Metachromatic leukodystrophy
| Metachromatic leukodystrophy | |
|---|---|
 | |
| Sulfatide | |
| Classification and external resources | |
| Specialty | endocrinology |
| ICD-10 | E75.25 |
| ICD-9-CM | 330.0 |
| OMIM | 250100 |
| DiseasesDB | 8080 |
| MedlinePlus | 001205 |
| eMedicine | ped/2893 |
| MeSH | D007966 |
Metachromatic leukodystrophy (MLD, also called arylsulfatase A deficiency) is a lysosomal storage disease which is commonly listed in the family of leukodystrophies as well as among the sphingolipidoses as it affects the metabolism of sphingolipids. Leukodystrophies affect the growth and/or development of myelin, the fatty covering which acts as an insulator around nerve fibers throughout the central and peripheral nervous systems. MLD involves cerebroside sulfate accumulation.[1][2] Metachromatic leukodystrophy, like most enzyme deficiencies, has an autosomal recessive inheritance pattern.[2]
Signs and symptoms
Like many other genetic disorders that affect lipid metabolism, there are several forms of MLD, which are late infantile, juvenile, and adult.
- In the late infantile form, which is the most common form of MLD (50–60%), affected children begin having difficulty walking after the first year of life, usually at 15–24 months. Symptoms include muscle wasting and weakness, muscle rigidity, developmental delays, progressive loss of vision leading to blindness, convulsions, impaired swallowing, paralysis, and dementia. Children may become comatose. Untreated, most children with this form of MLD die by age 5, often much sooner.
- Children with the juvenile form of MLD (onset between 3 and 10 years of age) usually begin with impaired school performance, mental deterioration, and dementia and then develop symptoms similar to the late infantile form but with slower progression. Age of death is variable, but normally within 10 to 15 years of symptom onset although some juveniles can live for several decades or longer after onset.
- The adult form commonly begins after age 16 often with an onset in the 4th or 5th decade of life and presents as a psychiatric disorder or progressive dementia. Adult-onset MLD usually progresses more slowly than the late infantile and juvenile forms, with a protracted course of a decade or more.
Palliative care can help with many of the symptoms and usually improves quality of life and longevity.
Carriers have low enzyme levels compared to their family population ("normal" levels vary from family to family) but even low enzyme levels are adequate to process the body's sulfatide.
Causes
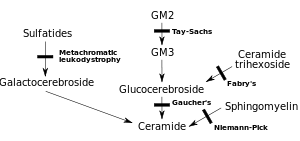
MLD is directly caused by a deficiency of the enzyme arylsulfatase A[3] (ARSA) and is characterized by enzyme activity in leukocytes that is less than 10% of normal controls.[4] However, assay of the ARSA enzyme activity alone is not sufficient for diagnosis; ARSA pseudodeficiency, which is characterized by enzyme activity that is 5~20% of normal controls does not cause MLD.[4] Without this enzyme, sulfatides build up in many tissues of the body, eventually destroying the myelin sheath of the nervous system. The myelin sheath is a fatty covering that protects nerve fibers. Without it, the nerves in the brain (central nervous system – CNS) and the peripheral nerves (peripheral nervous system – PNS) which control, among other things the muscles related to mobility, cease to function properly.
Arylsulfatase A is activated by saposin B (Sap B), a non-enzymatic proteinaceous cofactor.[5] When the arylsulfatase A enzyme level is normal but the sulfatides are still high – meaning that they are not being broken down because the enzyme is not activated – the resulting disease is saposin B deficiency, which presents similar to MLD.[4] Saposin B Deficiency is very rare, much more rare than traditional MLD.[4] The enzyme that is present is not "enabled" to a normal level of efficiency and can't break down the sulfatides which results in all of the same MLD symptoms and progression.[6]
A recent study contended sulfatide is not completely responsible for MLD because it is nontoxic. It has been suggested lysosulfatide, sulfatide which has had its acyl group removed, plays a role because of its cytotoxic properties in vitro.[7]
Genetics

MLD has an autosomal recessive inheritance pattern. The inheritance probabilities per birth are as follows:
- If both parents are carriers:
- 25% (1 in 4) children will have the disease
- 50% (2 in 4) children will be carriers, but unaffected
- 25% (1 in 4) children will be free of MLD – unaffected child that is not a carrier
- If one parent is affected and one is free of MLD:
- 0% (0) children will have the disorder – only one parent is affected, other parent always gives normal gene
- 100% (4 in 4) children will be carriers (but unaffected)
- If one parent is a carrier and the other is free of MLD:
- 50% (2 in 4) children will be carriers (but unaffected)
- 50% (2 in 4) children will be free of MLD – unaffected child that is not a carrier
In addition to these frequencies there is a 'pseudo'-deficiency that affects 7–15% of the population.[8][9] People with the pseudo deficiency do not have any MLD problems unless they also have affected status. With the current diagnostic tests, Pseudo-deficiency reports as low enzyme levels but sulfatide is processed normally so MLD symptoms do not exist. This phenomenon wreaks havoc with traditional approaches to Newborn Screening so new screening methods are being developed.
