Mammal
| Mammals Temporal range: 225–0 Ma (Kemp) or 167–0 Ma (Rowe) See discussion of dates in text | |
|---|---|

 | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Clade: | Amniota |
| Clade: | Synapsida |
| Clade: | Mammaliaformes |
| Class: | Mammalia Linnaeus, 1758 |
| Living subgroups | |
| |
Mammals are any vertebrates within the class Mammalia (/məˈmeɪli.ə/ from Latin mamma "breast"), a clade of endothermic amniotes distinguished from reptiles (including birds) by the possession of a neocortex (a region of the brain), hair, three middle ear bones and mammary glands. Females of all mammal species nurse their young with milk, secreted from the mammary glands.
Mammals include the biggest animals on the planet, the great whales. The basic body type is a terrestrial quadruped, but some mammals are adapted for life at sea, in the air, in trees, underground or on two legs. The largest group of mammals, the placentals, have a placenta, which enables the feeding of the fetus during gestation. Mammals range in size from the 30–40 mm (1.2–1.6 in) bumblebee bat to the 30-meter (98 ft) blue whale. With the exception of the five species of monotreme (egg-laying mammals), all modern mammals give birth to live young. Most mammals, including the six most species-rich orders, belong to the placental group. The largest orders are the rodents, bats and Soricomorpha (shrews and allies). The next three biggest orders, depending on the biological classification scheme used, are the Primates (apes and monkeys), the Cetartiodactyla (whales and even-toed ungulates), and the Carnivora (cats, dogs, seals, and allies).
Living mammals are divided into the Yinotheria (platypus and echidnas) and Theriiformes (all other mammals). There are around 5450 species of mammal, depending on which authority is cited. In some classifications, extant mammals are divided into two subclasses: the Prototheria, that is, the order Monotremata; and the Theria, or the infraclasses Metatheria and Eutheria. The marsupials constitute the crown group of the Metatheria, and include all living metatherians as well as many extinct ones; the placentals are the crown group of the Eutheria. While mammal classification at the family level has been relatively stable, several contending classifications regarding the higher levels—subclass, infraclass and order, especially of the marsupials—appear in contemporaneous literature. Much of the changes reflect the advances of cladistic analysis and molecular genetics. Findings from molecular genetics, for example, have prompted adopting new groups, such as the Afrotheria, and abandoning traditional groups, such as the Insectivora.
The mammals represent the only living Synapsida, which together with the Sauropsida form the Amniota clade. The early synapsid mammalian ancestors were sphenacodont pelycosaurs, a group that produced the non-mammalian Dimetrodon. At the end of the Carboniferous period, this group diverged from the sauropsid line that led to today's reptiles and birds. The line following the stem group Sphenacodontia split-off several diverse groups of non-mammalian synapsids—sometimes referred to as mammal-like reptiles—before giving rise to the proto-mammals (Therapsida) in the early Mesozoic era. The modern mammalian orders arose in the Paleogene and Neogene periods of the Cenozoic era, after the extinction of non-avian dinosaurs, and have been among the dominant terrestrial animal groups from 66 million years ago to the present.
Some mammals are intelligent, with some possessing large brains, self-awareness and tool use. Mammals can communicate and vocalize in several different ways, including the production of ultrasound, scent-marking, alarm signals, singing, and echolocation. Mammals can organize themselves into fission-fusion societies, harems, and hierarchies, but can also be solitary and territorial. Most mammals are polygynous, but some can be monogamous or polyandrous.
In human culture, domesticated mammals played a major role in the Neolithic revolution, causing farming to replace hunting and gathering, and leading to a major restructuring of human societies with the first civilizations. They provided, and continue to provide, power for transport and agriculture, as well as various commodities such as meat, dairy products, wool, and leather. Mammals are hunted or raced for sport, and are used as model organisms in science. Mammals have been depicted in art since Palaeolithic times, and appear in literature, film, mythology, and religion. Defaunation of mammals is primarily driven by anthropogenic factors, such as poaching and habitat destruction, though there are efforts to combat this.
Classification

|
Rodentia
Chiroptera
Soricomorpha
Primates
Carnivora
Artiodactyla
Diprotodontia
Lagomorpha
Didelphimorphia
Cetacea
Dasyuromorphia |
Afrosoricida
Erinaceomorpha
Cingulata
Peramelemorphia
Scandentia
Perissodactyla
Macroscelidea
Pilosa
Monotremata
Proboscidea |
Mammal classification has been through several iterations since Carl Linnaeus initially defined the class. No classification system is universally accepted; McKenna & Bell (1997) and Wilson & Reader (2005) provide useful recent compendiums.[1] George Gaylord Simpson's "Principles of Classification and a Classification of Mammals" (AMNH Bulletin v. 85, 1945) provides systematics of mammal origins and relationships that were universally taught until the end of the 20th century. Since Simpson's classification, the paleontological record has been recalibrated, and the intervening years have seen much debate and progress concerning the theoretical underpinnings of systematization itself, partly through the new concept of cladistics. Though field work gradually made Simpson's classification outdated, it remains the closest thing to an official classification of mammals.[2]
Most mammals, including the six most species-rich orders, belong to the placental group. The three largest orders in numbers of species are Rodentia: mice, rats, porcupines, beavers, capybaras and other gnawing mammals; Chiroptera: bats; and Soricomorpha: shrews, moles and solenodons. The next three biggest orders, depending on the biological classification scheme used, are the Primates including the apes, monkeys and lemurs; the Cetartiodactyla including whales and even-toed ungulates; and the Carnivora which includes cats, dogs, weasels, bears, seals and allies.[3] According to Mammal Species of the World, 5,416 species were identified in 2006. These were grouped into 1,229 genera, 153 families and 29 orders.[3] In 2008, the International Union for Conservation of Nature (IUCN) completed a five-year Global Mammal Assessment for its IUCN Red List, which counted 5,488 species.[4]
Definitions
The word "mammal" is modern, from the scientific name Mammalia coined by Carl Linnaeus in 1758, derived from the Latin mamma ("teat, pap"). In an influential 1988 paper, Timothy Rowe defined Mammalia phylogenetically as the crown group of mammals, the clade consisting of the most recent common ancestor of living monotremes (echidnas and platypuses) and therian mammals (marsupials and placentals) and all descendants of that ancestor.[5] Since this ancestor lived in the Jurassic period, Rowe's definition excludes all animals from the earlier Triassic, despite the fact that Triassic fossils in the Haramiyida have been referred to the Mammalia since the mid-19th century.[6] If Mammalia is considered as the crown group, its origin can be roughly dated as the first known appearance of animals more closely related to some extant mammals than to others. Ambondro is more closely related to monotremes than to therian mammals while Amphilestes and Amphitherium are more closely related to the therians; as fossils of all three genera are dated about 167 million years ago in the Middle Jurassic, this is a reasonable estimate for the appearance of the crown group.[7]
T. S. Kemp has provided a more traditional definition: "synapsids that possess a dentary–squamosal jaw articulation and occlusion between upper and lower molars with a transverse component to the movement" or, equivalently in Kemp's view, the clade originating with the last common ancestor of Sinoconodon and living mammals.[8] The earliest known synapsid satisfying Kemp's definitions is Tikitherium, dated 225 Ma, so the appearance of mammals in this broader sense can be given this Late Triassic date.[9][10]
McKenna/Bell classification
In 1997, the mammals were comprehensively revised by Malcolm C. McKenna and Susan K. Bell, which has resulted in the McKenna/Bell classification. Their 1997 book, Classification of Mammals above the Species Level,[11] is a comprehensive work on the systematics, relationships and occurrences of all mammal taxa, living and extinct, down through the rank of genus, though molecular genetic data challenge several of the higher level groupings. The authors worked together as paleontologists at the American Museum of Natural History, New York. McKenna inherited the project from Simpson and, with Bell, constructed a completely updated hierarchical system, covering living and extinct taxa that reflects the historical genealogy of Mammalia.[2]
Extinct groups are represented by a dagger (†).
Class Mammalia
- Subclass Prototheria: monotremes: echidnas and the platypus
- Subclass Theriiformes: live-bearing mammals and their prehistoric relatives
- Infraclass †Allotheria: multituberculates
- Infraclass †Eutriconodonta: eutriconodonts
- Infraclass Holotheria: modern live-bearing mammals and their prehistoric relatives
- Superlegion †Kuehneotheria
- Supercohort Theria: live-bearing mammals
- Cohort Marsupialia: marsupials
- Magnorder Australidelphia: Australian marsupials and the monito del monte
- Magnorder Ameridelphia: New World marsupials. Now considered paraphyletic, with shrew opossums being closer to australidelphians.[12]
- Cohort Placentalia: placentals
- Magnorder Xenarthra: xenarthrans
- Magnorder Epitheria: epitheres
- Superorder †Leptictida
- Superorder Preptotheria
- Grandorder Anagalida: lagomorphs, rodents and elephant shrews
- Grandorder Ferae: carnivorans, pangolins, †creodonts and relatives
- Grandorder Lipotyphla: insectivorans
- Grandorder Archonta: bats, primates, colugos and treeshrews
- Grandorder Ungulata: ungulates
- Order Tubulidentata incertae sedis: aardvark
- Mirorder Eparctocyona: †condylarths, whales and artiodactyls (even-toed ungulates)
- Mirorder †Meridiungulata: South American ungulates
- Mirorder Altungulata: perissodactyls (odd-toed ungulates), elephants, manatees and hyraxes
- Cohort Marsupialia: marsupials
Molecular classification of placentals
Molecular studies based on DNA analysis have suggested new relationships among mammal families over the last few years. Most of these findings have been independently validated by retrotransposon presence/absence data.[13] Classification systems based on molecular studies reveal three major groups or lineages of placental mammals—Afrotheria, Xenarthra and Boreoeutheria—which diverged in the Cretaceous. The relationships between these three lineages is contentious, and all three possible different hypotheses have been proposed with respect to which group is basal. These hypotheses are Atlantogenata (basal Boreoeutheria), Epitheria (basal Xenarthra) and Exafroplacentalia (basal Afrotheria).[14] Boreoeutheria in turn contains two major lineages—Euarchontoglires and Laurasiatheria.
Estimates for the divergence times between these three placental groups range from 105 to 120 million years ago, depending on the type of DNA used (such as nuclear or mitochondrial)[15] and varying interpretations of paleogeographic data.[14]
| Mammalia |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The cladogram above is based on Tarver et al. (2016)[16]
Group I: Superorder Afrotheria[17]
- Clade Afroinsectiphilia
- Order Macroscelidea: elephant shrews (Africa)
- Order Afrosoricida: tenrecs and golden moles (Africa)
- Order Tubulidentata: aardvark (Africa south of the Sahara)
- Clade Paenungulata
- Order Hyracoidea: hyraxes or dassies (Africa, Arabia)
- Order Proboscidea: elephants (Africa, Southeast Asia)
- Order Sirenia: dugong and manatees (cosmopolitan tropical)
Group II: Superorder Xenarthra[17]
- Order Pilosa: sloths and anteaters (neotropical)
- Order Cingulata: armadillos and extinct relatives (Americas)
Group III: Magnaorder Boreoeutheria[17]
- Superorder: Euarchontoglires (Supraprimates)
- Grandorder Euarchonta
- Order Scandentia: treeshrews (Southeast Asia).
- Order Dermoptera: flying lemurs or colugos (Southeast Asia)
- Order Primates: lemurs, bushbabies, monkeys, apes, humans (cosmopolitan)
- Grandorder Glires
- Order Lagomorpha: pikas, rabbits, hares (Eurasia, Africa, Americas)
- Order Rodentia: rodents (cosmopolitan)
- Grandorder Euarchonta
- Superorder: Laurasiatheria
- Order Eulipotyphla: shrews, hedgehogs, moles, solenodons
- Clade Scrotifera
- Order Chiroptera: bats (cosmopolitan)
- Clade Fereuungulata
- Clade Ferae
- Clade Euungulata
- Order Cetartiodactyla: cetaceans (whales, dolphins and porpoises) and even-toed ungulates, including pigs, cattle, deer and giraffes
- Order Perissodactyla: odd-toed ungulates, including horses, donkeys, zebras, tapirs and rhinoceroses
Evolution
Origins
Synapsida, a clade that contains mammals and their extinct relatives, originated during the Pennsylvanian subperiod, when they split from reptilian and avian lineages. Crown group mammals evolved from earlier mammaliaforms during the Early Jurassic. The cladogram takes Mammalia to be the crown group.[18]
| Mammaliaformes |
| ||||||||||||||||||||||||||||||||||||||||||||||||
Evolution from amniotes
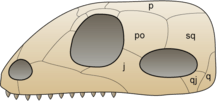
The first fully terrestrial vertebrates were amniotes. Like their amphibious tetrapod predecessors, they had lungs and limbs. Amniotic eggs, however, have internal membranes that allow the developing embryo to breathe but keep water in. Hence, amniotes can lay eggs on dry land, while amphibians generally need to lay their eggs in water.
The first amniotes apparently arose in the Pennsylvanian subperiod of the Carboniferous. They descended from earlier reptiliomorph amphibious tetrapods,[19] which lived on land that was already inhabited by insects and other invertebrates as well as ferns, mosses and other plants. Within a few million years, two important amniote lineages became distinct: the synapsids, which would later include the common ancestor of the mammals; and the sauropsids, which now include turtles, lizards, snakes, crocodilians, dinosaurs and birds.[20] Synapsids have a single hole (temporal fenestra) low on each side of the skull. One synapsid group, the pelycosaurs, included the largest and fiercest animals of the early Permian.[21] Nonmammalian synapsids are sometimes called "mammal-like reptiles".[22][23]
Therapsids, a group of synapsids, descended from pelycosaurs in the Middle Permian, about 265 million years ago, and became the dominant land vertebrates.[22] They differ from basal eupelycosaurs in several features of the skull and jaws, including: larger skulls and incisors which are equal in size in therapsids, but not for eupelycosaurs.[22] The therapsid lineage leading to mammals went through a series of stages, beginning with animals that were very similar to their pelycosaur ancestors and ending with probainognathian cynodonts, some of which could easily be mistaken for mammals. Those stages were characterized by:[24]
- The gradual development of a bony secondary palate.
- Progression towards an erect limb posture, which would increase the animals' stamina by avoiding Carrier's constraint. But this process was slow and erratic: for example, all herbivorous nonmammaliaform therapsids retained sprawling limbs (some late forms may have had semierect hind limbs); Permian carnivorous therapsids had sprawling forelimbs, and some late Permian ones also had semisprawling hindlimbs. In fact, modern monotremes still have semisprawling limbs.
- The dentary gradually became the main bone of the lower jaw which, by the Triassic, progressed towards the fully mammalian jaw (the lower consisting only of the dentary) and middle ear (which is constructed by the bones that were previously used to construct the jaws of reptiles).
First mammals
The Permian–Triassic extinction event about 252 million years ago, which was a prolonged event due to the accumulation of several extinction pulses, ended the dominance of carnivorous therapsids.[25] In the early Triassic, most medium to large land carnivore niches were taken over by archosaurs[26] which, over an extended period (35 million years), came to include the crocodylomorphs,[27] the pterosaurs and the dinosaurs;[28] however, large cynodonts like Trucidocynodon and traversodontids still occupied large sized carnivorous and herbivorous niches respectively. By the Jurassic, the dinosaurs had come to dominate the large terrestrial herbivore niches as well.[29]
The first mammals (in Kemp's sense) appeared in the Late Triassic epoch (about 225 million years ago), 40 million years after the first therapsids. They expanded out of their nocturnal insectivore niche from the mid-Jurassic onwards;[30] The Jurassic Castorocauda, for example, had adaptations for swimming, digging and catching fish.[31] Most, if not all, are thought to have remained nocturnal (the Nocturnal bottleneck), accounting for much of the typical mammalian traits.[32] The majority of the mammal species that existed in the Mesozoic Era were multituberculates, eutriconodonts and spalacotheriids.[33] The earliest known metatherian is Sinodelphys, found in 125 million-year-old Early Cretaceous shale in China's northeastern Liaoning Province. The fossil is nearly complete and includes tufts of fur and imprints of soft tissues.[34]

