1): 9–22. PMID 19096367. doi:10.1038/nrn2495.
- ↑ Swiss, V. A.; Nguyen, T.; Dugas, J.; Ibrahim, A.; Barres, B.; Androulakis, I. P.; Casaccia, P. (2011). Feng, Yue, ed. "Identification of a Gene Regulatory Network Necessary for the Initiation of Oligodendrocyte Differentiation". PLoS ONE. 6 (4): e18088. Bibcode:2011PLoSO...618088S. PMC 3072388
 . PMID 21490970. doi:10.1371/journal.pone.0018088.
. PMID 21490970. doi:10.1371/journal.pone.0018088. - ↑ Buller, B., Chopp, M., Ueno, Y., Zhang, L., Zhang, R. L., Morris, D., Zhang, Y. and Zhang, Z. G (2012). "Regulation of serum response factor by miRNA-200 and miRNA-9 modulates OPC differentiation". Glia. 60 (12): 1906–14. PMC 3474880
 . PMID 22907787. doi:10.1002/glia.22406.
. PMID 22907787. doi:10.1002/glia.22406. - 1 2 Hughes, E. G.; Kang, S. H.; Fukaya, M; Bergles, D. E. (2013). "Oligodendrocyte progenitors balance growth with self-repulsion to achieve homeostasis in the adult brain". Nature Neuroscience. 16 (6): 668–76. PMC 3807738
 . PMID 23624515. doi:10.1038/nn.3390.
. PMID 23624515. doi:10.1038/nn.3390. - ↑ Dawson, M. R.; Polito, A.; Levine, J. M.; Reynolds, R. (2003). "NG2-expressing glial progenitor cells: An abundant and widespread population of cycling cells in the adult rat CNS". Molecular and cellular neurosciences. 24 (2): 476–488. PMID 14572468. doi:10.1016/S1044-7431(03)00210-0.
- ↑ Ong, W. Y.; Levine, J. M. (1999). "A light and electron microscopic study of NG2 chondroitin sulfate proteoglycan-positive oligodendrocyte precursor cells in the normal and kainate-lesioned rat hippocampus". Neuroscience. 92 (1): 83–95. PMID 10392832. doi:10.1016/S0306-4522(98)00751-9.
- ↑ Bergles, D. E.; Jahr, C. E. (1997). "Synaptic Activation of Glutamate Transporters in Hippocampal Astrocytes". Neuron. 19 (6): 1297–1308. PMID 9427252. doi:10.1016/S0896-6273(00)80420-1.
- ↑ Ziskin, J. L.; Nishiyama, A.; Rubio, M.; Fukaya, M.; Bergles, D. E. (2007). "Vesicular release of glutamate from unmyelinated axons in white matter". Nature Neuroscience. 10 (3): 321–330. PMC 2140234
 . PMID 17293857. doi:10.1038/nn1854.
. PMID 17293857. doi:10.1038/nn1854. - ↑ Butt, A. M.; Duncan, A.; Hornby, M. F.; Kirvell, S. L.; Hunter, A.; Levine, J. M.; Berry, M. (1999). "Cells expressing the NG2 antigen contact nodes of Ranvier in adult CNS white matter". Glia. 26 (1): 84–91. PMID 10088675. doi:10.1002/(SICI)1098-1136(199903)26:1<84::AID-GLIA9>3.0.CO;2-L.
- ↑ Miller, R. H. (1996). "Oligodendrocyte origins". Trends in Neurosciences. 19 (3): 92–96. PMID 9054062. doi:10.1016/S0166-2236(96)80036-1.
- ↑ Maki, T; Maeda, M; Uemura, M; Lo, E. K.; Terasaki, Y; Liang, A. C.; Shindo, A; Choi, Y. K.; Taguchi, A; Matsuyama, T; Takahashi, R; Ihara, M; Arai, K (2015). "Potential interactions between pericytes and oligodendrocyte precursor cells in perivascular regions of cerebral white matter". Neuroscience Letters. 597: 164–9. PMC 4443478
 . PMID 25936593. doi:10.1016/j.neulet.2015.04.047.