For further information, see recessive gene and dominance relationship. Also, consult the MLD genetics page at MLD Foundation.
Diagnosis
Clinical examination and MRI are often the first steps in a MLD diagnosis. MRI can be indicative of MLD, but is not adequate as a confirming test.
An ARSA-A enzyme level blood test with a confirming urinary sulfatide test is the best biochemical test for MLD. The conforming urinary sulfatide is important to distinguish between MLD and pseudo-MLD blood results.
Genomic sequencing may also confirm MLD, however, there are likely more mutations than the over 200 already know which cause MLD that are not yet ascribed to MLD that cause MLD so in those cases a biochemical test is still warranted.
For further information, see the MLD Testing page at MLD Foundation.
Treatment
There is currently no therapy or cure for MLD in late infantile patients displaying symptoms, or for juvenile and adult onset with advanced symptoms. These patients typically receive clinical treatment focused on pain and symptom management.
Pre-symptomatic late infantile MLD patients, as well as those with juvenile or adult MLD that are either presymptomatic or displaying mild symptoms, can consider bone marrow transplantation (including stem cell transplantation), which may slow down progression of the disease in the central nervous system. However, results in the peripheral nervous system have been less dramatic, and the long-term results of these therapies have been mixed. Recent success has involved stem cells being taken from the bone marrow of children with the disorder and infecting the cells with a retro-virus, replacing the stem cells' mutated gene with the repaired gene before re-injecting it back into the patient where they multiplied. The children by the age of five were all in good condition and going to kindergarten when normally by this age, children with the disease can not even speak.[10]
Several therapy options are currently being investigated using clinical trials primarily in late infantile patients. These therapies include gene therapy, enzyme replacement therapy (ERT), substrate reduction therapy (SRT), and potentially enzyme enhancement therapy (EET).
A team of international researchers and foundations gathered in 2008 to form an international MLD Registry to create and manage a shared repository of knowledge, including the natural history of MLD. This consortium consisted of scientific, academic and industry resources. This registry never became operational.
Epidemiology
The incidence of metachromatic leukodystrophy is estimated to occur in 1 in 40,000 to 1 in 160,000 individuals worldwide.[11] There is a much higher incidence in certain genetically isolated populations, such as 1 in 75 in Habbanites (a small group of Jews who immigrated to Israel from southern Arabia), 1 in 2,500 in the western portion of the Navajo Nation, and 1 in 8,000 among Arab groups in Israel.[11]
As an autosomal recessive disease, 1 in 40,000 equates to a 1 in 100 carrier frequency in the general population.[12]
There are an estimated 3,600 MLD births per year, with 1,900 alive in the US, 3,100 in Europe, and 49,000 alive worldwide with MLD.[12]
MLD is considered a rare disease in the US and other countries.
Research
Bone marrow and stem cell transplant therapies
- Several trials are underway to continue to improve the effectiveness and reduce the risks of bone marrow and stem cell transplants. Cord blood transplants and reduced preparative routines are being studied.
Gene therapy
(current as of January 2017)
Two different approaches to gene therapy are currently being researched for MLD.
- Gene therapy with an autologous stem cell transplant – Italian researchers at the San Raffaele Telethon Institute tested a novel approach combining gene therapy with a stem cell transplant.[13] Recruiting for the Phase I/II Clinical Trial formally started on March 24, 2010 after approval from the Italian Authorities. Recruiting the initial cohort of 8 patients was completed in mid-March 2013. The trial was to test the efficacy and safety of autologous (using the patient's own cells) hematopoietic stem cell transplantation (HSCT) after genetic modification to deliver a super-therapeutic (over-expressing) ARSA enzyme to the nervous system by the route of the blood cells. Using the patient's own stem cells with genetic correction should reduce or eliminate the complications of graft vs. host disease and provide a long term solution to proper ARSA expression in MLD patients. Bench and animal tests showed positive results. The researchers published 2-year outcomes for the first three patients in July 2013. Results were described as promising.[10]
- The Phase I/II clinical trial is complete. No additional patients are being recruited while the data is analyzed and work progresses to improve the manufacturability and repeatable of the technology while an expansion to other geographies to increase access is being considered.