The oldest known fossil among the Eutheria ("true beasts") is the small shrewlike Juramaia sinensis, or "Jurassic mother from China", dated to 160 million years ago in the late Jurassic.[35] A later eutherian, Eomaia, dated to 125 million years ago in the early Cretaceous, possessed some features in common with the marsupials but not with the placentals, evidence that these features were present in the last common ancestor of the two groups but were later lost in the placental lineage.[36] In particular, the epipubic bones extend forwards from the pelvis. These are not found in any modern placental, but they are found in marsupials, monotremes, nontherian mammals and Ukhaatherium, an early Cretaceous animal in the eutherian order Asioryctitheria. This also applies to the multituberculates.[37] They are apparently an ancestral feature, which subsequently disappeared in the placental lineage. These epipubic bones seem to function by stiffening the muscles during locomotion, reducing the amount of space being presented, which placentals require to contain their fetus during gestation periods. A narrow pelvic outlet indicates that the young were very small at birth and therefore pregnancy was short, as in modern marsupials. This suggests that the placenta was a later development.[38]
The earliest known monotreme was Teinolophos, which lived about 120 million years ago in Australia.[39] Monotremes have some features which may be inherited from the original amniotes such as the same orifice to urinate, defecate and reproduce (cloaca) – as lizards and birds also do –[40] and they lay eggs which are leathery and uncalcified.[41]
Earliest appearances of features
Hadrocodium, whose fossils date from approximately 195 million years ago, in the early Jurassic, provides the first clear evidence of a jaw joint formed solely by the squamosal and dentary bones; there is no space in the jaw for the articular, a bone involved in the jaws of all early synapsids.[42]
The earliest clear evidence of hair or fur is in fossils of Castorocauda and Megaconus, from 164 million years ago in the mid-Jurassic. In the 1950s, it was suggested that the foramina (passages) in the maxillae and premaxillae (bones in the front of the upper jaw) of cynodonts were channels which supplied blood vessels and nerves to vibrissae (whiskers) and so were evidence of hair or fur;[43][44] it was soon pointed out, however, that foramina do not necessarily show that an animal had vibrissae, as the modern lizard Tupinambis has foramina that are almost identical to those found in the nonmammalian cynodont Thrinaxodon.[23][45] Popular sources, nevertheless, continue to attribute whiskers to Thrinaxodon.[46] Studies on Permian coprolites suggest that non-mammalian synapsids of the epoch already had fur, setting the evolution of hairs possibly as far back as dicynodonts.[47]
When endothermy first appeared in the evolution of mammals is uncertain, though it is generally agreed to have first evolved in non-mammalian therapsids.[47][48] Modern monotremes have lower body temperatures and more variable metabolic rates than marsupials and placentals,[49] but there is evidence that some of their ancestors, perhaps including ancestors of the therians, may have had body temperatures like those of modern therians.[50] Likewise, some modern therians like afrotheres and xenarthrans have secondarily developed lower body temperatures.[51]
The evolution of erect limbs in mammals is incomplete — living and fossil monotremes have sprawling limbs. The parasagittal (nonsprawling) limb posture appeared sometime in the late Jurassic or early Cretaceous; it is found in the eutherian Eomaia and the metatherian Sinodelphys, both dated to 125 million years ago.[52] Epipubic bones, a feature that strongly influenced the reproduction of most mammal clades, are first found in Tritylodontidae, suggesting that it is a synapomorphy between them and mammaliformes. They are omnipresent in non-placental mammaliformes, though Megazostrodon and Erythrotherium appear to have lacked them.[53]
It has been suggested that the original function of lactation (milk production) was to keep eggs moist. Much of the argument is based on monotremes, the egg-laying mammals.[54][55]
Rise of the mammals
Therian mammals took over the medium- to large-sized ecological niches in the Cenozoic, after the Cretaceous–Paleogene extinction event approximately 66 million years ago emptied ecological space once filled by non-avian dinosaurs and other groups of reptiles, as well as various other mammal groups,[56] and underwent an exponential increase in body size (megafauna).[57] Then mammals diversified very quickly; both birds and mammals show an exponential rise in diversity.[56] For example, the earliest known bat dates from about 50 million years ago, only 16 million years after the extinction of the dinosaurs.[58]
Molecular phylogenetic studies initially suggested that most placental orders diverged about 100 to 85 million years ago and that modern families appeared in the period from the late Eocene through the Miocene.[59] However, no placental fossils have been found from before the end of the Cretaceous.[60] The earliest undisputed fossils of placentals comes from the early Paleocene, after the extinction of the dinosaurs.[60] In particular, scientists have identified an early Paleocene animal named Protungulatum donnae as one of the first placental mammals.[61] however it has been reclassified as a non-placental eutherian.[62] Recalibrations of genetic and morphological diversity rates have suggested a Late Cretaceous origin for placentals, and a Paleocene origin for most modern clades.[63]
The earliest known ancestor of primates is Archicebus achilles[64] from around 55 million years ago.[64] This tiny primate weighed 20–30 grams (0.7–1.1 ounce) and could fit within a human palm.[64]
Anatomy and morphology
Distinguishing features
Living mammal species can be identified by the presence of sweat glands, including those that are specialized to produce milk to nourish their young.[65] In classifying fossils, however, other features must be used, since soft tissue glands and many other features are not visible in fossils.[66]
Many traits shared by all living mammals appeared among the earliest members of the group:
- Jaw joint - The dentary (the lower jaw bone, which carries the teeth) and the squamosal (a small cranial bone) meet to form the joint. In most gnathostomes, including early therapsids, the joint consists of the articular (a small bone at the back of the lower jaw) and quadrate (a small bone at the back of the upper jaw).[42]
- Middle ear - In crown-group mammals, sound is carried from the eardrum by a chain of three bones, the malleus, the incus and the stapes. Ancestrally, the malleus and the incus are derived from the articular and the quadrate bones that constituted the jaw joint of early therapsids.[67]
- Tooth replacement - Teeth are replaced once or (as in toothed whales and murid rodents) not at all, rather than being replaced continually throughout life.[68]
- Prismatic enamel - The enamel coating on the surface of a tooth consists of prisms, solid, rod-like structures extending from the dentin to the tooth's surface.[69]
- Occipital condyles - Two knobs at the base of the skull fit into the topmost neck vertebra; most other tetrapods, in contrast, have only one such knob.[70]
For the most part, these characteristics were not present in the Triassic ancestors of the mammals.[71] Nearly all mammaliaforms possess an epipubic bone, the exception being modern placentals.[72]
Biological systems

The majority of mammals have seven cervical vertebrae (bones in the neck), including bats, giraffes, whales and humans. The exceptions are the manatee and the two-toed sloth, which have just six, and the three-toed sloth which has nine cervical vertebrae.[73] All mammalian brains possess a neocortex, a brain region unique to mammals.[74] Placental mammals have a corpus callosum, unlike monotremes and marsupials.[75]
The lungs of mammals are spongy and honeycombed. Breathing is mainly achieved with the diaphragm, which divides the thorax from the abdominal cavity, forming a dome convex to the thorax. Contraction of the diaphragm flattens the dome, increasing the volume of the lung cavity. Air enters through the oral and nasal cavities, and travels through the larynx, trachea and bronchi, and expands the alveoli. Relaxing the diaphragm has the opposite effect, decreasing the volume of the lung cavity, causing air to be pushed out of the lungs. During exercise, the abdominal wall contracts, increasing pressure on the diaphragm, which forces air out quicker and more forcefully. The rib cage is able to expand and contract the chest cavity through the action of other respiratory muscles. Consequently, air is sucked into or expelled out of the lungs, always moving down its pressure gradient.[76][77] This type of lung is known as a bellows lung due to its resemblance to blacksmith bellows.[77]
The mammalian heart has four chambers, two upper atria, the receiving chambers, and two lower ventricles, the discharging chambers.[78] The heart has four valves, which separate its chambers and ensures blood flows in the correct direction through the heart (preventing backflow). After gas exchange in the pulmonary capillaries (blood vessels in the lungs), oxygen-rich blood returns to the left atrium via one of the four pulmonary veins. Blood flows nearly continuously back into the atrium, which acts as the receiving chamber, and from here through an opening into the left ventricle. Most blood flows passively into the heart while both the atria and ventricles are relaxed, but toward the end of the ventricular relaxation period, the left atrium will contract, pumping blood into the ventricle. The heart also requires nutrients and oxygen found in blood like other muscles, and is supplied via coronary arteries.[79]
The integumentary system is made up of three layers: the outermost epidermis, the dermis and the hypodermis. The epidermis is typically 10 to 30 cells thick; its main function is to provide a waterproof layer. Its outermost cells are constantly lost; its bottommost cells are constantly dividing and pushing upward. The middle layer, the dermis, is 15 to 40 times thicker than the epidermis. The dermis is made up of many components, such as bony structures and blood vessels. The hypodermis is made up of adipose tissue, which stores lipids and provides cushioning and insulation. The thickness of this layer varies widely from species to species;[80]:97 marine mammals require a thick hypodermis (blubber) for insulation, and right whales have the thickest blubber at 20 inches (51 cm).[81] Although other animals have features such as whiskers, feathers, setae, or cilia that superficially resemble it, no animals other than mammals have hair. It is a definitive characteristic of the class. Though some mammals have very little, careful examination reveals the characteristic, often in obscure parts of their bodies.[80]:61

Herbivores have developed a diverse range of physical structures to facilitate the consumption of plant material. To break up intact plant tissues, mammals have developed teeth structures that reflect their feeding preferences. For instance, frugivores (animals that feed primarily on fruit) and herbivores that feed on soft foliage have low-crowned teeth specialized for grinding foliage and seeds. Grazing animals that tend to eat hard, silica-rich grasses, have high-crowned teeth, which are capable of grinding tough plant tissues and do not wear down as quickly as low-crowned teeth.[82] Most carnivorous mammals have carnassialiforme teeth (of varying length depending on diet), long canines and similar tooth replacement patterns.[83]
The stomach of Artiodactyls is divided into four sections: the rumen, the reticulum, the omasum and the abomasum (only ruminants have a rumen). After the plant material is consumed, it is mixed with saliva in the rumen and reticulum and separates into solid and liquid material. The solids lump together to form a bolus (or cud), and is regurgitated. When the bolus enters the mouth, the fluid is squeezed out with the tongue and swallowed again. Ingested food passes to the rumen and reticulum where cellulytic microbes (bacteria, protozoa and fungi) produce cellulase, which is needed to break down the cellulose in plants.[84] Perissodactyls, in contrast to the ruminants, store digested food that has left the stomach in an enlarged cecum, where it is fermented by bacteria.[85] Carnivora have a simple stomach adapted to digest primarily meat, as compared to the elaborate digestive systems of herbivorous animals, which are necessary to break down tough, complex plant fibers. The caecum is either absent or short and simple, and the large intestine is not sacculated or much wider than the small intestine.[86]

The mammalian excretory system involves many components. Like most other land animals, mammals are ureotelic, and convert ammonia into urea, which is done by the liver as part of the urea cycle.[87] Bilirubin, a waste product derived from blood cells, is passed through bile and urine with the help of enzymes excreted by the liver.[88] The passing of bilirubin via bile through the intestinal tract gives mammalian feces a distinctive brown coloration.[89] Distinctive features of the mammalian kidney include the presence of the renal pelvis and renal pyramids, and of a clearly distinguishable cortex and medulla, which is due to the presence of elongated loops of Henle. Only the mammalian kidney has a bean shape, although there are some exceptions, such as the multilobed reniculate kidneys of pinnipeds, cetaceans and bears.[90][91] Most adult placental mammals have no remaining trace of the cloaca. In the embryo, the embryonic cloaca divides into a posterior region that becomes part of the anus, and an anterior region that has different fates depending on the sex of the individual: in females, it develops into the vestibule that receives the urethra and vagina, while in males it forms the entirety of the penile urethra.[91] However, the tenrecs, golden moles, and some shrews retain a cloaca as adults.[92] In marsupials, the genital tract is separate from the anus, but a trace of the original cloaca does remain externally.[91] Monotremes, which translates from Greek into "single hole", have a true cloaca.[93]
Sound production
As in all other tetrapods, mammals have a larynx that can quickly open and close to produce sounds, and a supralaryngeal vocal tract which filters this sound. The lungs and surrounding musculature provide the air stream and pressure required to phonate. The larynx controls the pitch and volume of sound, but the strength the lungs exert to exhale also contributes to volume. More primitive mammals, such as the echidna, can only hiss, as sound is achieved solely through exhaling through a partially close larynx. Other mammals phonate using vocal folds, as opposed to the vocal cords seen in birds and reptiles. The movement or tenseness of the vocal folds can result in many sounds such as purring and screaming. Mammals can change the position of the larynx, allowing them to breathe through the nose while swallowing through the mouth, and to create both oral and nasal sounds; nasal sounds, such as a dog whine, are generally soft sounds, and oral sounds, such as a dog bark, are generally loud.[94]
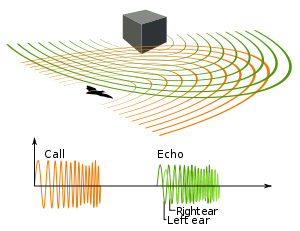
Some mammals have a large larynx and, thus, a low-pitched voice, namely the hammer-headed bat (Hypsignathus monstrosus) where the larynx can take up the entirety of the thoracic cavity while pushing the lungs, heart, and trachea into the abdomen.[95] Large vocal pads can also lower the pitch, as in the low-pitched roars of big cats.[96] The production of infrasound is possible in some mammals such as the African elephant (Loxodonta spp.) and baleen whales.[97][98] Small mammals with small larynxes have the ability to produced ultrasound, which can be detected by modifications to the middle ear and cochlea. Ultrasound is inaudible to birds and reptiles, which might have been important during the Mesozoic, when birds and reptiles were the dominant predators. This private channel is used by some rodents in, for example, mother-to-pup communication, and by bats when echolocating. Toothed whales also use echolocation, but, as opposed to the vocal membrane that extends upward from the vocal folds, they have a melon to manipulate sounds. Some mammals, namely the primates, have air sacs attached to the larynx, which may function to increase the volume of sound.[94]
The vocal production system is controlled by the cranial nerve nucleus in the brain, and supplied by the recurrent laryngeal nerve and the superior laryngeal nerve, branches of the vagus nerve. The vocal tract is supplied by the hypoglossal nerve and facial nerves. Electrical stimulation of the periaqueductal gray (PEG) region of the mammalian midbrain elicit vocalizations. The ability to learn new vocalizations is only exemplified in humans, seals, cetaceans, and possibly bats; in humans, this is the result of a direct connection between the motor cortex, which controls movement, and the motor neurons in the spinal cord.[94]
Fur

The fur of mammals has many uses protection, sensory purposes, waterproofing, and camouflage, with the primary usage being thermoregulation.[99] The types of hair include definitive, which may be shed after reaching a certain length; vibrissae, which are sensory hairs and are most commonly whiskers; pelage, which consists of guard hairs, under-fur, and awn hair; spines, which are a type of stiff guard hair used for defense in, for example, porcupines; bristles, which are long hairs usually used in visual signals, such as the mane of a lion; velli, often called "down fur," which insulates newborn mammals; and wool which is long, soft and often curly.[80]:99 Hair length is negligible in thermoregulation, as some tropical mammals, such as sloths, have the same length of fur length as some arctic mammals but with less insulation; and, conversely, other tropical mammals with short hair have the same insulating value as arctic mammals. The denseness of fur can increase an animal's insulation value, and arctic mammals especially have dense fur; for example, the musk ox has guard hairs measuring 12 inches (30 cm) as well as a dense underfur, which forms an airtight coat, allowing them to survive in temperatures of −40 °F (−40 °C).[80]:162–163 Some desert mammals, such as camels, use dense fur to prevent solar heat from reaching their skin, allowing the animal to stay cool; a camel's fur may reach 158 °F (70 °C) in the summer, but the skin stays at 104 °F (40 °C).[80]:188 Aquatic mammals, conversely, trap air in their fur to conserve heat by keeping the skin dry.[80]:162–163
Mammalian coats are colored for a variety of reasons, the major selective pressures including camouflage, sexual selection, communication and physiological processes such as temperature regulation. Camouflage is a powerful influence in a large number of mammals, as it helps to conceal individuals from predators or prey.[100] Aposematism, warning off possible predators, is the most likely explanation of the black-and-white pelage of many mammals which are able to defend themselves, such as in the foul-smelling skunk and the powerful and aggressive honey badger.[101] In arctic and subarctic mammals such as the arctic fox (Alopex lagopus), collared lemming (Dicrostonyx groenlandicus), stoat (Mustela erminea), and snowshoe hare (Lepus americanus), seasonal color change between brown in summer and white in winter is driven largely by camouflage.[102] Differences in female and male coat color may indicate nutrition and hormone levels, important in mate selection.[103] Some arboreal mammals, notably primates and marsupials, have shades of violet, green, or blue skin on parts of their bodies, indicating some distinct advantage in their largely arboreal habitat due to convergent evolution.[104] The green coloration of sloths, however, is the result of a symbiotic relationship with algae.[105] Coat color is sometimes sexually dimorphic, as in many primate species.[106] Coat color may influence the ability to retain heat, depending on how much light is reflected. Mammals with a darker colored coat can absorb more heat from solar radiation, and stay warmer, and some smaller mammals, such as voles, have darker fur in the winter. The white, pigmentless fur of arctic mammals, such as the polar bear, may reflect more solar radiation directly onto the skin.[80]:166–167[99]
Reproductive system


In male placentals, the penis is used both for urination and copulation. Depending on the species, an erection by be fueled by blood flow into vascular, spongy tissue or by muscular action. A penis may be contained in a sheath when not erect, and some placentals also have a penis bone (baculum). Marsupials typically have forked penises while the monotreme penis generally has four heads with only two functioning. The testes of most mammals descend into the scrotum which is typically posterior to the penis but is often anterior in marsupials. Female mammals generally have a clitoris, labia majora and labia minora on the outside, while the internal system contains paired oviducts, 1-2 uteri, 1-2 cervices and a vagina. Marsupials have two lateral vaginas and a medial vagina. The "vagina" of monotremes is better understood as a "urogenital sinus". The uterine systems of placental mammals can vary between a duplex, were there are two uteri and cervices which open into the vagina, a bipartite, were two uterine horns have a single cervix that connects to the vagina, a bicornuate, which consists where two uterine horns that are connected distally but separate medially creating a Y-shape, and a simplex, which has a single uterus.[107][108][80]:247, 220–21
Most mammals are viviparous, giving birth to live young. However, the five species of monotreme, the platypus and the four species of echidna, lay eggs. The monotremes have a sex determination system different from that of most other mammals.[109] In particular, the sex chromosomes of a platypus are more like those of a chicken than those of a therian mammal.[110]
Viviparous mammals are in the subclass Theria; those living today are in the marsupial and placental infraclasses. Marsupials have a short gestation period, typically shorter than its estrous cycle and gives birth to an undeveloped newborn that then undergoes further development; in many species, this takes place within a pouch-like sac, the marsupium, located in the front of the mother's abdomen. This is the plesiomorphic condition among viviparous mammals; the presence of epipubic bones in all non-placental mammals prevents the expansion of the torso needed for full pregnancy.[72] Even non-placental eutherians probably reproduced this way.[111] The placentals give birth to relatively complete and developed young, usually after long gestation periods.[112] They get their name from the placenta, which connects the developing fetus to the uterine wall to allow nutrient uptake.[113]
The mammary glands of mammals are specialized to produce milk, the primary source of nutrition for newborns. The monotremes branched early from other mammals and do not have the nipples seen in most mammals, but they do have mammary glands. The young lick the milk from a mammary patch on the mother's belly.[114]
Endothermy
Nearly all mammals are endothermic ("warm-blooded"). Most mammals also have hair to help keep them warm. Like birds, mammals can forage or hunt in weather and climates too cold for ectothermic ("cold-blooded") reptiles and insects. Endothermy requires plenty of food energy, so mammals eat more food per unit of body weight than most reptiles.[115] Small insectivorous mammals eat prodigious amounts for their size. A rare exception, the naked mole-rat produces little metabolic heat, so it is considered an operational poikilotherm.[116] Birds are also endothermic, so endothermy is not unique to mammals.[117]
Species lifespan
Among mammals, species maximum lifespan varies significantly (for example the shrew has a lifespan of two years, whereas the oldest bowhead whale is recorded to be 211 years).[118] Although the underlying basis for these lifespan differences is still uncertain, numerous studies indicate that the ability to repair DNA damages is an important determinant of mammalian lifespan. In a 1974 study by Hart and Setlow,[119] it was found that DNA excision repair capability increased systematically with species lifespan among seven mammalian species. Species lifespan was observed to be robustly correlated with the capacity to recognize DNA double-strand breaks as well as the level of the DNA repair protein Ku80.[118] In a study of the cells from sixteen mammalian species, genes employed in DNA repair were found to be up-regulated in the longer-lived species.[120] The cellular level of the DNA repair enzyme poly ADP ribose polymerase was found to correlate with species lifespan in a study of 13 mammalian species.[121] Three additional studies of a variety of mammalian species also reported a correlation between species lifespan and DNA repair capability.[122][123][124]
Locomotion
Terrestrial

