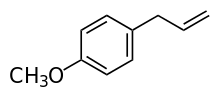
Estragole
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Methoxy-4-(prop-2-en-1-yl)benzene | |
| Other names
1-Methoxy-4-(2-propenyl)-benzene 1-Allyl-4-methoxybenzene Estragol Estragon p-Allylanisole Chavicyl methylether Methylchavicol Chavicol methylether Isoanethole 4-Allylanisole | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.004.935 |
| KEGG | |
| UNII | |
| |
| |
| Properties | |
| C10H12O | |
| Molar mass | 148.20 g/mol |
| Density | 0.946 g/cm3 |
| Boiling point | 216 °C (421 °F; 489 K) (95–96 °C at 12 mmHg) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Estragole (p-allylanisole, methyl chavicol) is a phenylpropene, a natural organic compound. Its chemical structure consists of a benzene ring substituted with a methoxy group and a propenyl group. It is an isomer of anethole, differing with respect to the location of the double bond. It is a colorless liquid, although impure samples can appear yellow. It is a component of various trees and plants, including turpentine (pine oil), anise, fennel, bay, tarragon, and basil. It is used in the preparation of fragrances.[1]
Production
Hundreds of tonnes of basil oil are produced annually by steam distillation of Ocimum basilicum (common basil). This oil is mainly estragole but also contains substantial amounts of linalool. Estragole is the primary constituent of essential oil of tarragon (comprising 60–75%), pine oil, turpentine, fennel, anise (2%[2]), Clausena anisata and Syzygium anisatum.
Estragole is used in perfumes and as a food additive for flavor. Upon treatment with potassium hydroxide, estragole converts to anethole.[1] A known use of estragole is in the synthesis of magnolol.
Safety
Estragole is suspected to be carcinogenic and genotoxic, as is indicated by a report of the European Union, Committee on Herbal Medicinal Products.[3] Several studies have clearly established that the profiles of metabolism, metabolic activation, and covalent binding are dose dependent and that the relative importance diminishes markedly at low levels of exposure (i.e. these events are not linear with respect to dose). In particular, rodent studies show that these events are minimal probably in the dose range of 1–10 mg/kg body weight, which is approximately 100-1000 times the anticipated human exposure to this substance. For these reasons it is concluded that the present exposure to estragole resulting from consumption of herbal medicinal products (short time use in adults at recommended posology) does not pose a significant cancer risk. In the meantime exposure of estragole to sensitive groups such as young children, pregnant and breastfeeding women should be minimized.
The Scientific Committee on Food from the Health & Consumer Protection Directorate-General took a more concerned position and concluded that "Estragole has been demonstrated to be genotoxic and carcinogenic. Therefore the existence of a threshold cannot be assumed and the Committee could not establish a safe exposure limit. Consequently, reductions in exposure and restrictions in use levels are indicated."[4]
See also
References
- 1 2 Karl-Georg Fahlbusch, Franz-Josef Hammerschmidt, Johannes Panten, Wilhelm Pickenhagen, Dietmar Schatkowski, Kurt Bauer, Dorothea Garbe, Horst Surburg "Flavors and Fragrances" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim: 2002. Published online: 15 January 2003; doi:10.1002/14356007.a11_141.
- ↑ Philip R. Ashurst (1999). chavicol Food Flavorings Check
|url=value (help). Springer. p. 11. - ↑ EMEA/HMPC/137212/2005, Committee on Herbal Medicinal Products. Final Public Statement on the Use of Herbal Medicinal Products Containing Estragole http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2010/04/WC500089960.pdf
- ↑ SCF/CS/FLAV/FLAVOUR/6, 26 September 2001, Opinion of the Scientific Committee on Food on Estragole (1-Allyl-4-methoxybenzene)http://ec.europa.eu/food/fs/sc/scf/out104_en.pdf