Maillard reaction
The Maillard reaction (/maɪˈjɑːr/ my-YAR; French pronunciation: [majaʁ]) is a chemical reaction between amino acids and reducing sugars that gives browned food its distinctive flavor. Seared steaks, pan-fried dumplings, cookies and other kinds of biscuits, breads, toasted marshmallows, and many other foods, undergo this reaction. It is named after French chemist Louis-Camille Maillard, who first described it in 1912 while attempting to reproduce biological protein synthesis.[1][2]
The reaction is a form of non-enzymatic browning which typically proceeds rapidly from around 140 to 165 °C (280 to 330 °F). At higher temperatures, caramelization and subsequently pyrolysis become more pronounced.
The reactive carbonyl group of the sugar reacts with the nucleophilic amino group of the amino acid, and forms a complex mixture of poorly characterized molecules responsible for a range of aromas and flavors. This process is accelerated in an alkaline environment (e.g., lye applied to darken pretzels; see Lye roll), as the amino groups (RNH3+) are deprotonated and, hence, have an increased nucleophilicity. The type of the amino acid determines the resulting flavor. This reaction is the basis for many of the flavoring industry's recipes. At high temperatures, a potential carcinogen called acrylamide can be formed.[3]
In the process, hundreds of different flavor compounds are created. These compounds, in turn, break down to form yet more new flavor compounds, and so on. Each type of food has a very distinctive set of flavor compounds that are formed during the Maillard reaction. It is these same compounds that flavor scientists have used over the years to make artificial flavors.
History
In 1913 Maillard published a paper to explain what happens when amino acids react with sugars at elevated temperatures.[1] However, it was chemist John E. Hodge, working at the U.S. Department of Agriculture in Peoria, Illinois, who published a paper in 1953 that established a mechanism for the Maillard reaction.[4][5]
Foods and products with Maillard reactions

The Maillard reaction is responsible for many colors and flavors in foods:
- The browning of various meats like steak, when seared and grilled
- The browning and umami taste in fried onions
- Coffee roasting
- The darkened crust of baked goods like pretzels, bagels, and toast
- The golden-brown color of French fries and other crisps
- Malted barley, found in malt whiskey or beer
- Dried or condensed milk
- Dulce de leche
- Black garlic
- Chocolate
- Lightly roasted peanuts
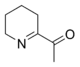
6-Acetyl-2,3,4,5-tetrahydropyridine is responsible for the biscuit or cracker-like flavor present in baked goods like bread, popcorn, and tortilla products. The structurally related compound 2-acetyl-1-pyrroline has a similar smell, and occurs also naturally without heating and gives varieties of cooked rice and the herb pandan (Pandanus amaryllifolius) their typical smells. Both compounds have odor thresholds below 0.06 ng/l.[6]


The browning reactions that occur when meat is roasted or seared are complicated, and occur mostly by Maillard browning[7] with contributions from other chemical reactions, including the breakdown of the tetrapyrrole rings of the muscle protein myoglobin.
Caramelization is an entirely different process from Maillard browning, though the results of the two processes are sometimes similar to the naked eye (and taste buds). Caramelization may sometimes cause browning in the same foods in which the Maillard reaction occurs, but the two processes are distinct. They both are promoted by heating, but the Maillard reaction involves amino acids, as discussed above, whereas caramelization is simply the pyrolysis of certain sugars.
In making silage, excess heat causes the Maillard reaction to occur, which reduces the amount of energy and protein available to the animals who feed on it.