Most vertebrates—the amphibians, the reptiles and some mammals such as humans and bears—are plantigrade, walking on the whole of the underside of the foot. Many mammals, such as cats and dogs, are digitigrade, walking on their toes, the greater stride length allowing more speed. Digitigrade mammals are also often adept at quiet movement.[125] Some animals such as horses are unguligrade, walking on the tips of their toes. This even further increases their stride length and thus their speed.[126] A few mammals, namely the great apes, are also known to walk on their knuckles, at least for their front legs. Giant anteaters[127] and platypuses[128] are also knuckle-walkers. Some mammals are bipeds, using only two limbs for locomotion, which can be seen in, for example, humans and the great apes. Bipedal species have a larger field of vision than quadrupeds, conserve more energy and have the ability to manipulate objects with their hands, which aids in foraging. Instead of walking, some bipeds hop, such as kangaroos and kangaroo rats.[129][130]
Animals will use different gaits for different speeds, terrain and situations. For example, horses show four natural gaits, the slowest horse gait is the walk, then there are three faster gaits which, from slowest to fastest, are the trot, the canter and the gallop. Animals may also have unusual gaits that are used occasionally, such as for moving sideways or backwards. For example, the main human gaits are bipedal walking and running, but they employ many other gaits occasionally, including a four-legged crawl in tight spaces.[131] Mammals show a vast range of gaits, the order that they place and lift their appendages in locomotion. Gaits can be grouped into categories according to their patterns of support sequence. For quadrupeds, there are three main categories: walking gaits, running gaits and leaping gaits.[132] Walking is the most common gait, where some feet are on the ground at any given time, and found in almost all legged animals. Running is considered to occur when at some points in the stride all feet are off the ground in a moment of suspension.[131]
Arboreal
.jpg)
Arboreal animals frequently have elongated limbs that help them cross gaps, reach fruit or other resources, test the firmness of support ahead and, in some cases, to brachiate (swing between trees).[133] Many arboreal species, such as tree porcupines, silky anteaters, spider monkeys and possums, use prehensile tails to grasp branches. In the spider monkey, the tip of the tail has either a bare patch or adhesive pad, which provides increased friction. Claws can be used to interact with rough substrates and re-orient the direction of forces the animal applies. This is what allows squirrels to climb tree trunks that are so large to be essentially flat from the perspective of such a small animal. However, claws can interfere with an animal's ability to grasp very small branches, as they may wrap too far around and prick the animal's own paw. Frictional gripping is used by primates, relying upon hairless fingertips. Squeezing the branch between the fingertips generates frictional force that holds the animal's hand to the branch. However, this type of grip depends upon the angle of the frictional force, thus upon the diameter of the branch, with larger branches resulting in reduced gripping ability. To control descent, especially down large diameter branches, some arboreal animals such as squirrels have evolved highly mobile ankle joints that permit rotating the foot into a 'reversed' posture. This allows the claws to hook into the rough surface of the bark, opposing the force of gravity. Small size provides many advantages to arboreal species: such as increasing the relative size of branches to the animal, lower center of mass, increased stability, lower mass (allowing movement on smaller branches) and the ability to move through more cluttered habitat.[133] Size relating to weight affects gliding animals such as the sugar glider.[134] Some species of primate, bat and all species of sloth achieve passive stability by hanging beneath the branch. Both pitching and tipping become irrelevant, as the only method of failure would be losing their grip.[133]
Aerial
Bats are the only mammals that can truly fly. They fly through the air at a constant speed by moving their wings up and down (usually with some fore-aft movement as well). Because the animal is in motion, there is some airflow relative to its body which, combined with the velocity of the wings, generates a faster airflow moving over the wing. This generates a lift force vector pointing forwards and upwards, and a drag force vector pointing rearwards and upwards. The upwards components of these counteract gravity, keeping the body in the air, while the forward component provides thrust to counteract both the drag from the wing and from the body as a whole.[135]
The wings of bats are much thinner and consist of more bones than that of birds, allowing bats to maneuver more accurately and fly with more lift and less drag.[136][137] By folding the wings inwards towards their body on the upstroke, they use 35% less energy during flight than birds.[138] The membranes are delicate, ripping easily; however, the tissue of the bat's membrane is able to regrow, such that small tears can heal quickly.[139] The surface of their wings is equipped with touch-sensitive receptors on small bumps called Merkel cells, also found on human fingertips. These sensitive areas are different in bats, as each bump has a tiny hair in the center, making it even more sensitive and allowing the bat to detect and collect information about the air flowing over its wings, and to fly more efficiently by changing the shape of its wings in response.[140]
Fossorial
Fossorial creatures live in subterranean environments. Many fossorial mammals were classified under the, now obsolete, order Insectivora, such as shrews, hedgehogs and moles. Fossorial mammals have a fusiform body, thickest at the shoulders and tapering off at the tail and nose. Unable to see in the dark burrows, most have degenerated eyes, but degeneration varies between species; pocket gophers, for example, are only semi-fossorial and have very small yet functional eyes, in the fully fossorial marsupial mole the eyes are degenerated and useless, talpa moles have vestigial eyes and the cape golden mole has a layer of skin covering the eyes. External ears flaps are also very small or absent. Truly fossorial mammals have short, stout legs as strength is more important than speed to a burrowing mammal, but semi-fossorial mammals have cursorial legs. The front paws are broad and have strong claws to help in loosening dirt while excavating burrows, and the back paws have webbing, as well as claws, which aids in throwing loosened dirt backwards. Most have large incisors to prevent dirt from flying into their mouth.[141]
Aquatic
Fully aquatic mammals, the cetaceans and sirenians, have lost their legs and have a tail fin to propel themselves through the water. Flipper movement is continuous. Whales swim by moving their tail fin and lower body up and down, propelling themselves through vertical movement, while their flippers are mainly used for steering. Their skeletal anatomy allows them to be fast swimmers. Most species have a dorsal fin to prevent themselves from turning upside-down in the water.[142][143] The flukes of sirenians are raised up and down in long strokes to move the animal forward, and can be twisted to turn. The forelimbs are paddle-like flippers which aid in turning and slowing.[144]
Semi-aquatic mammals, like pinnipeds, have two pairs of flippers on the front and back, the fore-flippers and hind-flippers. The elbows and ankles are enclosed within the body.[145][146] Pinnipeds have several adaptions for reducing drag. In addition to their streamlined bodies, they have smooth networks of muscle bundles in their skin that may increase laminar flow and make it easier for them to slip through water. They also lack arrector pili, so their fur can be streamlined as they swim.[147] They rely on their fore-flippers for locomotion in a wing-like manner similar to penguins and sea turtles.[148] Fore-flipper movement is not continuous, and the animal glides between each stroke.[146] Compared to terrestrial carnivorans, the fore-limbs are reduced in length, which gives the locomotor muscles at the shoulder and elbow joints greater mechanical advantage;[145] the hind-flippers serve as stabilizers.[147] Other semi-aquatic mammals include beavers, hippopotamuses, otters and platypuses.[149] Hippos are very large semi-aquatic mammals, and their barrel-shaped bodies have graviportal skeletal structures,[150] adapted to carrying their enormous weight, and their specific gravity allows them to sink and move along the bottom of a river.[151]
Behavior
Communication and vocalization
Many mammals communicate by vocalizing. Vocal communication serves many purposes, including in mating rituals, as warning calls,[153] to indicate food sources, and for social purposes. Males often call during mating rituals to ward off other males and to attract females, as in the roaring of lions and red deer.[154] The songs of the humpback whale may be signals to females;[155] they have different dialects in different regions of the ocean.[156] Social vocalizations include the territorial calls of gibbons, and the use of frequency in greater spear-nosed bats to distinguish between groups.[157] The vervet monkey gives a distinct alarm call for each of at least four different predators, and the reactions of other monkeys vary according to the call. For example, if an alarm call signals a python, the monkeys climb into the trees, whereas the eagle alarm causes monkeys to seek a hiding place on the ground.[152] Prairie dogs similarly have complex calls that signal the type, size, and speed of an approaching predator.[158] Elephants communicate socially with a variety of sounds including snorting, screaming, trumpeting, roaring and rumbling. Some of the rumbling calls are infrasonic, below the hearing range of humans, and can be heard by other elephants up to 6 miles (9.7 km) away at still times near sunrise and sunset.[159]
Mammals signal by a variety of means. Many give visual anti-predator signals, as when deer and gazelle stot, honestly indicating their fit condition and their ability to escape,[160][161] or when white-tailed deer and other prey mammals flag with conspicuous tail markings when alarmed, informing the predator that it has been detected.[162] Many mammals make use of scent-marking, sometimes possibly to help defend territory, but probably with a range of functions both within and between species.[163] Microbats and toothed whales including oceanic dolphins vocalize both socially and in echolocation.[164][165][166]
Feeding

To maintain a high constant body temperature is energy expensive – mammals therefore need a nutritious and plentiful diet. While the earliest mammals were probably predators, different species have since adapted to meet their dietary requirements in a variety of ways. Some eat other animals – this is a carnivorous diet (and includes insectivorous diets). Other mammals, called herbivores, eat plants, which contain complex carbohydrates such as cellulose. An herbivorous diet includes subtypes such as granivory (seed eating), folivory (leaf eating), frugivory (fruit eating), nectarivory (nectar eating), gummivory (gum eating) and mycophagy (fungus eating). The digestive tract of an herbivore is host to bacteria that ferment these complex substances, and make them available for digestion, which are either housed in the multichambered stomach or in a large cecum.[168] Some mammals are coprophagous, consuming feces to absorb the nutrients not digested when the food was first ingested.[80]:131–137 An omnivore eats both prey and plants. Carnivorous mammals have a simple digestive tract because the proteins, lipids and minerals found in meat require little in the way of specialized digestion. Exceptions to this include baleen whales who also house gut flora in a multi-chambered stomach, like terrestrial herbivores.[169]
The size of an animal is also a factor in determining diet type (Allen's rule). Since small mammals have a high ratio of heat-losing surface area to heat-generating volume, they tend to have high energy requirements and a high metabolic rate. Mammals that weigh less than about 18 ounces (510 g) are mostly insectivorous because they cannot tolerate the slow, complex digestive process of an herbivore. Larger animals, on the other hand, generate more heat and less of this heat is lost. They can therefore tolerate either a slower collection process (those that prey on larger vertebrates) or a slower digestive process (herbivores).[170] Furthermore, mammals that weigh more than 18 ounces (510 g) usually cannot collect enough insects during their waking hours to sustain themselves. The only large insectivorous mammals are those that feed on huge colonies of insects (ants or termites).[171]

_scavenging_a_narwhal_whale_(Monodon_monoceros)_carcass_-_journal.pone.0060797.g001-A.png)
Some mammals are omnivores and display varying degrees of carnivory and herbivory, generally leaning in favor of one more than the other. Since plants and meat are digested differently, there is a preference for one over the other, as in bears where some species may be mostly carnivorous and others mostly herbivorous.[173] They are grouped into three categories: mesocarnivory (50-70% meat), hypercarnivory (70% and greater of meat), and hypocarnivory (50% or less of meat). The dentition of hypocarnivores consists of dull, triangular carnassial teeth meant for grinding food. Hypercarnivores, however, have conical teeth and sharp carnassials meant for slashing, and in some cases strong jaws for bone-crushing, as in the case of hyenas, allowing them to consume bones; some extinct groups, notably the Machairodontinae, had saber-shaped canines.[172]
Some physiological carnivores consume plant matter and some physiological herbivores consuming meat. From a behavioral aspect, this would make them omnivores, but from the physiological standpoint, this may be due to zoopharmacognosy. Physiologically, animals must be able to obtain both energy and nutrients from plant and animal materials to be considered omnivorous. Thus, such animals are still able to be classified as carnivores and herbivores when they are just obtaining nutrients from materials originating from sources that do not seemingly complement their classification.[174] For example, it is well documented that some ungulates. such as giraffes, camels, and cattle, will gnaw on bones to consume particular minerals and nutrients.[175] Also, cats, which are generally regarded as obligate carnivores, occasionally eat grass to regurgitate indigestible material (such as hairballs), aid with hemoglobin production, and as a laxative.[176]
Many mammals, in the absence of sufficient food requirements in an environment, suppress their metabolism and conserve energy in a process known as hibernation.[177] In the period preceding hibernation, larger mammals, such as bears, become polyphagic to increase fat stores, whereas smaller mammals prefer to collect and stash food.[178] The slowing of the metabolism is accompanied by a decreased heart and respiratory rate, as well as a drop in internal temperatures, which can be around ambient temperature in some cases. For example, the internal temperatures of hibernating arctic ground squirrels can drop to −2.9 °C (26.8 °F), however the head and neck always stay above 0 °C (32 °F).[179] A few mammals in hot environments aestivate in times of drought or extreme heat, namely the fat-tailed dwarf lemur (Cheirogaleus medius).[180]
Intelligence
In intelligent mammals, such as primates, the cerebrum is larger relative to the rest of the brain. Intelligence itself is not easy to define, but indications of intelligence include the ability to learn, matched with behavioral flexibility. Rats, for example, are considered to be highly intelligent, as they can learn and perform new tasks, an ability that may be important when they first colonize a fresh habitat. In some mammals, food gathering appears to be related to intelligence: a deer feeding on plants has a brain smaller than a cat, which must think to outwit its prey.[171]
Tool use by animals may indicate different levels of learning and cognition. The sea otter uses rocks as essential and regular parts of its foraging behaviour (smashing abalone from rocks or breaking open shells), with some populations spending 21% of their time making tools.[181] Other tool use, such as chimpanzees using twigs to "fish" for termites, may be developed by watching others use tools and may even be a true example of animal teaching.[182] Tools may even be used in solving puzzles in which the animal appears to experience a "Eureka moment".[183] Other mammals that do not use tools, such as dogs, can also experience a Eureka moment.[184]
Brain size was previously considered a major indicator of the intelligence of an animal. Since most of the brain is used for maintaining bodily functions, greater ratios of brain to body mass may increase the amount of brain mass available for more complex cognitive tasks. Allometric analysis indicates that mammalian brain size scales at approximately the ⅔ or ¾ exponent of the body mass. Comparison of a particular animal's brain size with the expected brain size based on such allometric analysis provides an encephalisation quotient that can be used as another indication of animal intelligence.[185] Sperm whales have the largest brain mass of any animal on earth, averaging 8,000 cubic centimetres (490 in3) and 7.8 kilograms (17 lb) in mature males.[186]
Self-awareness appears to be a sign of abstract thinking. Self-awareness, although not well-defined, is believed to be a precursor to more advanced processes such as metacognitive reasoning. The traditional method for measuring this is the mirror test, which determines if an animal possesses the ability of self-recognition.[187] Mammals that have 'passed' the mirror test include Asian elephants (some pass, some do not);[188] chimpanzees;[189] bonobos;[190] orangutans;[191] humans, from 18 months (mirror stage);[192] bottlenose dolphins[lower-alpha 1][193] killer whales;[194] and false killer whales.[194]
Social structure

Eusociality is the highest level of social organization. These societies have an overlap of adult generations, the division of reproductive labor and cooperative caring of young. Usually insects, such as bees, ants and termites, have eusocial behavior, but it is demonstrated in two rodent species: the naked mole-rat[195] and the Damaraland mole-rat.[196]
Presociality is when animals exhibit more than just sexual interactions with members of the same species, but fall short of qualifying as eusocial. That is, presocial animals can display communal living, cooperative care of young, or primitive division of reproductive labor, but they do not display all of the three essential traits of eusocial animals. Humans and some species of Callitrichidae (marmosets and tamarins) are unique among primates in their degree of cooperative care of young.[197] Harry Harlow set up an experiment with rhesus monkeys, presocial primates, in 1958; the results from this study showed that social encounters are necessary in order for the young monkeys to develop both mentally and sexually.[198]
A fission-fusion society is a society that changes frequently in its size and composition, making up a permanent social group called the "parent group". Permanent social networks consist of all individual members of a community and often varies to track changes in their environment. In a fission–fusion society, the main parent group can fracture (fission) into smaller stable subgroups or individuals to adapt to environmental or social circumstances. For example, a number of males may break off from the main group in order to hunt or forage for food during the day, but at night they may return to join (fusion) the primary group to share food and partake in other activities. Many mammals exhibit this, such as primates (for example orangutans and spider monkeys),[199] elephants,[200] spotted hyenas,[201] lions,[202] and dolphins.[203]
Solitary animals defend a territory and avoid social interactions with the members of its species, except during breeding season. This is to avoid resource competition, as two individuals of the same species would occupy the same niche, and to prevent depletion of food.[204] A solitary animal, while foraging, can also be less conspicuous to predators or prey.[205]