. PMID 25936593. doi:10.1016/j.neulet.2015.04.047. - ↑ Birey, F; Aguirre, A (2015). "Age-Dependent Netrin-1 Signaling Regulates NG2+ Glial Cell Spatial Homeostasis in Normal Adult Gray Matter". Journal of Neuroscience. 35 (17): 6946–51. PMC 4412904
 . PMID 25926469. doi:10.1523/JNEUROSCI.0356-15.2015.
. PMID 25926469. doi:10.1523/JNEUROSCI.0356-15.2015. - ↑ Bergles, D. E.; Roberts, J. D. B.; Somogyi, P.; Jahr, C. E. (2000). "Glutamatergic synapses on oligodendrocyte precursor cells in the hippocampus". Nature. 405 (6783): 187–191. PMID 10821275. doi:10.1038/35012083.
- ↑ Steinhäuser, C.; Gallo, V. (1996). "News on glutamate receptors in glial cells". Trends in Neurosciences. 19 (8): 339–345. PMID 8843603. doi:10.1016/0166-2236(96)10043-6.
- ↑ Orduz, D; Maldonado, P. P.; Balia, M; Vélez-Fort, M; De Sars, V; Yanagawa, Y; Emiliani, V; Angulo, M. C. (2015). "Interneurons and oligodendrocyte progenitors form a structured synaptic network in the developing neocortex". ELife. 4. PMC 4432226
 . PMID 25902404. doi:10.7554/eLife.06953.
. PMID 25902404. doi:10.7554/eLife.06953. - ↑ Zhu, X.; Bergles, D. E.; Nishiyama, A. (2007). "NG2 cells generate both oligodendrocytes and gray matter astrocytes". Development. 135 (1): 145–157. PMID 18045844. doi:10.1242/dev.004895.
- ↑ Clarke, L. E.; Young, K. M.; Hamilton, N. B.; Li, H.; Richardson, W. D.; Attwell, D. (2012). "Properties and Fate of Oligodendrocyte Progenitor Cells in the Corpus Callosum, Motor Cortex, and Piriform Cortex of the Mouse". Journal of Neuroscience. 32 (24): 8173–8185. PMC 3378033
 . PMID 22699898. doi:10.1523/JNEUROSCI.0928-12.2012.
. PMID 22699898. doi:10.1523/JNEUROSCI.0928-12.2012. - ↑ Tsoa, R. W.; Coskun, V; Ho, C. K.; De Vellis, J; Sun, Y. E. (2014). "Spatiotemporally different origins of NG2 progenitors produce cortical interneurons versus glia in the mammalian forebrain". Proceedings of the National Academy of Sciences. 111 (20): 7444–9. Bibcode:2014PNAS..111.7444T. PMC 4034245
 . PMID 24799701. doi:10.1073/pnas.1400422111.
. PMID 24799701. doi:10.1073/pnas.1400422111. - ↑ Sakry, D; Yigit, H; Dimou, L; Trotter, J (2015). "Oligodendrocyte precursor cells synthesize neuromodulatory factors". PLoS ONE. 10 (5): e0127222. PMC 4429067
 . PMID 25966014. doi:10.1371/journal.pone.0127222.
. PMID 25966014. doi:10.1371/journal.pone.0127222. - 1 2 Sakry, D; Trotter, J (2015). "The role of the NG2 proteoglycan in OPC and CNS network function". Brain Research. 1638 (Pt B): 161–6. PMID 26100334. doi:10.1016/j.brainres.2015.06.003.
- 1 2 Sakry, D; Neitz, A; Singh, J; Frischknecht, R; Marongiu, D; Binamé, F; Perera, S. S.; Endres, K; Lutz, B; Radyushkin, K; Trotter, J; Mittmann, T (2014). "Oligodendrocyte precursor cells modulate the neuronal network by activity-dependent ectodomain cleavage of glial NG2". PLoS Biology. 12 (11): e1001993. PMC 4227637
 . PMID 25387269. doi:10.1371/journal.pbio.1001993.
