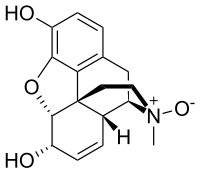
Morphine-''N''-oxide
Morphine-N-oxide
 |
| Names |
| IUPAC name
(4R,4aR,7S,7aR,12bS)-3-Methyl-2,3,4,4a,7,7a-hexahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinoline-7,9-diol 3-oxide |
| Identifiers |
| |
|
| |
|
| ChemSpider |
|
| ECHA InfoCard |
100.010.324 |
| EC Number |
211-355-8 |
| KEGG |
|
| |
|
InChI=1S/C17H19NO4/c1-18(21)7-6-17-10-3-5-13(20)16(17)22-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-,18?/m0/s1  Y YKey: AMAPEXTUMXQULJ-APQDOHRLSA-N  Y Y
|
CN1(=O)CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O CN1(=O)CC[C@]23[C@H]4OC5=C(O)C=CC(C[C@@H]1[C@@H]2C=C[C@@H]4O)=C35
|
| Properties |
| |
C17H19NO4 |
| Molar mass |
301.34 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
|
| Infobox references |
|
|
Morphine-N-oxide (genomorphine) is an active opioid metabolite of morphine. Morphine itself, in trials with rats, acts 11–22 times more potent than morphine-N-oxide subcutaneously and 39–89 times more potent intraperitoneally. However, pretreatment with amiphenazole or tacrine increases the potency of morphine-N-oxide in relation to morphine (intraperitoneally more so than in subcutaneous administration). A possible explanation is that morphine-N-oxide is rapidly inactivated in the liver and impairment of inactivation processes or enzymes increases functionality.[1]
Morphine-N-oxide can also form as a decomposition product of morphine outside the body and may show up in assays of opium and poppy straw concentrate. Codeine and the semi-synthetics such as heroin, dihydrocodeine, dihydromorphine, hydromorphone, and hydrocodone also have equivalent amine oxide derivatives.
Morphine-N-Oxide has a DEA ACSCN of 9307 and annual production quota of 655 grammes in 2013. It is a Schedule I controlled substance in the US.[2]
See also
References
|
|---|
| MOR |
- PAMs: BMS-986121
- BMS-986122
|
|---|
| DOR | |
|---|
| KOR |
- Agonists: 6'-GNTI
- 8-CAC
- 18-MC
- 14-Methoxymetopon
- β-Chlornaltrexamine
- β-Funaltrexamine
- Adrenorphin (metorphamide)
- Akuuamicine
- Alazocine (SKF-10047)
- Allomatrine
- Apadoline
- Asimadoline
- BAM-12P
- BAM-18P
- BAM-22P
- Big dynorphin
- Bremazocine
- BRL-52537
- Butorphan
- Butorphanol
- BW-373U86
- Cebranopadol
- Ciprefadol
- CR665
- Cyclazocine
- Cyclorphan
- Cyprenorphine
- Diamorphine (heroin)
- Diacetylnalorphine
- Difelikefalin
- Dihydroetorphine
- Dihydromorphine
- Diprenorphine
- Dynorphin A
- Dynorphin B (rimorphin)
- Eluxadoline
- Enadoline
- Eptazocine
- Erinacine E
- Ethylketazocine
- Etorphine
- Fedotozine
- Fentanyl
- Gemazocine
- GR-89696
- GR-103545
- Hemorphin-4
- Herkinorin
- HS665
- Hydromorphone
- HZ-2
- Ibogaine
- ICI-199,441
- ICI-204,448
- Ketamine
- Ketazocine
- Laudanosine
- Leumorphin (dynorphin B-29)
- Levallorphan
- Levomethorphan
- Levorphanol
- Lexanopadol
- Lofentanil
- LPK-26
- Lufuradom
- Matrine
- MB-1C-OH
- Menthol
- Metazocine
- Metkefamide
- Mianserin
- Mirtazapine
- Morphine
- Moxazocine
- MR-2034
- N-MPPP
- Nalbuphine
- Nalbuphine sebacate
- NalBzOH
- Nalfurafine
- Nalmefene
- Nalodeine (N-allylnorcodeine)
- Nalorphine
- Naltriben
- Niravoline
- Norbuprenorphine
- Norbuprenorphine-3-glucuronide
- Noribogaine
- Norketamine
- O-Desmethyltramadol
- Oripavine
- Oxilorphan
- Oxycodone
- Pentazocine
- Pethidine (meperidine)
- Phenazocine
- Proxorphan
- Racemethorphan
- Racemorphan
- RB-64
- Salvinorin A (salvia)
- Salvinorin B ethoxymethyl ether
- Salvinorin B methoxymethyl ether
- Samidorphan
- Spiradoline (U-62,066)
- TH-030418
- Thienorphine
- Tifluadom
- Tricyclic antidepressants (e.g., amitriptyline, desipramine, imipramine, nortriptyline)
- U-50,488
- U-54,494A
- U-69,593
- Xorphanol
|
|---|
| NOP | |
|---|
| Unsorted | |
|---|
| Others |
- Others: Kyotorphin (met-enkephalin releaser/degradation stabilizer)
|
|---|
See also: Receptor/signaling modulators • Signaling peptide/protein receptor modulators |

 . PMID 5687589. doi:10.1111/j.1476-5381.1968.tb07055.x.
. PMID 5687589. doi:10.1111/j.1476-5381.1968.tb07055.x.