In a hierarchy, individuals are either dominant or submissive. A despotic hierarchy is where one individual is dominant while the others are submissive, as in wolves and lemurs,[206] and a pecking order is a linear ranking of individuals where there is a top individual and a bottom individual. Pecking orders may also be ranked by sex, where the lowest individual of a sex has a higher ranking than the top individual of the other sex, as in hyenas.[207] Dominant individuals, or alphas, have a high chance of reproductive success, especially in harems where one or a few males (resident males) have exclusive breeding rights to females in a group.[208] Non-resident males can also be accepted in harems, but some species, such as the common vampire bat (Desmodus rotundus), may be more strict.[209]
Some mammals are perfectly monogamous, meaning that they mate for life and take no other partners (even after the original mate’s death), as with wolves, Eurasian beavers, and otters.[210][211] There are three types of polygamy: either one or multiple dominant males have breeding rights (polygyny), multiple males that females mate with (polyandry), or multiple males have exclusive relations with multiple females (polygynandry). It is much more common for polygynous mating to happen, which, excluding leks, are estimated to occur in up to 90% of mammals.[212] Lek mating occurs in harems, wherein one or a few males protect their harem of females from other males who would otherwise mate with the females, as in elephant seals;[213] or males congregate around females and try to attract them with various courtship displays and vocalizations, as in harbor seals.[214]
All higher mammals (excluding monotremes) share two major adaptations for care of the young: live birth and lactation. These imply a group-wide choice of a degree of parental care. They may build nests and dig burrows to raise their young in, or feed and guard them often for a prolonged period of time. Many mammals are K-selectors, and invest more time and energy into their young than do r-selectors. When two animals mate, they both share an interest in the success of the offspring, though often to different extremes. Mammalian females, both r- and K-selectors, exhibit some degree of maternal aggression, another example of parental care, which may be targeted against other females of the species or the young of other females; however, some mammals may "aunt" the infants of other females, and care for them. Mammalian males may play a role in child rearing, as with tenrecs, however this varies species to species, even within the same genus. For example, the males of the southern pig-tailed macaque (Macaca nemestrina) do not participate in child care, whereas the males of the Japanese macaque (M. fuscata) do.[215]
Humans and other mammals
In human culture

Non-human mammals play a wide variety of roles in human culture. They are the most popular of pets, with tens of millions of dogs, cats and other animals including rabbits and mice kept by families around the world.[216][217][218] Mammals such as mammoths, horses and deer are among the earliest subjects of art, being found in Upper Paleolithic cave paintings such as at Lascaux.[219] Major artists such as Albrecht Dürer, George Stubbs and Edwin Landseer are known for their portraits of mammals.[220] Many species of mammals have been hunted for sport and for food; deer and wild boar are especially popular as game animals.[221][222][223] Mammals such as horses and dogs are widely raced for sport, often combined with betting on the outcome.[224][225] There is a tension between the role of animals as companions to humans, and their existence as individuals with rights of their own.[226] Mammals further play a wide variety of roles in literature,[227][228][229] film,[230] mythology, and religion.[231][232][233]
Uses and importance

Domestic mammals form a large part of the livestock raised for meat across the world. They include (2011) around 1.4 billion cattle, 1.2 billion sheep, 1 billion domestic pigs,[234][235] and (1985) over 700 million rabbits.[236] Working domestic animals including cattle and horses have been used for work and transport from the origins of agriculture, their numbers declining with the arrival of mechanised transport and agricultural machinery. In 2004 they still provided some 80% of the power for the mainly small farms in the third world, and some 20% of the world's transport, again mainly in rural areas. In mountainous regions unsuitable for wheeled vehicles, pack animals continue to transport goods.[237] Mammal skins provide leather for shoes, clothing and upholstery.[238] Wool from mammals including sheep, goats and alpacas has been used for centuries for clothing.[239][240] Mammals serve a major role in science as experimental animals, both in fundamental biological research, such as in genetics,[241] and in the development of new medicines, which must be tested exhaustively to demonstrate their safety.[242] Millions of mammals, especially mice and rats, are used in experiments each year.[243] A knockout mouse is a genetically modified mouse with an inactivated gene, replaced or disrupted with an artificial piece of DNA. They enable the study of sequenced genes whose functions are unknown.[244][245] A small percentage of the mammals are non-human primates, used in research for their similarity to humans.[246][247][248]
Charles Darwin, Jared Diamond and others have noted the importance of domesticated mammals in the Neolithic development of agriculture and of civilization, causing farmers to replace hunter-gatherers around the world.[lower-alpha 2][250] This transition from hunting and gathering to herding flocks and growing crops was a major step in human history. The new agricultural economies, based on domesticated mammals, caused "radical restructuring of human societies, worldwide alterations in biodiversity, and significant changes in the Earth's landforms and its atmosphere... momentous outcomes".[251]
Hybrids
Hybrids are offspring resulting from the breeding of two genetically distinct individuals, which usually will result in a high degree of heterozygosity, though hybrid and heterozygous are not synonymous. The deliberate or accidental hybridizing of two or more species of closely related animals through captive breeding is a human activity which has been in existence for millennia and has grown for economic purposes.[252] Hybrids between different subspecies within a species (such as between the Bengal tiger and Siberian tiger) are known as intra-specific hybrids. Hybrids between different species within the same genus (such as between lions and tigers) are known as interspecific hybrids or crosses. Hybrids between different genera (such as between sheep and goats) are known as intergeneric hybrids.[253] Natural hybrids will occur in hybrid zones, where two populations of species within the same genera or species living in the same or adjacent areas will interbreed with each other. Some hybrids have been recognized as species, such as the red wolf (though this is controversial).[254]
Artificial selection, the deliberate selective breeding of domestic animals, is being used to breed back recently extinct animals in an attempt to achieve an animal breed with a phenotype that resembles that extinct wildtype ancestor. A breeding-back (intraspecific) hybrid may be very similar to the extinct wildtype in appearance, ecological niche and to some extent genetics, but the initial gene pool of that wild type is lost forever with its extinction. As a result, bred-back breeds are at best vague look-alikes of extinct wildtypes, as Heck cattle are of the aurochs.[255]
Purebred wild species evolved to a specific ecology can be threatened with extinction[256] through the process of genetic pollution, the uncontrolled hybridization, introgression genetic swamping which leads to homogenization or out-competition from the heterosic hybrid species.[257] When new populations are imported or selectively bred by people, or when habitat modification brings previously isolated species into contact, extinction in some species, especially rare varieties, is possible.[258] Interbreeding can swamp the rarer gene pool and create hybrids, depleting the purebred gene pool. For example, the endangered wild water buffalo is most threatened with extinction by genetic pollution from the domestic water buffalo. Such extinctions are not always apparent from a morphological standpoint. Some degree of gene flow is a normal evolutionary process, nevertheless, hybridization threatens the existence of rare species.[259][260]
Threats