. PMID 25387269. doi:10.1371/journal.pbio.1001993. - ↑ Donna J. Osterhout; Amy Wolven; Rebecca M. Wolf; Marilyn D. Resh & Moses V. Chao (1999). "Morphological Differentiation of Oligodendrocytes Requires Activation of Fyn Tyrosine Kinase" (PDF). Journal of Cell Biol.
- ↑ Spassky, N; Olivier, C; Cobos, I; Lebras, B; Goujet-Zalc, C; Martínez, S; Zalc, B; Thomas, J. L. (2001). "The early steps of oligodendrogenesis: Insights from the study of the plp lineage in the brain of chicks and rodents". Developmental neuroscience. 23 (4–5): 318–26. PMID 11756747. doi:10.1159/000048715.
- 1 2 3 4 Pfeiffer, Steve E. Warrington, Art E. Bansal, Rashmi. (1993). "The oligodendrocyte and its many cellular processes". Trends in Cell Biology. 3 (6): 191–7. PMID 14731493. doi:10.1016/0962-8924(93)90213-K.
- ↑ Kessaris, N.; Fogarty, M.; Iannarelli, P.; Grist, M.; Wegner, M.; Richardson, W. D. (2005). "Competing waves of oligodendrocytes in the forebrain and postnatal elimination of an embryonic lineage". Nature Neuroscience. 9 (2): 173–179. PMID 16388308. doi:10.1038/nn1620.
- ↑ El Waly, B; MacChi, M; Cayre, M; Durbec, P (2014). "Oligodendrogenesis in the normal and pathological central nervous system". Frontiers in Neuroscience. 8: 145. PMC 4054666
 . PMID 24971048. doi:10.3389/fnins.2014.00145.
. PMID 24971048. doi:10.3389/fnins.2014.00145. - ↑ Wang, H.; Rusielewicz, T.; Tewari, A.; Leitman, E. M.; Einheber, S.; Melendez-Vasquez, C. V. (2012). "Myosin II is a negative regulator of oligodendrocyte morphological differentiation". Journal of Neuroscience Research. 90 (8): 1547–1556. PMC 3370114
 . PMID 22437915. doi:10.1002/jnr.23036.
. PMID 22437915. doi:10.1002/jnr.23036. - ↑ Tripathi, R. B.; Clarke, L. E.; Burzomato, V; Kessaris, N; Anderson, P. N.; Attwell, D; Richardson, W. D. (2011). "Dorsally and ventrally derived oligodendrocytes have similar electrical properties but myelinate preferred tracts". Journal of Neuroscience. 31 (18): 6809–19. PMC 4227601
 . PMID 21543611. doi:10.1523/JNEUROSCI.6474-10.2011.
. PMID 21543611. doi:10.1523/JNEUROSCI.6474-10.2011. - ↑ Dimou, L.; Simon, C.; Kirchhoff, F.; Takebayashi, H.; Gotz, M. (2008). "Progeny of Olig2-Expressing Progenitors in the Gray and White Matter of the Adult Mouse Cerebral Cortex". Journal of Neuroscience. 28 (41): 10434–10442. PMID 18842903. doi:10.1523/JNEUROSCI.2831-08.2008.
- ↑ Káradóttir, R; Hamilton, N. B.; Bakiri, Y; Attwell, D (2008). "Spiking and nonspiking classes of oligodendrocyte precursor glia in CNS white matter". Nature Neuroscience. 11 (4): 450–6. PMC 2615224
 . PMID 18311136. doi:10.1038/nn2060.
. PMID 18311136. doi:10.1038/nn2060. - ↑ Doetsch, F; Caillé, I; Lim, D. A.; García-Verdugo, J. M.; Alvarez-Buylla, A (1999). "Subventricular zone astrocytes are neural stem cells in the adult mammalian brain". Cell. 97 (6): 703–16. PMID 10380923. doi:10.1016/s0092-8674(00)80783-7.
- ↑ Ortega, F; Gascón, S; Masserdotti, G; Deshpande, A; Simon, C; Fischer, J; Dimou, L; Chichung Lie, D; Schroeder, T; Berninger, B (2013). "Oligodendrogliogenic and neurogenic adult subependymal zone neural stem cells constitute distinct lineages and exhibit differential responsiveness to Wnt signalling". Nature Cell Biology. 15 (6): 602–13. PMID 23644466. doi:10.1038/ncb2736.