- Recruiting was completed for the 20 patient cohort in April 2015, which includes an expansion in December 2014 to add 6 additional patients.
- Inclusion criteria are pre-symptomatic late infantiles and both pre- and early-symptomatic juveniles. See details on inclusion criteria and the trial protocol here.[14]
- The trial was at a single center at the San Raffaele Institute in Milan, Italy. All costs were to be paid by the researchers. This was a 3-year study. In March 2013, the last of the 8 primary trial patients started therapy. The trial had several compassionate access patients and ultimately was expanded to 20 patients
- In late 2013 GSK exercised its option for the San Rafaelle gene therapy technology and is working with the Milan Investigators to prepare for the next phase of study.[15]
- Intracerebral Gene therapy – A Phase I/II Clinical Trial started recruiting in Paris in late March, 2013 for an Intracerebral Gene Therapy clinical trial where special "vectors" carrying genetically modified material are directly injected into a dozen sites in the brain. The hope is that the corrected cells and the enzyme they produce will then diffuse into surrounding areas of the brain. Extensive work in the lab and some encouraging ALD studies provided the basis for this trial.
- Recruiting 5 late infantiles between the ages of 6 and 48 months for a two-year study
- Must have first shown symptoms in the last 12 months
- Additional trial inclusion criteria and information can be found here
Enzyme replacement therapy (ERT)
(current as of January 2017)
- Shire, with headquarters in Switzerland and a major research center in Lexington, MA, is developing and studying their intrathecal SHP 611 (formerly HGT-1110) ERT [Enzyme Replacement Therapy].
- Clinical Trial
- Recruiting for the clinical trial[16] started January, 2012 and was fully recruited by mid-2014.
- a Fourth cohort was recruited during the first half of 2016. This cohort is fully populated and no new patients are being recruited. Data from this cohort will be gathered by late 2016 with another 3–6 months of outcome analysis expected before a decision is made on what the next drug development and Trial plans will be.
- Phase I/II data is scheduled to be presented in February 2017 at the LDN/WORLD conference.
- Early (post-40 week) results showed the drug was well tolerated at all doses and the 100 mg dose showed the slowest decline in GMFM-88 scores over the trial period. Data continues to be studied.[17]
- Trial Centers
- Trial centers were opened in Europe, South America and Australia
- Patients were successfully recruited in all trial centers
- Inclusion Criteria
- 1st symptoms before age 30 months, currently 7 years old or younger
- Ambulatory – be able to walk 10 steps while holding only one hand.
- Additional clinical trial information & inclusion criteria, can be found on the MLD Foundation website here and at the Clinical Trials.gov site.
- The clinical trial is a 38-week multi-site study of 18 children in three different dosing cohorts. The 'no treatment' placebo arm was removed from the trial in June 2012.[18]
- Patients must go to one of five trial sites for their every other week enzyme infusions: Copenhagen Denmark, Paris France, Tübingen Germany, Sydney Australia, or Porto Alegre Brazil. Derqui, Argentina is awaiting approval.
- A new intrathecal port from a new vendor was approved for use starting December 2013. See the MLD Foundation website for more details.
- SHP611 has orphan product status in both Europe[19] and the United States.[20]
- History: Shire suspended development of the Metazyme intravenous ERT product in 2010. It was in clinical trial when it was acquired from Zymenex in 2008 (subsequently renamed HGT-1111 by Shire) after it was shown to not have sufficient efficacy by a Phase I/II clinical trial in Europe. The initial study completed September 2008 and the extension study completed October 2010 with the cessation of product supply to trial participants.[21][22]
Substrate reduction therapy
- Biomarin South (formerly Zacharon before being acquired by Biomarin in January 2013[23]) from San Diego had initiated a drug discovery program for MLD. This program is based on using assays which measure sulfatide accumulation in cultured fibroblasts as a means to discover and develop small molecule drugs for MLD. (This approach differs from other approaches which have measured enzyme activity to discover effective drugs.) As of July 2011, Zacharon has begun adapting the assays it developed for other lysosomal storage diseases so that they can be employed to discover and develop drugs for MLD. (current March 2013)
- The Cooper Health System (New Jersey) sponsored a clinical trial underway to determine the safety and efficacy of a Vitamin K antagonist (Warfarin) in treating Metachromatic Leukodystrophy (MLD) in 2009. No results are known to have been published.[24] (current March 2013)
Natural history studies
- A natural history study (NHS) launched in Washington, DC in January 2014 to study 30 patients with additional study centers opened in the US, Europe, South America, Southeast Asia, and South America. Due to challenges in recruiting this study has been cancelled.