The loss of species from ecological communities, defaunation, is primarily driven by human activity.[261] This has resulted in empty forests, ecological communities depleted of large vertebrates.[262][263] In the Quaternary extinction event, the mass die-off of megafaunal variety coincided with the appearance of humans, suggesting a human influence. One hypothesis is that humans hunted large mammals, such as the woolly mammoth, into extinction.[264][265]
Various species are predicted to become extinct in the near future,[266] among them the rhinoceros,[267] primates,[268] pangolins,[269] and giraffes.[270] Hunting alone threatens hundreds of mammalian species around the world.[271][272] Scientists claim that the growing demand for meat is contributing to biodiversity loss as this is a significant driver of deforestation and habitat destruction; species-rich habitats, such as significant portions of the Amazon rainforest, are being converted to agricultural land for meat production.[273][274][275] According to the World Wildlife Fund's 2016 Living Planet Index, global wildlife populations have declined 58% since 1970, primarily due to habitat destruction, over-hunting and pollution. They project that if current trends continue, 67% of wildlife could disappear by 2020.[276][277] Another influence is over-hunting and poaching, which can reduce the overall population of game animals,[278] especially those located near villages,[279] as in the case of peccaries.[280] The effects of poaching can especially be seen in the ivory trade with African elephants.[281] Marine mammals are at risk from entanglement from fishing gear, notably cetaceans, with discard mortalities ranging from 65,000 to 86,000 individuals annually.[282]
Several courses of actions are being taken globally, notably the Convention on Biological Diversity, otherwise known as the Rio Accord, which includes 189 signatory countries that are focused on identifying endangered species and habitats.[283] Another notable conservation organization is the IUCN, which has a membership of over 1,200 governmental and non-governmental organizations.[284]
Recent extinctions can be directly attributable to human influences.[285][261] The IUCN characterizes 'recent' extinction as those that have occurred past the cut-off point of 1500,[286] and around 80 mammal species have gone extinct since that time and 2015.[287] Some species, such as the Père David's deer[288] are extinct in the wild, and survive solely in captive populations. Other species, such as the Florida panther, are ecologically extinct, surviving in such low numbers that they essentially have no impact on the ecosystem.[289]:318 Other populations are only locally extinct (extirpated), still existing elsewhere, but reduced in distribution,[289]:75–77 as with the extinction of gray whales in the Atlantic.[290]
Notes
- ↑ Decreased latency to approach the mirror, repetitious head circling and close viewing of the marked areas were considered signs of self-recognition since they do not have arms and cannot touch the marked areas.[193]
- ↑ Diamond discussed this matter further in his 1997 book Guns, Germs, and Steel.[249]
See also
- List of recently extinct mammals – during recorded history
- List of prehistoric mammals
- List of monotremes and marsupials
- List of placental mammals
- List of mammal genera – living mammals
- List of mammalogists
- Lists of mammals by population size
- Lists of mammals by region
- List of threatened mammals of the United States
- Mammals described in the 2000s
- Mammals in culture
- Prehistoric mammals
References
- ↑ Vaughan, Terry A.; Ryan, James M.; Czaplewski, Nicholas J. (2013). "Classification of Mammals". Mammalogy (6 ed.). Jones and Bartlett Learning. ISBN 978-1-284-03209-3.
- 1 2 Szalay, Frederick S. (1999). "Classification of Mammals above the Species Level: Review". Journal of Vertebrate Paleontology. 19 (1): 191–195. JSTOR 4523980. doi:10.1080/02724634.1999.10011133.
- 1 2 Wilson, D.E.; Reeder, D.M., eds. (2005). "Preface and introductory material". Mammal Species of the World: A Taxonomic and Geographic Reference (3rd ed.). Johns Hopkins University Press. p. xxvi. ISBN 978-0-8018-8221-0. OCLC 62265494.
- ↑ "Mammals". The IUCN Red List of Threatened Species. IUCN. April 2010. Retrieved 23 August 2016.
- ↑ Rowe, T. (1988). "Definition, diagnosis, and origin of Mammalia" (PDF). Journal of Vertebrate Paleontology. 8 (3): 241–264. doi:10.1080/02724634.1988.10011708.
- ↑ Lyell, Charles (1871). The Student's Elements of Geology. London: John Murray. p. 347. ISBN 978-1-345-18248-4.
- ↑ Cifelli, Richard L.; Davis, Brian M. (2003). "Marsupial origins". Science. 302 (5652): 1899–1900. PMID 14671280. doi:10.1126/science.1092272.
- ↑ Kemp, T. S. (2005). The Origin and Evolution of Mammals (PDF). United Kingdom: Oxford University Press. p. 3. ISBN 0-19-850760-7. OCLC 232311794.
- ↑ Datta, P. M. (2005). "Earliest mammal with transversely expanded upper molar from the Late Triassic (Carnian) Tiki Formation, South Rewa Gondwana Basin, India". Journal of Vertebrate Paleontology. 25 (1): 200–207. doi:10.1671/0272-4634(2005)025[0200:EMWTEU]2.0.CO;2.
- ↑ Luo, Zhe-Xi; Martin, Thomas (2007). "Analysis of Molar Structure and Phylogeny of Docodont Genera" (PDF). Bulletin of Carnegie Museum of Natural History. 39: 27–47. doi:10.2992/0145-9058(2007)39[27:AOMSAP]2.0.CO;2. Retrieved April 8, 2013.
- ↑ McKenna, Malcolm C.; Bell, Susan Groag (1997). Classification of Mammals above the Species Level. New York: Columbia University Press. ISBN 0-231-11013-8. OCLC 37345734.
- ↑ Nilsson, M. A.; Churakov, G.; Sommer, M.; van Tran, N.; Zemann, A.; Brosius, J.; Schmitz, J. (2010). "Tracking Marsupial Evolution Using Archaic Genomic Retroposon Insertions". PLoS Biology. 8 (7): e1000436. PMC 2910653
 . PMID 20668664. doi:10.1371/journal.pbio.1000436.
. PMID 20668664. doi:10.1371/journal.pbio.1000436. - ↑ Kriegs, Jan Ole; Churakov, Gennady; Kiefmann, Martin; Jordan, Ursula; Brosius, Jürgen; Schmitz, Jürgen (2006). "Retroposed Elements as Archives for the Evolutionary History of Placental Mammals". PLoS Biology. 4 (4): e91. PMC 1395351
 . PMID 16515367. doi:10.1371/journal.pbio.0040091.
. PMID 16515367. doi:10.1371/journal.pbio.0040091. - 1 2 Nishihara, H.; Maruyama, S.; Okada, N. (2009). "Retroposon analysis and recent geological data suggest near-simultaneous divergence of the three superorders of mammals". Proceedings of the National Academy of Sciences. 106 (13): 5235–5240. PMC 2655268
 . PMID 19286970. doi:10.1073/pnas.0809297106.
. PMID 19286970. doi:10.1073/pnas.0809297106. - ↑ Springer, Mark S.; Murphy, William J.; Eizirik, Eduardo; O'Brien, Stephen J. (2003). "Placental mammal diversification and the Cretaceous–Tertiary boundary". Proceedings of the National Academy of Sciences. 100 (3): 1056–1061. PMC 298725
 . PMID 12552136. doi:10.1073/pnas.0334222100.
. PMID 12552136. doi:10.1073/pnas.0334222100. - ↑ Tarver, James E.; dos Reis, Mario; Mirarab, Siavash; Moran, Raymond J.; Parker, Sean; O’Reilly, Joseph E.; King, Benjamin L.; O’Connell, Mary J.; Asher, Robert J.; Warnow, Tandy; Peterson, Kevin J.; Donoghue, Philip C. J.; Pisani, Davide (2016). "The Interrelationships of Placental Mammals and the Limits of Phylogenetic Inference". Genome Biology and Evolution. 8 (2): 330–344. doi:10.1093/gbe/evv261.
- 1 2 3 Springer, Mark S.; Meredith, Robert W.; Janecka, Jan E.; Murphy, William J. (2011). "The Historical Biogeography of Mammalia". Philosophical Transactions of the Royal Society B. 366 (1577): 2478–2502. PMC 3138613
 . PMID 21807730. doi:10.1098/rstb.2011.0023.
. PMID 21807730. doi:10.1098/rstb.2011.0023. - ↑ Jin Meng, Yuanqing Wang & Chuankui Li (2011). "Transitional mammalian middle ear from a new Cretaceous Jehol eutriconodont". Nature. 472 (7342): 181–185. Bibcode:2011Natur.472..181M. PMID 21490668. doi:10.1038/nature09921.
- ↑ Ahlberg, P. E. & Milner, A. R. (April 1994). "The Origin and Early Diversification of Tetrapods". Nature. 368 (6471): 507–514. Bibcode:1994Natur.368..507A. doi:10.1038/368507a0.
- ↑ "Amniota – Palaeos". Archived from the original on 2010-12-20.
- ↑ "Synapsida overview – Palaeos". Archived from the original on 2010-12-20.
- 1 2 3 Kemp, T. S. (2006). "The origin and early radiation of the therapsid mammal-like reptiles: a palaeobiological hypothesis" (PDF). Journal of Evolutionary Biology. 19 (4): 1231–47. PMID 16780524. doi:10.1111/j.1420-9101.2005.01076.x.
- 1 2 Bennett, A. F.; Ruben, J. A. (1986). "The metabolic and thermoregulatory status of therapsids". In Hotton III, N.; MacLean, P. D.; Roth, J. J.; Roth, E. C. The ecology and biology of mammal-like reptiles. Washington D. C.: Smithsonian Institution Press. pp. 207–218. ISBN 978-0-87474-524-5.
- ↑ Kermack, D.M.; Kermack, K.A. (1984). The evolution of mammalian characters. Washington D.C.: Croom Helm. ISBN 0-7099-1534-9. OCLC 10710687.
- ↑ Tanner LH, Lucas SG & Chapman MG (2004). "Assessing the record and causes of Late Triassic extinctions" (PDF). Earth-Science Reviews. 65 (1–2): 103–139. Bibcode:2004ESRv...65..103T. doi:10.1016/S0012-8252(03)00082-5. Archived from the original on October 25, 2007.
- ↑ Stephen L. Brusatte. "Superiority, Competition, and Opportunism in the Evolutionary Radiation of Dinosaurs".
- ↑ Gauthier, J.A. (1986). "Saurischian monophyly and the origin of birds". In Padian, K. (ed.). The Origin of Birds and the Evolution of Flight. Memoirs of the California Academy of Sciences. 8. San Francisco: California Academy of Sciences. pp. 1–55.
- ↑ Sereno, P.C. (1991). "Basal archosaurs: phylogenetic relationships and functional implications". Memoirs of the Society of Vertebrate Paleontology. 2: 1–53. JSTOR 3889336. doi:10.2307/3889336.
- ↑ MacLeod, N; Rawson, P. F.; Forey, P. L.; Banner, F. T.; Boudagher-Fadel, M. K.; Bown, P. R.; Burnett, J. A.; Chambers, P.; Culver, S.; Evans, S. E.; Jeffery, C.; Kaminski, M. A.; Lord, A. R.; Milner, A. C.; Milner, A. R.; Morris, N.; Owen, E.; Rosen, B. R.; Smith, A. B.; Taylor, P. D.; Urquhart, E.; Young, J. R. (1997). "The Cretaceous–Tertiary biotic transition". Journal of the Geological Society. 154 (2): 265–292. doi:10.1144/gsjgs.154.2.0265.
- ↑ Hunt, David M.; Hankins, Mark W.; Collin, Shaun P.; Marshall, N. J. Evolution of Visual and Non-visual Pigments. London: Springer. p. 73. ISBN 978-1-4614-4354-4. OCLC 892735337.
- ↑ Bakalar, Nicholas (2006). "Jurassic "Beaver" Found; Rewrites History of Mammals". National Geographic News. Retrieved 28 May 2016.
- ↑ Hall, M. I.; Kamilar, J. M.; Kirk, E. C. (24 October 2012). "Eye shape and the nocturnal bottleneck of mammals". Proceedings of the Royal Society B: Biological Sciences. 279 (1749): 4962–4968. PMC 3497252
 . PMID 23097513. doi:10.1098/rspb.2012.2258.
. PMID 23097513. doi:10.1098/rspb.2012.2258. - ↑ Luo, Zhe-Xi (2007). "Transformation and diversification in early mammal evolution". Nature. 450 (7172): 1011–19. Bibcode:2007Natur.450.1011L. PMID 18075580. doi:10.1038/nature06277.
- ↑ Pickrell, John (2003). "Oldest Marsupial Fossil Found in China". National Geographic News. Retrieved 28 May 2016.
- 1 2 Luo, Zhe-Xi; Yuan, Chong-Xi; Meng, Qing-Jin; Ji, Qiang (2011). "A Jurassic eutherian mammal and divergence of marsupials and placentals". Nature. 476 (7361): 442–445. Bibcode:2011Natur.476..442L. PMID 21866158. doi:10.1038/nature10291.
- ↑ Ji, Qiang; Luo, Zhe-Xi; Yuan, Chong-Xi; Wible, John R.; Zhang, Jian-Ping; Georgi, Justin A. (2002). "The earliest known eutherian mammal". Nature. 416: 816–822. PMID 11976675. doi:10.1038/416816a.
- ↑ M. J. Novacek; G. W. Rougier; J. R. Wible; M. C. McKenna; D. Dashzeveg & I. Horovitz (1997). "Epipubic bones in eutherian mammals from the Late Cretaceous of Mongolia". Nature. 389 (6650): 483–486. Bibcode:1997Natur.389..483N. PMID 9333234. doi:10.1038/39020.
- ↑ Power, Michael L.; Schulkin, Jay (2012). "Evolution of Live Birth in Mammals". Evolution of the Human Placenta. Baltimore: Johns Hopkins University Press. p. 68. ISBN 978-1-4214-0643-5.
- ↑ Rowe, Timothy; Rich, Thomas H.; Vickers-Rich, Patricia; Springer, Mark; Woodburne, Michael O. (2007). "The oldest platypus and its bearing on divergence timing of the platypus and echidna clades". Proceedings of the National Academy of Sciences. 105 (4): 1238–1242. PMC 2234122
 . PMID 18216270. doi:10.1073/pnas.0706385105.
. PMID 18216270. doi:10.1073/pnas.0706385105. - ↑ Grant, Tom (1995). "Reproduction". The Platypus: A Unique Mammal. Sydney: University of New South Wales. p. 55. ISBN 978-0-86840-143-0. OCLC 33842474.
- ↑ Goldman, Armond S. (2012). "Evolution of Immune Functions of the Mammary Gland and Protection of the Infant". Breastfeeding Medicine. 7 (3): 132–142. doi:10.1089/bfm.2012.0025.
- 1 2 Rose, Kenneth D. (2006). The Beginning of the Age of Mammals. Baltimore: Johns Hopkins University Press. pp. 82–83. ISBN 978-0-8018-8472-6. OCLC 646769601.
- ↑ Brink, A.S. (1955). "A study on the skeleton of Diademodon". Palaeontologia Africana. 3: 3–39.
- ↑ Kemp, T.S. (1982). Mammal-like reptiles and the origin of mammals. London: Academic Press. p. 363. ISBN 978-0-12-404120-2. OCLC 8613180.
- ↑ Estes, R. (1961). "Cranial anatomy of the cynodont reptile Thrinaxodon liorhinus". Bulletin of the Museum of Comparative Zoology (1253): 165–180.
- ↑ "Thrinaxodon: The Emerging Mammal". National Geographic Daily News. February 11, 2009. Retrieved August 26, 2012.
- 1 2 Bajdek, Piotr; Qvarnström, Martin; Owocki, Krzysztof; Sulej, Tomasz; Sennikov, Andrey G.; Golubev, Valeriy K.; Niedźwiedzki, Grzegorz (2015). "Microbiota and food residues including possible evidence of pre-mammalian hair in Upper Permian coprolites from Russia". Lethaia. doi:10.1111/let.12156.
- ↑ Botha-Brink, Jennifer; Angielczyk, Kenneth D. (2010). "Do extraordinarily high growth rates in Permo-Triassic dicynodonts (Therapsida, Anomodontia) explain their success before and after the end-Permian extinction?". Zoological Journal of the Linnean Society. 160 (2): 341–365. doi:10.1111/j.1096-3642.2009.00601.x.
- ↑ Paul, G.S. (1988). Predatory Dinosaurs of the World. New York: Simon and Schuster. p. 464. ISBN 978-0-671-61946-6. OCLC 18350868.
- ↑ J.M. Watson & J.A.M. Graves (1988). "Monotreme Cell-Cycles and the Evolution of Homeothermy". Australian Journal of Zoology. CSIRO. 36 (5): 573–584. doi:10.1071/ZO9880573.
- ↑ McNab, Brian K. (1980). "Energetics and the limits to the temperate distribution in armadillos". Journal of Mammalogy (American Society of Mammalogists). 61 (4): 606–627. JSTOR 1380307. doi:10.2307/1380307.
- ↑ Kielan−Jaworowska, Z.; Hurum, J.H.. (2006). "Limb posture in early mammals: Sprawling or parasagittal" (PDF). Acta Palaeontologica Polonica. 51 (3): 10237–10239.
- ↑ Lillegraven, Jason A.; Kielan-Jaworowska, Zofia; Clemens, William A. (1979). Mesozoic Mammals: The First Two-Thirds of Mammalian History. University of California Press. p. 321. ISBN 978-0-520-03951-3. OCLC 5910695.
- ↑ Oftedal, O.T. (2002). "The mammary gland and its origin during synapsid evolution". Journal of Mammary Gland Biology and Neoplasia. 7 (3): 225–252. PMID 12751889. doi:10.1023/A:1022896515287.
- ↑ Oftedal, O.T. (2002). "The origin of lactation as a water source for parchment-shelled eggs". Journal of Mammary Gland Biology and Neoplasia. 7 (3): 253–266. PMID 12751890. doi:10.1023/A:1022848632125.
- 1 2 Sahney, S., Benton, M.J. and Ferry, P.A. (2010). "Links between global taxonomic diversity, ecological diversity and the expansion of vertebrates on land" (PDF). Biology Letters. 6 (4): 544–547. PMC 2936204
 . PMID 20106856. doi:10.1098/rsbl.2009.1024.
. PMID 20106856. doi:10.1098/rsbl.2009.1024. - ↑ Smith, F. A.; Boyer, A. G.; Brown, J. H.; Costa, D. P.; Dayan, T.; Ernest, S. K. M.; Evans, A. R.; Fortelius, M.; Gittleman, J. L.; Hamilton, M. J.; Harding, L. E.; Lintulaakso, K.; Lyons, S. K.; McCain, C.; Okie, J. G.; Saarinen, J. J.; Sibly, R. M.; Stephens, P. R.; Theodor, J.; Uhen, M. D. (2010). "The Evolution of Maximum Body Size of Terrestrial Mammals". Science. 330 (6008): 1216–1219. Bibcode:2010Sci...330.1216S. doi:10.1126/science.1194830.
- ↑ Simmons, Nancy B.; Seymour, Kevin L.; Habersetzer, Jörg; Gunnell, Gregg F. (2007). "Primitive Early Eocene bat from Wyoming and the evolution of flight and echolocation". Nature. 451: 818–821. PMID 18270539. doi:10.1038/nature06549.
