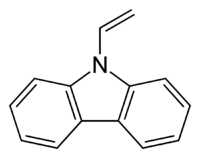
''N''-Vinylcarbazole
 | |
 | |
| Names | |
|---|---|
| IUPAC name
9-ethenyl-9H-carbazole | |
| Other names
9-vinyl-9H-carbazole, NVC | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.014.596 |
| EC Number | 216-055-0 |
| PubChem CID |
|
| RTECS number | FE6350000 |
| |
| |
| Properties | |
| C14H11N | |
| Molar mass | 193.244 g mol−1 |
| Appearance | pale brown crystalline solid[2] |
| Melting point | 66 °C (151 °F; 339 K) |
| Boiling point | 154 to 155 °C (309 to 311 °F; 427 to 428 K) 3 mmHg[2] |
| insoluble | |
| Solubility in diethyl ether | very soluble |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
N-Vinylcarbazole is an organic compound used as a monomer in the production of poly(vinylcarbazole),[3] a conductive polymer, in which conductivity is photon-dependent. The compound is used in the photoreceptors of photocopiers.[4] Upon exposure to γ-irradiation, N-vinylcarbazole undergoes solid-state polymerisation.[5]
Synthesis
One possible synthesis, patented by Nippon Shokubai, is based on the reaction of carbazole with ethylene carbonate at 300 °C (572 °F)-500 °C (932 °F) under partial vacuum and in the presence of alkaline earth metal and alkaline metal catalyst and silica. This reaction produces N-(2-Hydroxyethyl) carbazole, which in turn generates vinylcarbazole through water eliminiation.[3]
Related compounds
References
- ↑ Lide, David R. (2008). CRC Handbook of Chemistry and Physics, 89th Edition. CRC Press. pp. 3–518. ISBN 978-0-8493-0488-0.
- 1 2 Sigma-Aldrich 9-Vinylcarbazole product page
- 1 2 Conti, Francsco (June 2006). "Nuova via di sintesi del vinilcarbazolo". La Chimica & L'Industria (in Italian). Società Chimica Italiana (5): 82.
- ↑ G. Burton; J. Holman; J. Lazonby; G. Pilling; D. Waddington (2000). Chemical Storylines (2nd ed.). Heinemann Educational Publishers. pp. 121–122. ISBN 0-435-63119-5.
- ↑ K. Tsutsui; K. Hirotsu; M. Umesaki; M. Kurahashi; A. Shimada; T. Higuchi (1976). "Structural chemistry of polymerizable monomers. I. Crystal structure of N-vinylcarbazole". Acta Crystallogr. B. 32: 3049–3053. doi:10.1107/S0567740876009527.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.