More information here (current January 2017)
- A natural history study has been underway in Pittsburgh, PA since November 2012.
Research & Clinical Trial updates provided by MLD Foundation
See also
References
- ↑ "metachromatic leukodystrophy" at Dorland's Medical Dictionary
- 1 2 Le, Tao; Bhushan, Vikas; Hofmann, Jeffrey (2012). First Aid for the USMLE Step 1. McGraw-Hill. p. 117.
- ↑ Poeppel P, Habetha M, Marcão A, Büssow H, Berna L, Gieselmann V (March 2005). "Missense mutations as a cause of metachromatic leukodystrophy, Degradation of arylsulfatase A in the endoplasmic reticulum". FEBS J. 272 (5): 1179–88. PMID 15720392. doi:10.1111/j.1742-4658.2005.04553.x.
- 1 2 3 4 Fluharty, Arvan. "Arylsulfatase A Deficiency: Metachromatic Leukodystrophy, ARSA Deficiency". GeneReviews, 2006
- ↑ Kishimoto Y, Hiraiwa M, O'Brien JS. Saposins: structure, function, distribution, and molecular genetics. J Lipid Res. 1992 Sep;33(9):1255-67. PMID 1402395.
- ↑ "Genetics". MLD Foundation.
- ↑ Blomqvist, M.; Gieselmann, V.; Månsson, J. E. (2011). "Accumulation of lysosulfatide in the brain of arylsulfatase A-deficient mice". Lipids in Health and Disease. 10 (1): 28. PMC 3041674
 . PMID 21299873. doi:10.1186/1476-511X-10-28.
. PMID 21299873. doi:10.1186/1476-511X-10-28. - ↑ Hohenschutz, C; Eich P; Friedl W; Waheed A; Conzelmann E; Propping P. (April 1989). "Pseudodeficiency of arylsulfatase A". Human Genetics. 82 (1): 45–8. PMID 2565866. doi:10.1007/bf00288270.
- ↑ Herz, Barbara; Bach, G. (1984). "Arylsulfatase A in pseudodeficiency". Human Genetics. 66: 147–150. doi:10.1007/BF00286589.
- 1 2 Biffi A, Montini E, Lorioli L, et al. (2013). "Lentiviral hematopoietic stem cell gene therapy benefits metachromatic leukodystrophy". Science. 341 (6148): 1233158. PMID 23845948. doi:10.1126/science.1233158.
- 1 2 Metachromatic leukodystrophy at Genetics Home Reference. Reviewed September 2007
- 1 2 "MLD 101: Genetics". www.mldfoundation.org. January 6, 2017. Retrieved January 6, 2017.
- ↑ Biffi A, Lucchini G, Rovelli A, Sessa M (October 2008). "Metachromatic leukodystrophy: an overview of current and prospective treatments". Bone Marrow Transplant. 42 Suppl 2: S2–6. PMID 18978739. doi:10.1038/bmt.2008.275.
- ↑ "MLD gene therapy - San Raffaele - MLD Foundation". mldfoundation.org.
- ↑ "GSK Product Pipeline". GSK. March 2014. Retrieved 29 June 2014.
- ↑ "Multicenter Study of HGT-1110 Administered Intrathecally in Children With Metachromatic Leukodystrophy (MLD) - Full Text View - ClinicalTrials.gov". clinicaltrials.gov.
- ↑ Shire. "March 2015 Quarterly Report" (PDF). Shire Corporate Site. Retrieved 20 May 2015.
- ↑ "NCT01510028 on 2012_06_15: ClinicalTrials.gov Archive". clinicaltrials.gov.
- ↑ "European Medicines Agency - Human medicines - EU/3/10/813". www.ema.europa.eu.
- ↑ "FDA/OOPD issued 27-February-2008".
- ↑ "Long-term Metazym Treatment of Patients With Late Infantile Metachromatic Leukodystrophy (MLD) - Full Text View - ClinicalTrials.gov". clinicaltrials.gov.
- ↑ "Open-Label Extension Study of Recombinant Human Arylsulfatase A (HGT-1111) in Late Infantile MLD - Full Text View - ClinicalTrials.gov". clinicaltrials.gov.
- ↑
- ↑ "Effect of Warfarin in the Treatment of Metachromatic Leukodystrophy - Full Text View - ClinicalTrials.gov". clinicaltrials.gov.
External links
Some portions of this article are courtesy of the public domain text available at the National Institute of Neurological Disorders and Stroke:
- "NINDS Metachromatic Leukodystrophy Information Page". Retrieved 2009-06-07.