- ↑ Bininda-Emonds, O.R.P.; Cardillo, M.; Jones, K.E.; Beck, Robin M. D.; Grenyer, Richard; Price, Samantha A.; Vos, Rutger A.; et al. (2007). "The delayed rise of present-day mammals" (PDF). Nature. 446 (7135): 507–511. Bibcode:2007Natur.446..507B. PMID 17392779. doi:10.1038/nature05634.
- 1 2 Wible, J. R.; Rogier, G. W.; Novacek, M. J.; Asher, R. J. (2007). "Cretaceous eutherians and Laurasian origin for placental mammals near the K/T boundary". Nature. 447 (7147): 1003–06. Bibcode:2007Natur.447.1003W. PMID 17581585. doi:10.1038/nature05854.
- ↑ O'Leary, Maureen A.; Bloch, Jonathan I.; Flynn, John J.; Gaudin, Timothy J.; Giallombardo, Andres; Giannini, Norberto P.; Goldberg, Suzann L.; Kraatz, Brian P.; Luo, Zhe-Xi; Meng, Jin; Novacek, Michael J.; Perini, Fernando A.; Randall, Zachary S.; Rougier, Guillermo; Sargis, Eric J.; Silcox, Mary T.; Simmons, Nancy b.; Spaulding, Micelle; Velazco, Paul M.; Weksler, Marcelo; Wible, John r.; Cirranello, Andrea L.; Cirranello, Andrea L. (8 February 2013). "The Placental Mammal Ancestor and the Post–K-Pg Radiation of Placentals". Science. 339 (6120): 662–667. Bibcode:2013Sci...339..662O. PMID 23393258. doi:10.1126/science.1229237. Retrieved 9 February 2013.
- ↑ Halliday, Thomas J. D.; Upchurch, Paul; Goswami, Anjali (2015). "Resolving the relationships of Paleocene placental mammals". Biological Reviews. doi:10.1111/brv.12242.
- ↑ Halliday, Thomas John Dixon; Upchurch, Paul; Goswami, Anjali (2016). "Eutherians experienced elevated evolutionary rates in the immediate aftermath of the Cretaceous–Palaeogene mass extinction" (PDF). Proceedings of the Royal Society B. 283 (1833): 20153026. PMC 4936024
 . PMID 27358361. doi:10.1098/rspb.2015.3026.
. PMID 27358361. doi:10.1098/rspb.2015.3026. - 1 2 3 Ni, Xijun; Gebo, Daniel L.; Dagosto, Marian; Meng, Jin; Tafforeau, Paul; Flynn, John J.; Beard, K. Christopher (6 June 2013). "The oldest known primate skeleton and early haplorhine evolution". Nature. 498 (7452): 60–64. Bibcode:2013Natur.498...60N. PMID 23739424. doi:10.1038/nature12200.
- ↑ Romer, Sherwood A.; Parsons, Thomas S. (1977). The Vertebrate Body. Philadelphia: Holt-Saunders International. pp. 129–145. ISBN 978-0-03-910284-5. OCLC 60007175.
- ↑ Purves, William K.; Sadava, David E.; Orians, Gordon H.; Helle, H. C. (2001). Life: The Science of Biology (6 ed.). New York: Sinauer Associates, Inc. p. 593. ISBN 978-0-7167-3873-2. OCLC 874883911.
- ↑ Anthwal, Neal; Joshi, Leena; Tucker, Abigail S. (2012). "Evolution of the mammalian middle ear and jaw: adaptations and novel structures". Journal of Anatomy. 222 (1): 147–160. PMC 3552421
 . PMID 22686855. doi:10.1111/j.1469-7580.2012.01526.x.
. PMID 22686855. doi:10.1111/j.1469-7580.2012.01526.x. - ↑ van Nievelt, Alexander F. H.; Smith, Kathleen K. (2005). "To replace or not to replace: the significance of reduced functional tooth replacement in marsupial and placental mammals". Paleobiology. 31 (2): 324–346. doi:10.1666/0094-8373(2005)031[0324:trontr]2.0.co;2.
- ↑ Mao, Fangyuan; Wang, Yuanqing; Meng, Jin (2015). "A Systematic Study on Tooth Enamel Microstructures of Lambdopsalis bulla (Multituberculate, Mammalia) - Implications for Multituberculate Biology and Phylogeny". PLOS ONE. 10 (5): e0128243. PMC 4447277
 . PMID 26020958. doi:10.1371/journal.pone.0128243.
. PMID 26020958. doi:10.1371/journal.pone.0128243. - ↑ Osborn, Henry F. (1900). "Origin of the Mammalia, III. Occipital Condyles of Reptilian Tripartite Type". The American Naturalist. 34 (408): 943–947. JSTOR 2453526. doi:10.1086/277821.
- ↑ Crompton, A. W.; Jenkins, Jr., F. A. (1973). "Mammals from Reptiles: A Review of Mammalian Origins". Annual Review of Earth and Planetary Sciences. 1: 131–155. doi:10.1146/annurev.ea.01.050173.001023.
- 1 2 Power, Michael L.; Schulkin, Jay (2013). The Evolution Of The Human Placenta. Baltimore: Johns Hopkins University Press. pp. 1890–1891. ISBN 978-1-4214-0643-5. OCLC 940749490.
- ↑ Dierauf, Leslie A.; Gulland, Frances M. D. (2001). CRC Handbook of Marine Mammal Medicine: Health, Disease, and Rehabilitation (2 ed.). Boca Raton: CRC Press. p. 154. ISBN 978-1-4200-4163-7. OCLC 166505919.
- ↑ Lui, J. H.; Hansen, D. V.; Kriegstein, A. R. (2011). "Development and Evolution of the Human Neocortex". Cell. 146 (1): 18–36. PMC 3610574
 . PMID 21729779. doi:10.1016/j.cell.2011.06.030.
. PMID 21729779. doi:10.1016/j.cell.2011.06.030. - ↑ Keeler, Clyde E. (1933). "Absence of the Corpus callosum as a Mendelizing Character in the House Mouse". Proceedings of the National Academy of Sciences of the United States of America. 19 (6): 609–11. Bibcode:1933PNAS...19..609K. JSTOR 86284. PMC 1086100
 . PMID 16587795. doi:10.1073/pnas.19.6.609.
. PMID 16587795. doi:10.1073/pnas.19.6.609. - ↑ Levitzky, Michael G. (2013). "Mechanics of Breathing". Pulmonary physiology (8 ed.). New York: McGraw-Hill Medical. ISBN 978-0071793131. OCLC 940633137.
- 1 2 Umesh, Kumar B. (2011). "Pulmonary Anatomy and Physiology". Handbook of Mechanical Ventilation (1 ed.). New Delhi: Jaypee Brothers Medical Publishing. p. 12. ISBN 978-93-80704-74-6. OCLC 945076700.
- ↑ Standring, Susan; Borley, Neil R. (2008). Gray's anatomy: the anatomical basis of clinical practice (40 ed.). London: Churchill Livingstone. pp. 960–962. ISBN 978-0-8089-2371-8. OCLC 213447727.
- ↑ Betts, J. Gordon; Desaix, Peter; Johnson, Eddie; Johnson, Jody E.; Korol, Oksana; Kruse, Dean; Poe, Brandon; Wise, James A.; Womble, Mark; Young, Kelly A. (2013). Anatomy & physiology. Houston: Rice University Press. pp. 787–846. ISBN 978-1-938168-13-0. OCLC 898069394.
- 1 2 3 4 5 6 7 8 9 Feldhamer, George A.; Drickamer, Lee C.; Vessey, Stephen H.; Merritt, Joseph H.; Krajewski, Carey (2007). Mammalogy: Adaptation, Diversity, Ecology (3 ed.). Baltimore: Johns Hopkins University Press. ISBN 978-0-8018-8695-9. OCLC 124031907.
- ↑ Tinker, Spencer W. (1988). Whales of the World. Brill Archive. p. 51. ISBN 978-0-935848-47-2.
- ↑ Romer, A. S. (1959). The vertebrate story (4 ed.). Chicago: University of Chicago Press. ISBN 978-0-226-72490-4.
- ↑ de Muizon, Christian; Lange-Badré, Brigitte (1997). "Carnivorous dental adaptations in tribosphenic mammals and phylogenetic reconstruction". Lethaia. 30 (4): 353–366. doi:10.1111/j.1502-3931.1997.tb00481.x.
- ↑ Langer, Peter (1984). "Comparative Anatomy of the Stomach in Mammalian Herbivores". Quarterly Journal of Experimental Physiology. 69 (3): 615–625. PMID 6473699. doi:10.1113/expphysiol.1984.sp002848.
- ↑ Vaughan, Terry A.; Ryan, James M.; Czaplewski, Nicholas J. (2011). "Perissodactyla". Mammalogy (5 ed.). Jones and Bartlett. p. 322. ISBN 978-0-7637-6299-5. OCLC 437300511.
- ↑ Flower, William H.; Lydekker, Richard (1946). An Introduction to the Study of Mammals Living and Extinct. London: Adam and Charles Black. p. 496. ISBN 978-1-110-76857-8.
- ↑ Sreekumar, S. (2010). Basic Physiology. PHI Learning Pvt. Ltd. pp. 180–181. ISBN 978-8120-34107-4.
- ↑ Cheifetz, Adam S. (2010). Oxford American Handbook of Gastroenterology and Hepatology. Oxford: Oxford University Press, USA. p. 165. ISBN 0199830126.
- ↑ Kuntz, Erwin (2008). Hepatology: Textbook and Atlas. Germany: Springer. p. 38. ISBN 978-3-540-76838-8.
- ↑ Ortiz, Rudy M. (2001). "Osmoregulation in Marine Mammals". Journal of Experimental Biology. 204 (11): 1831–1844. PMID 11441026.
- 1 2 3 Roman, Alfred Sherwood; Parsons, Thomas S. (1977). The Vertebrate Body. Philadelphia: Holt-Saunders International. pp. 396–399. ISBN 978-0-03-910284-5.
- ↑ Biological Reviews - Cambridge Journals
- ↑ Dawkins, R.; Wong, Y. (2016). The Ancestor's Tale: A Pilgrimage to the Dawn of Evolution (2nd ed.). Boston: Mariner Books. p. 281. ISBN 978-0-544-85993-7.
- 1 2 3 Fitch, W. T. (2006). "Production of Vocalizations in Mammals". In Brown, K. Encyclopedia of Language and Linguistics (PDF). Oxford: Elsevier. pp. 115–121.
- ↑ Langevin, Paul; Barclay, Robert M. R. (1990). "Hypsignathus monstrosus". Mammalian Species. 357: 1–4. doi:10.2307/3504110.
- ↑ Weissengruber, G. E.; Forstenpointner, G.; Peters, G.; Kübber-Heiss, A.; W. T., Fitch (2002). "Hyoid apparatus and pharynx in the lion (Panthera leo), jaguar (Panthera onca), tiger (Panthera tigris), cheetah (Acinonyx jubatus), liger (Panthera leo × Panthera tigris), Tigon (Panthera tigris x Panthera leo) and the domestic cat. (Felis silvestris f. catus)". Journal of Anatomy. 201 (3): 195–209. PMC 1570911
 . PMID 12363272. doi:10.1046/j.1469-7580.2002.00088.x.
. PMID 12363272. doi:10.1046/j.1469-7580.2002.00088.x. - ↑ Stoeger, Angela S.; Heilmann, Gunnar; Zeppelzauer, Matthias; Ganswindt, André; Hensman, Sean; Charlton, Benjamin D. (2012). "Visualizing Sound Emission of Elephant Vocalizations: Evidence for Two Rumble Production Types". PLOS ONE. 7 (11): e48907. PMC 3498347
 . PMID 23155427. doi:10.1371/journal.pone.0048907.
. PMID 23155427. doi:10.1371/journal.pone.0048907. - ↑ Clark, C. W. (2004). "Baleen whale infrasonic sounds: Natural variability and function". Journal of the Acoustical Society of America. 115 (5): 2554. doi:10.1121/1.4783845.
- 1 2 Dawson, T. J.; Webster, K. N.; Maloney, S. K. (2014). "The fur of mammals in exposed environments; do crypsis and thermal needs necessarily conflict? The polar bear and marsupial koala compared". Journal of Comparative Physiology B. 184 (2): 273–284. doi:10.1007/s00360-013-0794-8.
- ↑ Caro, Tim (2005). "The Adaptive Significance of Coloration in Mammals" (PDF). BioScience. 55 (2): 125–136. doi:10.1641/0006-3568(2005)055[0125:tasoci]2.0.co;2.
- ↑ Caro, Tim (February 2009). "Contrasting coloration in terrestrial mammals". Phil Trans Royal Soc B. 364 (1516): 537–548. PMC 2674080
 . PMID 18990666. doi:10.1098/rstb.2008.0221.
. PMID 18990666. doi:10.1098/rstb.2008.0221. - ↑ Mills, L. Scott; Zimova, Marketa; Oyler, Jared; Running, Steven; Abatzoglou, John T.; Lukacs, Paul M. (April 2013). "Camouflage mismatch in seasonal coat color due to decreased snow duration". PNAS. 110 (8): 7360–7365. PMC 3645584
 . PMID 23589881. doi:10.1073/pnas.1222724110.
. PMID 23589881. doi:10.1073/pnas.1222724110. - ↑ Bradley et. al, Brenda (2012). "Coat Color Variation and Pigmentation Gene Expression in Rhesus Macaques (Macaca Mulatta)" (PDF). Journal of Mammalian Evolution. 20: 263–70. doi:10.1007/s10914-012-9212-3.
- ↑ Prum, Richard O.; Torres, Rodolfo H. (2004). "Structural colouration of mammalian skin: convergent evolution of coherently scattering dermal collagen arrays" (PDF). Journal of Experimental Biology. 207 (12): 2157–72. doi:10.1242/jeb.00989.
- ↑ Suutari, Milla; Majaneva, Markus; Fewer, David P.; Voirin, Bryson; Aiello, Annette; Friedl, Thomas; Chiarello, Adriano G.; Blomster, Jaanika (2010). "Molecular evidence for a diverse green algal community growing in the hair of sloths and a specific association with Trichophilus welckeri (Chlorophyta, Ulvophyceae)". Evolutionary Biology. 10 (86). PMC 2858742
 . PMID 20353556. doi:10.1186/1471-2148-10-86.
. PMID 20353556. doi:10.1186/1471-2148-10-86. - ↑ Plavcan, J. M. (2001). "Sexual dimorphism in primate evolution". American Journal of Physical Anthropology. 116 (33): 25–53. PMID 11786990. doi:10.1002/ajpa.10011.
- ↑ Maxwell, Kenneth E. (2013). The Sex Imperative: An Evolutionary Tale of Sexual Survival. Springer. pp. 112–13. ISBN 9781489959881.
- ↑ Vaughan, Terry A; et al. (2011). Mammalogy. Jones & Bartlett Publishers. p. 387. ISBN 9781449644376.
- ↑ Wallis M.C., Waters P.D., Delbridge M.L., Kirby P.J., Pask A.J., Grützner F., Rens W., Ferguson-Smith M.A., Graves J.A.M.; Waters; Delbridge; Kirby; Pask; Grützner; Rens; Ferguson-Smith; Graves; et al. (2007). "Sex determination in platypus and echidna: autosomal location of SOX3 confirms the absence of SRY from monotremes". Chromosome Research. 15 (8): 949–959. PMID 18185981. doi:10.1007/s10577-007-1185-3.
- ↑ Marshall Graves, Jennifer A. (2008). "Weird Animal Genomes and the Evolution of Vertebrate Sex and Sex Chromosomes" (PDF). Annual Review of Genetics. 42: 568–586. PMID 18983263. doi:10.1146/annurev.genet.42.110807.091714.
- ↑ Novacek, Michael J.; Rougier, Guillermo W.; Wible, John R.; McKenna, Malcolm C.; Dashzeveg, Demberelyin; Horovitz, Inés (1997). "Epipubic bones in eutherian mammals from the Late Cretaceous of Mongolia". Nature. 389: 483–486. PMID 9333234. doi:10.1038/39020.
- ↑ Morgan, Sally (2005). "Mammal Behavior and Lifestyle". Mammals. Chicago: Raintree. p. 6. ISBN 978-1-4109-1050-9. OCLC 53476660.
- ↑ Verma, P. S.; Pandey, B. P. (2013). ISC Biology Book I for Class XI. New Delhi: S. Chand and Company. p. 288. ISBN 978-81-219-2557-0.
- ↑ Oftedal, O. T. (2002). "The mammary gland and its origin during synapsid evolution". Journal of Mammary Gland Biology and Neoplasia. 7 (3): 225–252. PMID 12751889. doi:10.1023/a:1022896515287.
- ↑ Campbell, Neil A.; Reece, Jane B. (2002). Biology (6 ed.). Benjamin Cummings. p. 845. ISBN 978-080536-624-2. OCLC 47521441.
- ↑ Buffenstein, Rochelle; Yahav, Shlomo (1991). "Is the naked mole-rat Hererocephalus glaber an endothermic yet poikilothermic mammal?". Journal of Thermal Biology. 16 (4): 227–232. doi:10.1016/0306-4565(91)90030-6.
- ↑ Schmidt-Nielsen, Knut; Duke, James B. (1997). "Temperature Effects". Animal Physiology: Adaptation and Environment (5 ed.). Cambridge. p. 218. ISBN 978-0-521-57098-5. OCLC 35744403.
- 1 2 Lorenzini, A.; Johnson, F. B.; Oliver, A.; Tresini, M.; Smith, J. S.; Hdeib, M.; Sell, C.; Cristofalo, V. J.; Stamato, T. D. (2009). "Significant correlation of species longevity with DNA double strand break recognition but not with telomere length". Mech. Ageing Dev. 130 (11-12): 784–792. PMC 2799038
 . PMID 19896964. doi:10.1016/j.mad.2009.10.004.
. PMID 19896964. doi:10.1016/j.mad.2009.10.004. - ↑ Hart, R. W.; Setlow, R. B. (1974). "Correlation between deoxyribonucleic acid excision-repair and life-span in a number of mammalian species". Proceedings of the National Academy of Sciences of the U.S.A. 71 (6): 2169–2173. PMC 388412
 . PMID 4526202.
. PMID 4526202. - ↑ Ma S, Upneja A, Galecki A, Tsai YM, Burant CF, Raskind S, Zhang Q, Zhang ZD, Seluanov A, Gorbunova V, Clish CB, Miller RA, Gladyshev VN (2016). "Cell culture-based profiling across mammals reveals DNA repair and metabolism as determinants of species longevity". Elife. 5. PMC 5148604
 . PMID 27874830. doi:10.7554/eLife.19130.
. PMID 27874830. doi:10.7554/eLife.19130. - ↑ Grube K, Bürkle A (1992). "Poly(ADP-ribose) polymerase activity in mononuclear leukocytes of 13 mammalian species correlates with species-specific life span". Proceedings National Academy Sciences of the U.S.A. 89 (24): 11759–11763. PMC 50636
 . PMID 1465394.
. PMID 1465394. - ↑ Francis, A. A.; Lee, W. H.; Regan, J. D. (1981). "The relationship of DNA excision repair of ultraviolet-induced lesions to the maximum life span of mammals". Mechanisms of Ageing of Development. 16 (2): 181–189. PMID 7266079.
- ↑ Treton, J. A.; Courtois, Y. (1982). "Correlation between DNA excision repair and mammalian lifespan in lens epithelial cells". Cell Biol. Int. Rep. 6 (3): 253–60. PMID 7060140.
- ↑ Maslansky, C. J.; Williams, G. M. (1985). "Ultraviolet light-induced DNA repair synthesis in hepatocytes from species of differing longevities". Mechanisms of Ageing of Development. 29 (2): 191–203. PMID 3974310.
- ↑ "Leg and foot". Archived from the original on 2008-04-04. Retrieved 3 August 2008.
- ↑ Walker, Warren F.; Homberger, Dominique G. (1998). Anatomy and Dissection of the Fetal Pig (5 ed.). New York: W. H. Freeman and Company. p. 3. ISBN 978-0-7167-2637-1. OCLC 40576267.
- ↑ Orr, CM. (2005). "Knuckle-walking anteater: a convergence test of adaptation for purported knuckle-walking features of African Hominidae". Am. J. Phys. Anthropol. 128 (3): 639–58. PMID 15861420. doi:10.1002/ajpa.20192.
- ↑ Fish, FE; Frappell, PB; Baudinette, RV; MacFarlane, PM (2001). "Energetics of terrestrial locomotion of the platypus Ornithorhynchus anatinus" (PDF). The Journal of Experimental Biology. 204 (Pt 4): 797–803. PMID 11171362.
- ↑ Dhingra, P. (2004). "Comparative Bipedalism – How the Rest of the Animal Kingdom Walks on two legs". Anthropological Science. 131 (231).
- ↑ Alexander, R. M. (2004). "Bipedal animals, and their differences from humans". Journal of Anatomy. 204 (5): 321–330. PMC 1571302
 . PMID 15198697. doi:10.1111/j.0021-8782.2004.00289.x.
. PMID 15198697. doi:10.1111/j.0021-8782.2004.00289.x. - 1 2 Dagg, Anne I. (1973). "Gaits in Mammals". Mammal Review. 3 (4): 135–154. doi:10.1111/j.1365-2907.1973.tb00179.x.