- ↑ Menn, B; Garcia-Verdugo, J. M.; Yaschine, C; Gonzalez-Perez, O; Rowitch, D; Alvarez-Buylla, A (2006). "Origin of oligodendrocytes in the subventricular zone of the adult brain". Journal of Neuroscience. 26 (30): 7907–18. PMID 16870736. doi:10.1523/JNEUROSCI.1299-06.2006.
- ↑ Colak, D; Mori, T; Brill, M. S.; Pfeifer, A; Falk, S; Deng, C; Monteiro, R; Mummery, C; Sommer, L; Götz, M (2008). "Adult neurogenesis requires Smad4-mediated bone morphogenic protein signaling in stem cells". Journal of Neuroscience. 28 (2): 434–46. PMID 18184786. doi:10.1523/JNEUROSCI.4374-07.2008.
- ↑ Dessaud, E; Ribes, V; Balaskas, N; Yang, L. L.; Pierani, A; Kicheva, A; Novitch, B. G.; Briscoe, J; Sasai, N (2010). "Dynamic assignment and maintenance of positional identity in the ventral neural tube by the morphogen sonic hedgehog". PLoS Biology. 8 (6): e1000382. PMC 2879390
 . PMID 20532235. doi:10.1371/journal.pbio.1000382.
. PMID 20532235. doi:10.1371/journal.pbio.1000382. - ↑ Kim, H; Shin, J; Kim, S; Poling, J; Park, H. C.; Appel, B (2008). "Notch-regulated oligodendrocyte specification from radial glia in the spinal cord of zebrafish embryos". Developmental Dynamics. 237 (8): 2081–9. PMC 2646814
 . PMID 18627107. doi:10.1002/dvdy.21620.
. PMID 18627107. doi:10.1002/dvdy.21620. - ↑ Lu, Q. R.; Sun, T; Zhu, Z; Ma, N; Garcia, M; Stiles, C. D.; Rowitch, D. H. (2002). "Common developmental requirement for Olig function indicates a motor neuron/oligodendrocyte connection". Cell. 109 (1): 75–86. PMID 11955448. doi:10.1016/s0092-8674(02)00678-5.
- ↑ Relucio, J; Menezes, M. J.; Miyagoe-Suzuki, Y; Takeda, S; Colognato, H (2012). "Laminin regulates postnatal oligodendrocyte production by promoting oligodendrocyte progenitor survival in the subventricular zone". Glia. 60 (10): 1451–67. PMID 22706957. doi:10.1002/glia.22365.
- ↑ Zhao, X; He, X; Han, X; Yu, Y; Ye, F; Chen, Y; Hoang, T; Xu, X; Mi, Q. S.; Xin, M; Wang, F; Appel, B; Lu, Q. R. (2010). "MicroRNA-mediated control of oligodendrocyte differentiation". Neuron. 65 (5): 612–26. PMC 2855245
 . PMID 20223198. doi:10.1016/j.neuron.2010.02.018.
. PMID 20223198. doi:10.1016/j.neuron.2010.02.018. - ↑ Bunge, Mary Bartlett; Bunge, Richard P.; Ris, Hans (1961). "Ultrastructural Study of Remyelination in an Experimental Lesion in Adult Cat Spinal Cord". Journal of the Neurological Sciences. 10: 67–94. PMC 2225064
 . PMID 13688845. doi:10.1083/jcb.10.1.67.
. PMID 13688845. doi:10.1083/jcb.10.1.67. - 1 2 Pierre, O. & Gregoire, A. (1965). "Electron Microscopic Features of Multiple Sclerosis Lesions". Brain. 88 (5): 937–52. PMID 5864468. doi:10.1093/brain/88.5.937.
- ↑ Blakemore, W.F. (1974). "Pattern of remyelination in the CNS". Nature. 249 (5457): 577–578. Bibcode:1974Natur.249..577B. doi:10.1038/249577a0.