- ↑ Roberts, Tristan D. M. (1995). Understanding Balance: The Mechanics of Posture and Locomotion. San Diego: Nelson Thornes. p. 211. ISBN 978-1-56593-416-0. OCLC 33167785.
- 1 2 3 Cartmill, M. (1985). "Climbing". In Hildebrand, M.; Bramble, D. M.; Liem, K. F.; Wake, D. B. Functional Vertebrate Morphology. Cambridge: Belknap Press. pp. 73–88. ISBN 978-0-674-32775-7. OCLC 11114191.
- ↑ Vernes, Karl (2001). "Gliding Performance of the Northern Flying Squirrel (Glaucomys sabrinus) in Mature Mixed Forest of Eastern Canada" (PDF). Journal of Mammalogy. 82 (4): 1026–1033. doi:10.1644/1545-1542(2001)082<1026:GPOTNF>2.0.CO;2.
- ↑ A. Barba, Lorena (October 2011). "Bats – the only flying mammals". Bio-Aerial Locomotion. Retrieved 20 May 2016.
- ↑ "Bats In Flight Reveal Unexpected Aerodynamics". ScienceDaily. 2007. Retrieved July 12, 2016.
- ↑ Hedenström, Anders; Johansson, L. C. (2015). "Bat flight: aerodynamics, kinematics and flight morphology" (PDF). Journal of Experimental Biology. 218: 653–663. PMID 25740899. doi:10.1242/jeb.031203.
- ↑ "Bats save energy by drawing in wings on upstroke". ScienceDaily. 2012. Retrieved July 12, 2016.
- ↑ Taschek, Karen (2008). Hanging with Bats: Ecobats, Vampires, and Movie Stars. Albuquerque, New Mexico: University of New Mexico Press. p. 14. ISBN 978-0-8263-4403-8. OCLC 191258477.
- ↑ Sterbing-D'Angeloa, Susanne; Chadhab, Mohit; Chiuc, Chen; Falkc, Ben; Xianc, Wei; Barceloc, Janna; Zookd, John M.; Mossa, Cynthia F. (2011). "Bat wing sensors support flight control" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 108 (27): 11291–11296. PMC 3131348
 . PMID 21690408. doi:10.1073/pnas.1018740108.
. PMID 21690408. doi:10.1073/pnas.1018740108. - ↑ Shimer, H. W. (1903). "Adaptations to Aquatic, Arboreal, Fossorial and Cursorial Habits in Mammals. III. Fossorial Adaptations". The American Naturalist. 37 (444): 819–825. JSTOR 2455381. doi:10.1086/278368.
- ↑ Perry, D. A. (1949). "The anatomical basis of swimming in Whales". Journal of Zoology. 119 (1): 49–60. doi:10.1111/j.1096-3642.1949.tb00866.x.
- ↑ Fish, F. E.; Hui, C. A. (1991). "Dolphin swimming — a review" (PDF). Mammal Review. 21 (4): 181–195. doi:10.1111/j.1365-2907.1991.tb00292.x.
- ↑ Marsh, Helene (1989). "Chapter 57: Dugongidae". Fauna of Australia (PDF). 1. Canberra: Australian Government Publications. ISBN 978-0-644-06056-1. OCLC 27492815. Archived from the original on 2013-05-11.
- 1 2 Berta, pp. 62–64.
- 1 2 Fish, F. E. (2003). "Maneuverability by the sea lion Zalophus californianus: Turning performance of an unstable body design". Journal of Experimental Biology. 206 (4): 667–74. PMID 12517984. doi:10.1242/jeb.00144.
- 1 2 Riedman, M. (1990). The Pinnipeds: Seals, Sea Lions, and Walruses. University of California Press. ISBN 978-0-520-06497-3. OCLC 19511610.
- ↑ Fish, F. E. (1996). "Transitions from drag-based to lift-based propulsion in mammalian swimming". Integrative and Comparative Biology. 36 (6): 628–41. doi:10.1093/icb/36.6.628.
- ↑ Fish, Frank E. (2000). "Biomechanics and Energetics in Aquatic and Semiaquatic Mammals: Platypus to Whale" (PDF). Physiological and Biochemical Zoology. 73 (6): 683–698. PMID 11121343. doi:10.1086/318108. Archived from the original (PDF) on 2016-08-04.
- ↑ Eltringham, S. K. (1999). "Anatomy and Physiology". The Hippos. London: T & AD Poyser Ltd. p. 8. ISBN 978-0-8566-1131-5. OCLC 42274422.
- ↑ "Hippopotamus Hippopotamus amphibius". National Geographic. Archived from the original on 2014-11-25. Retrieved 30 April 2016.
- 1 2 Seyfarth, R. M.; Cheney, D. L.; Marler, Peter (1980). "Vervet Monkey Alarm Calls: Semantic communication in a Free-Ranging Primate" (PDF). Animal Behaviour. 28 (4): 1070–1094. doi:10.1016/S0003-3472(80)80097-2.
- ↑ Zuberbühler, Klause (2001). "Predator-specific alarm calls in Campbell's monkeys, Cercopithecus campbelli". Behavioral Ecology and Sociobiology. 50 (5): 414–442. JSTOR 4601985. doi:10.1007/s002650100383.
- ↑ Slabbekoorn, Hans; Smith, Thomas B. (2002). "Bird song, ecology and speciation". Philosophical Transactions: Biology Sciences. 357 (1420): 493–503. doi:10.1098/rstb.2001.1056.
- ↑ Bannister, John L. (2008). "Baleen Whales (Mysticetes)". In F. Perrin, William; Würsig, Bernd; Thewissen, J. G. M. Encyclopedia of Marine Mammals (2 ed.). Academic Press. pp. 80–89. ISBN 978-0-12-373553-9.
- ↑ Norris, Scott (2002). "Creatures of Culture? Making the Case for Cultural Systems in Whales and Dolphins" (PDF). BioScience. 52 (1): 9–14. doi:10.1641/0006-3568(2002)052[0009:COCMTC]2.0.CO;2.
- ↑ Boughman, Janette W. (1998). "Vocal learning by greater spear-nosed bats". Proceedings: Biological Sciences. 265 (1392): 227–233. doi:10.1098/rspb.1998.0286.
- ↑ "Prairie dogs' language decoded by scientists". CBC News. 21 June 2013. Retrieved 20 May 2015.
- ↑ Mayell, Hillary (3 March 2004). "Elephants Call Long-Distance After-Hours". National Geographic. Retrieved 15 November 2016.
- ↑ Maynard Smith, John; Harper, David (2003). Animal Signals. Oxford Series in Ecology and Evolution. Oxford University Press. pp. 61–63. ISBN 978-0-19-852684-1. OCLC 54460090.
- ↑ FitzGibbon, C. D.; Fanshawe, J. H. (1988). "Stotting in Thomson’s gazelles: an honest signal of condition" (PDF). Behavioral Ecology and Sociobiology. 23 (2): 69–74. doi:10.1007/bf00299889. Archived from the original (PDF) on 2014-02-25.
- ↑ Bildstein, Keith L. (May 1983). "Why White-Tailed Deer Flag Their Tails". The American Naturalist. 121 (5): 709–715. JSTOR 2460873. doi:10.1086/284096.
- ↑ Johnson, Roger P. (August 1973). "Scent Marking in Mammals". Animal Behaviour. 21 (3): 521–535. doi:10.1016/S0003-3472(73)80012-0.
- ↑ Schevill, W.E.; McBride, A.F. (1956). "Evidence for echolocation by cetaceans". Deep-Sea Research. 3 (2): 153–154. Bibcode:1956DSR.....3..153S. doi:10.1016/0146-6313(56)90096-x.
- ↑ Wilson, W.; Moss, C. (2004). Thomas, J., ed. "Echolocation in Bats and Dolphins". Chicago University Press: 22. ISBN 978-0-226-79599-7. OCLC 50143737.
- ↑ Au, Whitlow W. L. (1993). The Sonar of Dolphins. Springer-Verlag. ISBN 978-3-540-97835-0. OCLC 26158593.
- ↑ Naugher, K. B. (2004). "Anteaters (Myrmecophagidae)". In Hutchins, M.; Kleiman, D. G.; Geist, V.; McDade, M. С. Grzimek's Animal Life Encyclopedia. 13 (2 ed.). Gale. pp. 171–179. ISBN 978-0-7876-7750-3. OCLC 471032508.
- ↑ Langer, Peter (1984). "Comparative Anatomy of the Stomach in Mammalian Herbivores". Quarterly Journal of Experimental Physiology. 69: 615–625. PMID 6473699. doi:10.1113/expphysiol.1984.sp002848.
- ↑ Sanders, Jon G.; Beichman, Annabel C.; Roman, Joe; Scott, Jarrod J.; Emerson, David; McCarthy, James J.; Girguis, Peter R. (2015). "Baleen whales host a unique gut microbiome with similarities to both carnivores and herbivores". Nature Communications. 6: 8285. PMC 4595633
 . PMID 26393325. doi:10.1038/ncomms9285.
. PMID 26393325. doi:10.1038/ncomms9285. - ↑ Speaksman, J. R. (1996). "Energetics and the evolution of body size in small terrestrial mammals" (PDF). Symposia of the Zoological Society of London (69): 69–81.
- 1 2 Don E. Wilson; David Burnie, eds. (2001). Animal: The Definitive Visual Guide to the World's Wildlife (1st ed.). DK Publishing. pp. 86–89. ISBN 978-0-7894-7764-4. OCLC 46422124.
- 1 2 van Valkenburgh, Blaire (2007). "Déjà vu: the evolution of feeding morphologies in the Carnivora". Integrative and Comparative Biology. 47 (1): 147–163. PMID 21672827. doi:10.1093/icb/icm016.
- ↑ Sacco, Tyson; van Valkenburgh, Blaire (2004). "Ecomorphological indicators of feeding behaviour in the bears (Carnivora: Ursidae)". Journal of Zoology. 263 (1): 41–54. doi:10.1017/S0952836904004856.
- ↑ Singer, M. S.; Bernays, E. A. (2003). "Understanding omnivory needs a behavioral perspective". Ecology. 84 (10): 2532–2537. doi:10.1890/02-0397.
- ↑ Hutson, Jarod M.; Burke, Chrissina C.; Haynes, Gary (2013-12-01). "Osteophagia and bone modifications by giraffe and other large ungulates". Journal of Archaeological Science. 40 (12): 4139–4149. doi:10.1016/j.jas.2013.06.004.
- ↑ "Why Do Cats Eat Grass?". Pet MD. Retrieved 13 January 2017.
- ↑ Geiser, Fritz (2004). "Metabolic Rate and Body Temperature Reduction During Hibernation and Daily Torpor". Annu. Rev. Physiol. 66: 239–274. PMID 14977403. doi:10.1146/annurev.physiol.66.032102.115105.
- ↑ Humphries, M. M.; Thomas, D.W.; Kramer, D.L. (2003). "The role of energy availability in mammalian hibernation: A cost-benefit approach". Physiological and Biochemical Zoology. 76 (2): 165–179. PMID 12794670. doi:10.1086/367950.
- ↑ Barnes, Brian M. (1989). "Freeze Avoidance in a Mammal: Body Temperatures Below 0 °C in an Arctic Hibernator". Science. 244 (4912): 1593–1595. PMID 2740905. doi:10.1126/science.2740905.
- ↑ Geiser, Fritz (2010). "Aestivation in Mammals and Birds". In Navas, Carlos Arturo; Carvalho, José Eduardo. Aestivation: Molecular and Physiological Aspects. Springer-Verlag. pp. 95–113. ISBN 978-3-642-02420-7. doi:10.1007/978-3-642-02421-4.
- ↑ Mann, Janet; Patterson, Eric M. (2013). "Tool Use by Aquatic Animals" (PDF). Philosophical Transactions of the Royal Society B. 368 (1630): 20120424. doi:10.1098/rstb.2012.0424.
- ↑ Raffaele, Paul (2011). Among the Great Apes: Adventures on the Trail of Our Closest Relatives. New York: Harper. p. 83. ISBN 978-0061671-84-5. OCLC 674694369.
- ↑ Köhler, Wolfgang (1925). The Mentality of Apes. Liveright. ISBN 978-0-87140-108-3. OCLC 2000769.
- ↑ McGowan, R. T.; Rehn, T.; Norling, Y.; Keeling, L. J. (2014). "Positive affect and learning: exploring the "Eureka Effect" in dogs". Animal Cognition. 17 (13): 577–587. PMID 24096703. doi:10.1007/s10071-013-0688-x.
- ↑ Karbowski, Jan (2007). "Global and regional brain metabolic scaling and its functional consequences". BioMed Central Biology. 5 (18). doi:10.1186/1741-7007-5-18.
- ↑ Marino, Lori (2007). "Cetacean Brains: How Aquatic Are They?". The Anatomical Record. 290 (6): 694–700. doi:10.1002/ar.20530.
- ↑ Gallup, Jr., G. G. (1970). "Chimpanzees: Self recognition". Science. 167 (3914): 86–87. Bibcode:1970Sci...167...86G. PMID 4982211. doi:10.1126/science.167.3914.86.
- ↑ Plotnik, J.M., de Waal, F.B.M. and Reiss, D. (2006). "Self-recognition in an Asian elephant" (PDF). PNAS. 103 (45): 17053–17057. Bibcode:2006PNAS..10317053P. doi:10.1073/pnas.0608062103.
- ↑ S., Robert (1986). "Ontogeny of mirror behavior in two species of great apes". American Journal of Primatology. 10 (2): 109–117. doi:10.1002/ajp.1350100202.
- ↑ Walraven, V., van Elsacker, L. and Verheyen, R. (1995). "Reactions of a group of pygmy chimpanzees (Pan paniscus) to their mirror images: evidence of self-recognition". Primates. 36: 145–150. doi:10.1007/bf02381922.
- ↑ Leakey, Richard (1994). "The Origin of the Mind". The Origin Of Humankind. New York: BasicBooks. p. 150. ISBN 978-0-465-05313-1. OCLC 30739453.
- ↑ Archer, John (1992). Ethology and Human Development. Rowman & Littlefield. pp. 215–218. ISBN 978-0-389-20996-6. OCLC 25874476.
- 1 2 Marten, K.; Psarakos, S. (1995). "Evidence of self-awareness in the bottlenose dolphin (Tursiops truncatus)". In Parker, S.T.; Mitchell, R.; Boccia, M. Self-awareness in Animals and Humans: Developmental Perspectives. Cambridge: Cambridge University Press. pp. 361–379. ISBN 978-0-521-44108-7. OCLC 28180680.
- 1 2 Delfour, F. & Marten, K. (2001). "Mirror image processing in three marine mammal species: Killer whales (Orcinus orca), false killer whales (Pseudorca crassidens) and California sea lions (Zalophus californianus)". Behavioural Processes. 53 (3): 181–190. PMID 11334706. doi:10.1016/s0376-6357(01)00134-6.
- ↑ Jarvis, J. U. M. (1981). "Eusociality in a mammal: cooperative breeding in naked mole-rat colonies". Science. 212 (4494): 571–573. JSTOR 1686202. doi:10.1126/science.7209555.
- ↑ Jacobs, D.S.; et al. (1991). "The colony structure and dominance hierarchy of the Damaraland mole-rat, Cryptomys damarensis (Rodentia: Bathyergidae) from Namibia". Journal of Zoology. 224 (4): 553–576. doi:10.1111/j.1469-7998.1991.tb03785.x.
- ↑ Hardy, Sarah B. (2009). Mothers and Others: The Evolutionary Origins of Mutual Understanding. Boston: Belknap Press of Harvard University Press. pp. 92–93.
- ↑ Harlow, H. F.; Suomi, S. J. (1971). "Social Recovery by Isolation-Reared Monkeys". Proceedings of the National Academy of Sciences of the United States of America. 68 (7): 1534–1538. doi:10.1073/pnas.68.7.1534.
- ↑ van Schaik, Carel P. (1999). "The Socioecology of Fission-Fusion Sociality in Orangutans". Biomedical and Life Sciences. 40 (1): 69–86. doi:10.1007/BF02557703.
- ↑ Archie, Elizabeth A.; Cynthia J. Moss; Susan C. Alberts (March 2005). "The ties that bind: genetic relatedness predicts the fission and fusion of social groups in wild African elephants". Proceedings of the Royal Society B. 273: 513–522. PMC 1560064
 . PMID 16537121. doi:10.1098/rspb.2005.3361.
. PMID 16537121. doi:10.1098/rspb.2005.3361. - ↑ Smith, Jennifer E.; Sandra K. Memenis; Kay E. Holekamp (2007). "Rank-related partner choice in the fission–fusion society of the spotted hyena (Crocuta crocuta)" (PDF). Behavioral Ecology and Sociobiology. 61 (5): 753–765. doi:10.1007/s00265-006-0305-y.
- ↑ Matoba, Tomoyuki; Kutsukake, Nobuyuki; Hasegawa, Toshikazu (2013). Hayward, Matt, ed. "Head Rubbing and Licking Reinforce Social Bonds in a Group of Captive African Lions, Panthera leo". PLoS ONE. 8 (9): e73044. PMC 3762833
 . PMID 24023806. doi:10.1371/journal.pone.0073044.
. PMID 24023806. doi:10.1371/journal.pone.0073044. - ↑ Krützen, Michael; Barré, Lynne M.; Connor, Richard C.; Mann, Janet; Sherwin, William B. (2004). "‘O father: where art thou?’— Paternity assessment in an open fission–fusion society of wild bottlenose dolphins (Tursiops sp.) in Shark Bay, Western Australia". Molecular Ecology. 13 (7): 1975–1990. PMID 15189218. doi:10.1111/j.1365-294X.2004.02192.x.
- ↑ Martin, Claude (1991). The Rainforests of West Africa: Ecology — Threats — Conservation (1 ed.). Springer. ISBN 978-3-0348-7726-8. doi:10.1007/978-3-0348-7726-8.
- ↑ le Roux, Aliza; Michael I. Cherry; Lorenz Gygax (5 May 2009). "Vigilance behaviour and fitness consequences: comparing a solitary foraging and an obligate group-foraging mammal". Behavioral Ecology and Sociobiology. 63: 1097–1107. doi:10.1007/s00265-009-0762-1.
- ↑ Palagi, Elisabetta; Norscia, Ivan (2015). Samonds, Karen E., ed. "The Season for Peace: Reconciliation in a Despotic Species (Lemur catta)". PLoS ONE. 10 (11): e0142150. PMC 4646466
 . PMID 26569400. doi:10.1371/journal.pone.0142150.
. PMID 26569400. doi:10.1371/journal.pone.0142150. - ↑ East, Marion L.; Hofer, Heribert (2000). "Male spotted hyenas (Crocuta crocuta) queue for status in social groups dominated by females". Behavioral Ecology. 12 (15): 558–568. doi:10.1093/beheco/12.5.558.
- ↑ Samuels, A.; Silk, J. B.; Rodman, P. (1984). "Changes in the dominance rank and reproductive behavior of male bonnet macaques (Macaca radiate)". Animal Behaviour. 32: 994–1003. doi:10.1016/s0003-3472(84)80212-2.
- ↑ Delpietro, H.A.; Russo, R.G. (2002). "Observations of the common vampire bat (Desmodus rotundus) and the hairy-legged vampire bat (Diphylla ecaudata) in captivity". Mammalian Biology. 67 (2): 65–78. doi:10.1078/1616-5047-00011.
- ↑ Kleiman, Devra G. (1977). "Monogamy in Mammals". The Quarterly Review of Biology. 52 (1): 39–69. PMID 857268. doi:10.1086/409721.
- ↑ Holland, B.; Rice, W. R. (1998). "Perspective: Chase-Away Sexual Selection: Antagonistic Seduction vs. Resistance" (PDF). Evolution. 52: 1–7. doi:10.2307/2410914.
- ↑ Clutton-Brock, T. H. (1989). "Review Lecture: Mammalian Mating Systems". Proceedings of the Royal Society of London. Series B, Biological Sciences. 236 (1285): 339–372. PMID 2567517. doi:10.1098/rspb.1989.0027.
- ↑ Leboeuf, J. B. (1972). "Sexual behavior in the northern elephant seal Mirounga angustirostris". Behaviour. 41 (1): 1–26. JSTOR 4533425. PMID 5062032. doi:10.1163/156853972X00167.
- ↑ Boness, D. J.; Bowen, D.; Buhleier, B. M.; Marshall, G. J. (2006). "Mating tactics and mating system of an aquatic-mating pinniped: the harbor seal, Phoca vitulina" (PDF). Behavioral Ecology and Sociobiology. 61: 119–30. doi:10.1007/s00265-006-0242-9.
- ↑ Klopfer, P. H. (1981). "Origins of Parental Care". In Gubernick, D. J. Parental Care in Mammals. New York: Plenum Press. ISBN 978-1-4613-3150-6. OCLC 913709574.
- ↑ Murthy, Rekha; Bearman, Gonzalo; Brown, Sherrill; Bryant, Kristina (2015). "Animals in Healthcare Facilities: Recommendations to Minimize Potential Risks" (PDF). Infection Control and Hospital Epidemiology. 36 (5): 495–516. doi:10.1017/ice.2015.15.
- ↑ The Humane Society of the United States. "U.S. Pet Ownership Statistics". Retrieved 27 April 2012.
- ↑ USDA. "U.S. Rabbit Industry profile" (PDF). Retrieved 10 July 2013.
- ↑ McKie, Robin (26 May 2013). "Prehistoric cave art in the Dordogne". The Guardian. Retrieved 9 November 2016.
- ↑ Jones, Jonathan (27 June 2014). "The top 10 animal portraits in art". The Guardian. Retrieved 24 June 2016.
- ↑ "Deer Hunting in the United States: An Analysis of Hunter Demographics and Behavior Addendum to the 2001 National Survey of Fishing, Hunting, and Wildlife-Associated Recreation Report 2001-6". Fishery and Wildlife Service (USA). Retrieved 24 June 2016.
- ↑ "Recreational Hog Hunting Popularity Soaring". Gramd View Outdoors. Retrieved 24 June 2016.
- ↑ Nguyen, Jenny; Wheatley, Rick (2015). Hunting For Food: Guide to Harvesting, Field Dressing and Cooking Wild Game. F+W Media. pp. 6–77. ISBN 978-1-4403-3856-4. Chapters on hunting deer, wild hog (boar), rabbit, and squirrel.
- ↑ "Horse racing". Archived from the original on 21 December 2013. Retrieved 6 May 2014.
- ↑ Genders, Roy (1981). Encyclopaedia of Greyhound Racing. Pelham Books. ISBN 978-0-7207-1106-6. OCLC 9324926.
- ↑ Plous, S. (1993). "The Role of Animals in Human Society". Journal of Social Issues. 49 (1): 1–9. doi:10.1111/j.1540-4560.1993.tb00906.x.
- ↑ Fowler, Karen Joy (26 March 2014). "Top 10 books about intelligent animals". The Guardian. Retrieved 9 November 2016.
- ↑ Gamble, Nikki; Yates, Sally (2008). Exploring Children's Literature (2 ed.). Los Angeles: Sage. ISBN 978-1-4129-3013-0. OCLC 71285210.
- ↑ "Books for Adults". Seal Sitters. Retrieved 9 November 2016.
- ↑ Paterson, Jennifer (2013). "Animals in Film and Media". Oxford Bibliographies. doi:10.1093/obo/9780199791286-0044.
- ↑ Johns, Catherine (2011). Cattle: History, Myth, Art. London: The British Museum Press. ISBN 978-0-7141-5084-0. OCLC 665137673.
- ↑ Robert Hans van Gulik. Hayagrīva: The Mantrayānic Aspect of Horse-cult in China and Japan. Brill Archive. p. 9.
- ↑ Grainger, Richard (24 June 2012). "Lion Depiction across Ancient and Modern Religions". ALERT. Archived from the original on 23 September 2016. Retrieved November 6, 2016.
- ↑ "Graphic detail Charts, maps and infographics. Counting chickens". The Economist. 27 July 2011. Retrieved November 6, 2016.
- ↑ Cattle Today. "Breeds of Cattle at CATTLE TODAY". Cattle-today.com. Retrieved November 6, 2016.
- ↑ Lukefahr, S.D.; Cheeke, P.R. "Rabbit project development strategies in subsistence farming systems". Food and Agriculture Organization. Retrieved November 6, 2016.
- ↑ Pond, Wilson G. (2004). Encyclopedia of Animal Science. New York: CRC Press. pp. 248–250. ISBN 978-0-8247-5496-9. OCLC 57033325.
- ↑ "History of Leather". Moore & Giles. Retrieved 10 November 2016.
- ↑ Braaten, Ann W. (2005). "Wool". In Steele, Valerie. Encyclopedia of Clothing and Fashion. 3. Thomson Gale. pp. 441–443. ISBN 978-0-684-31394-8. OCLC 963977000.
- ↑ Quiggle, Charlotte. "Alpaca: An Ancient Luxury." Interweave Knits Fall 2000: 74-76.
- ↑ "Genetics Research". Animal Health Trust. Retrieved November 6, 2016.
- ↑ "Drug Development". Animal Research.info. Retrieved November 6, 2016.
- ↑ "EU statistics show decline in animal research numbers". Speaking of Research. 2013. Retrieved November 6, 2016.
- ↑ Helen R. Pilcher (2003). "It's a knockout". Nature. doi:10.1038/news030512-17. Retrieved November 6, 2016.
- ↑ Y Zan et al., Production of knockout rats using ENU mutagenesis and a yeast-based screening assay, Nat. Biotechnol. (2003).Archived June 11, 2010, at the Wayback Machine.
- ↑ "The supply and use of primates in the EU". European Biomedical Research Association. 1996. Archived from the original on 2012-01-17.
- ↑ Carlsson, H. E.; Schapiro, S. J.; Farah, I.; Hau, J. (2004). "Use of primates in research: A global overview". American Journal of Primatology. 63 (4): 225–237. PMID 15300710. doi:10.1002/ajp.20054.
- ↑ Weatherall, D., et al., (The Weatherall Committee) (2006). The use of non-human primates in research (PDF) (Report). London, UK: Academy of Medical Sciences. Archived from the original (PDF) on 2013-03-23.
- ↑ Diamond, J. M. (1997). "Part 2: The rise and spread of food production". Guns, Germs, and Steel: the Fates of Human Societies (1 ed.). New York: W.W. Norton & Company. ISBN 978-0-393-03891-0. OCLC 35792200.
- ↑ Larson, Greger; Burger, Joachim (April 2013). "A population genetics view of animal domestication" (PDF). Trends in Genetics. 29 (4): 197–205. doi:10.1016/j.tig.2013.01.003.
- ↑ Zeder, Melinda A. (August 2008). "Domestication and early agriculture in the Mediterranean Basin: Origins, diffusion, and impact". PNAS. 105 (33): 11597–11604. PMC 2575338
 . PMID 18697943. doi:10.1073/pnas.0801317105.
. PMID 18697943. doi:10.1073/pnas.0801317105. - ↑ Price, E. (2008). Principles and applications of domestic animal behavior: an introductory text. Sacramento: Cambridge University Press. ISBN 978-1-84593-398-2. OCLC 226038028.
- ↑ Taupitz, Jochen; Weschka, Marion (2009). CHIMBRIDS - Chimeras and Hybrids in Comparative European and International Research. Heidelberg: Springer. p. 13. ISBN 978-3-540-93869-9. OCLC 495479133.
- ↑ Chambers, Steven M.; Fain, Steven R.; Fazio, Bud; Amaral, Michael (2012). "An account of the taxonomy of North American wolves from morphological and genetic analyses". North American Fauna. 77: 2. doi:10.3996/nafa.77.0001.
- ↑ van Vuure, T. (2005). Retracing the Aurochs – History, Morphology and Ecology of an extinct wild Ox. Pensoft Publishers. ISBN 978-954-642-235-4. OCLC 940879282.
- ↑ Mooney, H. A.; Cleland, E. E. (2001). "The evolutionary impact of invasive species". PNAS. 98 (10): 5446–5451. Bibcode:2001PNAS...98.5446M. PMC 33232
 . PMID 11344292. doi:10.1073/pnas.091093398.
. PMID 11344292. doi:10.1073/pnas.091093398. - ↑ Le Roux, Johannes J.; Foxcroft, Llewellyn C.; Herbst, Marna; MacFadyen, Sandra (2014). "Genetic analysis shows low levels of hybridization between African wildcats (Felis silvestris lybica) and domestic cats (F. s. catus) in South Africa". Ecology and Evolution. 5 (2): 288–299. PMC 4314262
 . PMID 25691958. doi:10.1002/ece3.1275.
. PMID 25691958. doi:10.1002/ece3.1275. - ↑ Wilson, Andrew (2003). "Australia's state of the forests report". p. 107.
- ↑ Rhymer, J. M.; Simberloff, D. (November 1996). "Extinction by Hybridization and Introgression". Annual Review of Ecology and Systematics. Annual Reviews. 27: 83–109. doi:10.1146/annurev.ecolsys.27.1.83.
- ↑ Potts, Brad M. (2001). Barbour, Robert C.; Hingston, Andrew B., eds. Genetic pollution from farm forestry using eucalypt species and hybrids : a report for the RIRDC/L&WA/FWPRDC Joint Venture Agroforestry Program. Rural Industrial Research and Development Corporation of Australia. ISBN 978-0-642-58336-9. OCLC 48794104.
- 1 2 Dirzo, Rodolfo; Young, Hillary S.; Galetti, Mauro; Ceballos, Gerardo; Isaac, Nick J. B.; Collen, Ben (2014). "Defaunation in the Anthropocene" (PDF). Science. 345 (6195): 401–406. doi:10.1126/science.1251817.
- ↑ Primack, Richard (2014). Essentials of Conservation Biology (6 ed.). Sunderland, MA: Sinauer Associates, Inc. Publishers. pp. 217–245. ISBN 978-1-605-35289-3. OCLC 876140621.
- ↑ Vignieri, Sacha (2014). "Vanishing fauna". Science. 345 (6195): 392–395. doi:10.1126/science.345.6195.392.
- ↑ Burney, David A.; Flannery, Timothy F. (2005). "Fifty millennia of catastrophic extinctions after human contact" (PDF). Trends in Ecology and Evolution. 20 (7): 395–401. PMID 16701402. doi:10.1016/j.tree.2005.04.022. Archived from the original on 2010-06-10.
- ↑ Diamond, J. (1984). "Historic extinctions: a Rosetta stone for understanding prehistoric extinctions". In Martin, P. S.; Klein, R. G. Quaternary extinctions: A prehistoric revolution. Tucson: University of Arizona Press. pp. 824–862. ISBN 978-0-8165-1100-6. OCLC 10301944.
- ↑ 7 Iconic Animals Humans Are Driving to Extinction. Live Science. November 22, 2013.
- ↑ Poachers Drive Javan Rhino to Extinction in Vietnam by John R. Platt October 25, 2011 Scientific American
- ↑ Estrada, Alejandro; Garber, Paul A.; Rylands, Anthony B.; Roos, Christian; Fernandez-Duque, Eduardo; Di Fiore, Anthony; Anne-Isola Nekaris, K.; Nijman, Vincent; Heymann, Eckhard W.; Lambert, Joanna E.; Rovero, Francesco; Barelli, Claudia; Setchell, Joanna M.; Gillespie, Thomas R.; Mittermeier, Russell A.; Arregoitia, Luis Verde; de Guinea, Miguel; Gouveia, Sidney; Dobrovolski, Ricardo; Shanee, Sam; Shanee, Noga; Boyle, Sarah A.; Fuentes, Agustin; MacKinnon, Katherine C.; Amato, Katherine R.; Meyer, Andreas L. S.; Wich, Serge; Sussman, Robert W.; Pan, Ruliang; Kone, Inza; Li, Baoguo (January 18, 2017). "Impending extinction crisis of the world’s primates: Why primates matter". Science Advances. 3 (1): e1600946. doi:10.1126/sciadv.1600946.
- ↑ Fletcher, Martin (January 31, 2015). "Pangolins: why this cute prehistoric mammal is facing extinction". The Telegraph. Retrieved 3 February 2017.
- ↑ Carrington, Damian (December 8, 2016). "Giraffes facing extinction after devastating decline, experts warn". The Guardian. Retrieved 3 February 2017.
- ↑ Pennisi, Elizabeth (October 18, 2016). "People are hunting primates, bats, and other mammals to extinction". Science. Retrieved 3 February 2017.
- ↑ Ripple, William J.; Abernethy, Katharine; Betts, Matthew G.; Chapron, Guillaume; Dirzo, Rodolfo; Galetti, Mauro; Levi, Taal; Lindsey, Peter A.; Macdonald, David W.; Machovina, Brian; Newsome, Thomas M.; Peres, Carlos A.; Wallach, Arian D.; Wolf, Christopher; Young, Hillary (2016). "Bushmeat hunting and extinction risk to the world's mammals". Royal Society Open Science. 3: 1–16. doi:10.1098/rsos.160498.
- ↑ Williams, Mark; Zalasiewicz, Jan; Haff, P. K.; Schwägerl, Christian; Barnosky, Anthony D.; Ellis, Erle C. (2015). "The Anthropocene Biosphere". The Anthropocene Review. 2 (3): 196–219. doi:10.1177/2053019615591020.
- ↑ Morell, Virginia (August 11, 2015). "Meat-eaters may speed worldwide species extinction, study warns". Science. Retrieved 3 February 2017.
- ↑ Machovina, B.; Feeley, K. J.; Ripple, W. J. (2015). "Biodiversity conservation: The key is reducing meat consumption". Science of The Total Environment. 536: 419–431. PMID 26231772. doi:10.1016/j.scitotenv.2015.07.022.
- ↑ "World on track to lose two-thirds of wild animals by 2020, major report warns". The Guardian. Retrieved 3 February 2017.
- ↑ Report 2016: risk and resilience in a new era (Report). Living Planet. World Wildlife Fund. pp. 1–148. ISBN 978-2-940529-40-7. OCLC 961331618.
- ↑ Redford, K. H. (1992). "The empty forest" (PDF). BioScience. 42 (6): 412–422. JSTOR 1311860. doi:10.2307/1311860.
- ↑ Peres, Carlos A.; Nascimento, Hilton S. (2006). "Impact of Game Hunting by the Kayapo´ of South-eastern Amazonia: Implications for Wildlife Conservation in Tropical Forest Indigenous Reserves". Human Exploitation and Biodiversity Conservation. Topics in Biodiversity and Conservation. 3. pp. 287–313. ISBN 978-1-4020-5283-5. OCLC 207259298.
- ↑ Altrichter, M.; Boaglio, G. (2004). "Distribution and Relative Abundance of Peccaries in the Argentine Chaco: Associations with Human Factors". Biological Conservation. 116 (2): 217–225. doi:10.1016/S0006-3207(03)00192-7.
- ↑ "African Elephant". IUCN Red List of Threatened Species. Retrieved 3 February 2017.
- ↑ Alverson, D. L.; Freeburg, M. H.; Murawski, S. A.; Pope, J. G. (1996) [1994]. "Bycatch of Marine Mammals". A global assessment of fisheries bycatch and discards. Rome: Food and Agriculture Organization of the United Nations. ISBN 978-92-5-103555-9. OCLC 31424005.
- ↑ Glowka, Lyle; Burhenne-Guilmin, Françoise; Synge, Hugh; McNeely, Jeffrey A.; Gündling, Lothar (1994). IUCN environmental policy and law paper. Guide to the Convention on Biodiversity. International Union for Conservation of Nature. ISBN 978-2-8317-0222-3. OCLC 32201845.
- ↑ "About IUCN". International Union for Conservation of Nature. Retrieved 3 February 2017.
- ↑ Ceballos, Gerardo; Ehrlich, Paul R.; Barnosky, Anthony D.; García, Andrés; Pringle, Robert M.; Palmer, Todd M. (2015). "Accelerated modern human–induced species losses: Entering the sixth mass extinction". Science Advances. 1 (5): e1400253. doi:10.1126/sciadv.1400253.
- ↑ Fisher, Diana O.; Blomberg, Simon P. (2011). "Correlates of rediscovery and the detectability of extinction in mammals". Proceedings of the Royal Society B: Biological Sciences. 278 (1708): 1090–1097. PMC 3049027
 . PMID 20880890. doi:10.1098/rspb.2010.1579.
. PMID 20880890. doi:10.1098/rspb.2010.1579. - ↑ Ceballos, G.; Ehrlich, A. H.; Ehrlich, P. R. (2015). The Annihilation of Nature: Human Extinction of Birds and Mammals. Baltimore: Johns Hopkins University Press. p. 69. ISBN 978-1-4214-1718-9.
- ↑ Zhigang, J; Harris, RB (2008). "Elaphurus davidianus". IUCN Red List of Threatened Species. Version 2008. International Union for Conservation of Nature. Retrieved 2012-05-20.
- 1 2 McKinney, Michael L.; Schoch, Robert; Yonavjak, Logan (2013). "Conserving Biological Resources". Environmental Science: Systems and Solutions (5 ed.). Jones & Bartlett Learning. ISBN 978-1-4496-6139-7. OCLC 777948078.
- ↑ Perrin, William F.; Würsig, Bernd G.; Thewissen, J. G. M. (2009). Encyclopedia of marine mammals. Academic Press. p. 404. ISBN 978-0-12-373553-9. OCLC 455328678.
Further reading
- Brown W.M. (2001). "Natural selection of mammalian brain components". Trends in Ecology and Evolution. 16 (9): 471–473. doi:10.1016/S0169-5347(01)02246-7.
- Jaffa, Khalaf-von; Taher, Norman Ali Bassam Ali (2006). "Mammalia Palaestina: The Mammals of Palestine". The Palestinian Biological Bulletin (55): 1–46.
- McKenna, Malcolm C.; Bell, Susan K. (1997). Classification of Mammals Above the Species Level. New York: Columbia University Press. ISBN 978-0-231-11013-6. OCLC 37345734.
- Nowak, Ronald M. (1999). Walker's mammals of the world (6 ed.). Baltimore: Johns Hopkins University Press. ISBN 978-0-8018-5789-8. OCLC 937619124.
- Simpson, George Gaylord (1945). "The principles of classification and a classification of mammals". Bulletin of the American Museum of Natural History. 85: 1–350.
- Murphy, William J.; Eizirik, Eduardo; O'Brien, Stephen J.; Madsen, Ole; Scally, Mark; Douady, Christophe J.; Teeling, Emma; Ryder, Oliver A.; Stanhope, Michael J.; de Jong, Wilfried W.; Springer, Mark S. (2001). "Resolution of the Early Placental Mammal Radiation Using Bayesian Phylogenetics". Science. 294 (5550): 2348–2351. PMID 11743200. doi:10.1126/science.1067179.
- Springer, Mark S.; Stanhope, Michael J.; Madsen, Ole; de Jong, Wilfried W. (2004). "Molecules consolidate the placental mammal tree" (PDF). Trends in Ecology and Evolution. 19 (8): 430–438. PMID 16701301. doi:10.1016/j.tree.2004.05.006.
- Vaughan, Terry A.; Ryan, James M.; Capzaplewski, Nicholas J. (2000). Mammalogy (4 ed.). Fort Worth, Texas: Saunders College Publishing. ISBN 978-0-03-025034-7. OCLC 42285340.
- Ole Kriegs, Jan; Churakov, Gennady; Kiefmann, Martin; Jordan, Ursula; Brosius, Juergen; Schmitz, Juergen (2006). "Retroposed Elements as Archives for the Evolutionary History of Placental Mammals". PLoS Biol. 4 (4): e91. PMC 1395351
 . PMID 16515367. doi:10.1371/journal.pbio.0040091.
. PMID 16515367. doi:10.1371/journal.pbio.0040091. - MacDonald, David W.; Norris, Sasha (2006). The Encyclopedia of Mammals (3 ed.). London: Brown Reference Group. ISBN 978-0-681-45659-4. OCLC 74900519.
External links
- BBC Wildlife Finder – video clips from the BBC's natural history archive
- Biodiversitymapping.org – All mammal orders in the world with distribution maps
- Paleocene Mammals, a site covering the rise of the mammals, paleocene-mammals.de
- Evolution of Mammals, a brief introduction to early mammals, enchantedlearning.com
- Mammal Species, collection of information sheets about various mammal species, learnanimals.com
- European Mammal Atlas EMMA from Societas Europaea Mammalogica, European-mammals.org
- Marine Mammals of the World—An overview of all marine mammals, including descriptions, both fully aquatic and semi-aquatic, noaa.gov
- Mammalogy.org The American Society of Mammalogists was established in 1919 for the purpose of promoting the study of mammals, and this website includes a mammal image library