- ↑ Smith, K.J., Bostock, H. Hall, S.M. (1982). "Saltatory conduction precedes remyelination in axons demyelinated with lysophosphatidyl choline". Journal of the Neurological Sciences. 54 (1): 13–31. PMID 6804606. doi:10.1016/0022-510X(82)90215-5.
- ↑ Albert, Monika Antel, Jack Bruic, Wolfgang Stadelmann, Christine (2007). "Extensive Remyelination in patients with Chronic Multiple Sclerosis". Brain Pathology. 17 (2): 129–38. PMID 17388943. doi:10.1111/j.1750-3639.2006.00043.x.
- ↑ Keirstead, H.S., Blakemore, W.F. (1997). "Identification of post-mitotic oligodendrocytes incapable of remyelination within the demyelinated adult spinal cord". J Neuropathol Exp Neurol. 56 (11): 1191–201. ISSN 0022-3069. PMID 9370229. doi:10.1097/00005072-199711000-00003.
- ↑ Ffrench-Constant, C. Raff, M. C. (1986). "Proliferating bipotential glial progenitor cells in adult rat optic nerve". Nature. 319 (6053): 499–502. Bibcode:1986Natur.319..499F. PMID 3945333. doi:10.1038/319499a0.
- ↑ Scolding, N. J. Rayner, P. J. Sussman, J. Shaw, C. Compston, D. A. (1995). "A proliferative adult human oligodendrocyte progenitor". NeuroReport. 6 (3): 441–5. ISSN 0959-4965. PMID 7766839. doi:10.1097/00001756-199502000-00009.
- ↑ Zhang, S. C. Ge, B. Duncan, I. D.; Ge; Duncan (1999). "Adult brain retains the potential to generate oligodendroglial progenitors with extensive myelination capacity". Proc Natl Acad Sci U S A. 96 (7): 4089–94. Bibcode:1999PNAS...96.4089Z. ISSN 0027-8424. PMC 22425
 . PMID 10097168. doi:10.1073/pnas.96.7.4089.
. PMID 10097168. doi:10.1073/pnas.96.7.4089. - ↑ Luyt, K. Varadi, A. Halfpenny, C. A. Scolding, N. J. Molnar, E. (2004). "Metabotropic glutamate receptors are expressed in adult human glial progenitor cells". Biochem Biophys Res Commun. 319 (1): 120–9. PMID 15158450. doi:10.1016/j.bbrc.2004.04.158.
- ↑ Heese, K. Hock, C. Otten, U. (1998). "Inflammatory signals induce neurotrophin expression in human microglial cells". J Neurochem. 70 (2): 699–707. ISSN 0022-3042. PMID 9453564. doi:10.1046/j.1471-4159.1998.70020699.x.
- 1 2 Niehaus, A. Shi, J. Grzenkowski, M. Diers-Fenger, M. Archelos, J. Hartung, H. P. Toyka, K. Bruck, W. Trotter, J. (2000). "Patients with active relapsing-remitting multiple sclerosis synthesize antibodies recognizing oligodendrocyte progenitor cell surface protein: implications for remyelination". Annals of Neurology. 48 (3): 362–71. ISSN 0364-5134. PMID 10976643. doi:10.1002/1531-8249(200009)48:3<362::AID-ANA11>3.0.CO;2-6.
- ↑ Lakatos, A. Franklin, R. J. Barnett, S. C. (2000). "Olfactory ensheathing cells and Schwann cells differ in their in vitro interactions with astrocytes". Glia. 32 (3): 214–25. ISSN 0894-1491. PMID 11102963. doi:10.1002/1098-1136(200012)32:3<214::AID-GLIA20>3.0.CO;2-7.
- ↑ Behrens, T. E. Johansen-Berg, H. Woolrich, M. W. Smith, S. M. Wheeler-Kingshott, C. A. Boulby, P. A. Barker, G. J. Sillery, E. L. Sheehan, K. Ciccarelli, O. Thompson, A. J. Brady, J. M. Matthews, P. M. (2003). "Non-invasive mapping of connections between human thalamus and cortex using diffusion imaging". Annals of Neurology. 6 (7): 750–757. doi:10.1038/nn1075.