Chemical mechanism
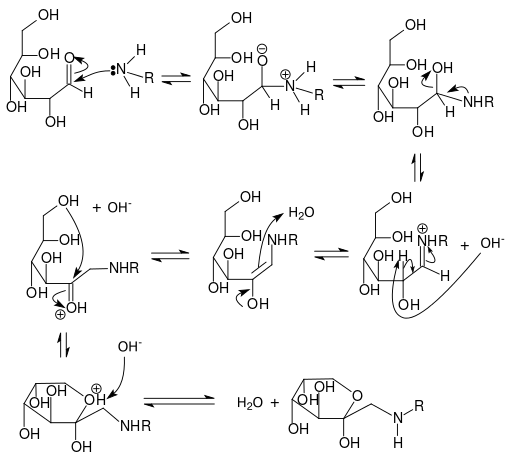
- The carbonyl group of the sugar reacts with the amino group of the amino acid, producing N-substituted glycosylamine and water
- The unstable glycosylamine undergoes Amadori rearrangement, forming ketosamines
- There are several ways for the ketosamines to react further:
- Produce 2 water and reductones
- Diacetyl, aspirin, pyruvaldehyde and other short-chain hydrolytic fission products can be formed
- Produce brown nitrogenous polymers and melanoidins
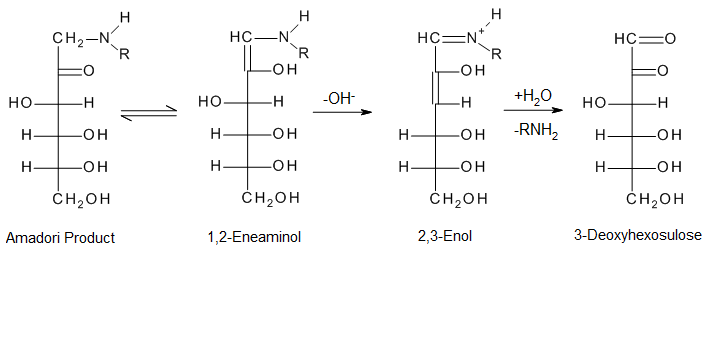
The open-chain Amadori product undergo further dehydration and de-amination to produce dicarbonyls.[8] This is a crucial intermediate.
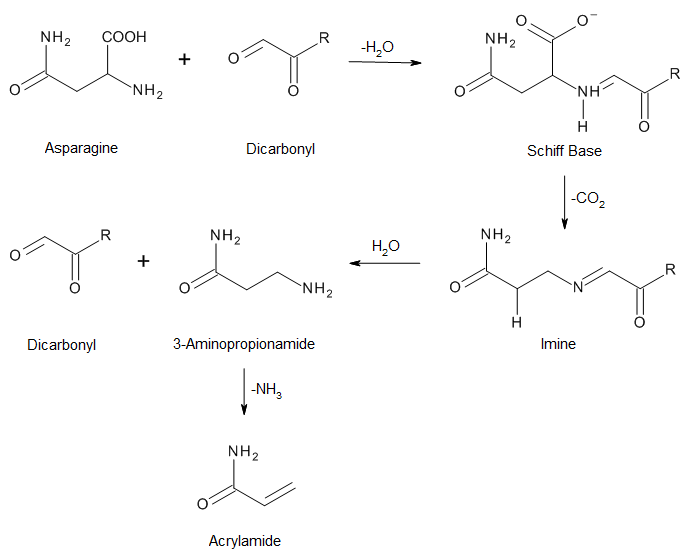
Dicarbonyls react with amine to produce Strecker aldehyde through Strecker degradation.[9]
Acrylamide, a possible human carcinogen,[10] can be generated as a by-product of Maillard reaction between reducing sugars and amino acids, especially asparagine, both of which are present in most food products.[11][12]
Physiology
The Maillard reaction also occurs in the human body. It is a step in the formation of advanced glycation endproducts (AGEs).[13] It is tracked by measuring pentosidine.
Although the Maillard reaction has been studied most extensively in foods, it has also shown a correlation in numerous different diseases in the human body, in particular degenerative eye diseases. In general, these diseases are due to the accumulation of AGEs on nucleic acids, proteins, and lipids. Though AGEs have numerous origins, they can form from the oxidation and dehydration of Amadori adducts, which themselves are products of nonenzymatic Maillard reactions. Apart from ocular diseases, whose correlation with Maillard chemistry has been more recently studied, the formation of AGEs has also proven to contribute to a wide range of human diseases that include diabetic complications, pulmonary fibrosis, and neurodegeneration. The positron emission tomography imaging agent fluorodeoxyglucose has been shown to undergo the Maillard reaction to form fluorodeoxyglycosylamine.
Receptor systems in the body have been suggested to have evolved to remove glycation-modified molecules, such as AGEs, to eliminate their effects. The adverse effects of AGE accumulation appear to be mediated by numerous different AGE receptors. Examples include AGE-R1, galectin-3, CD36, and, most noted, RAGE, the receptor for AGEs.
Advanced glycation in numerous different locations within the eye can prove detrimental. In the cornea, whose endothelial cells have been known to express RAGE and galectin-3, the accumulation of AGEs is associated with thickened corneal stroma, corneal edema, and morphological changes within patients with diabetes. Within the lens, Maillard chemistry has been studied extensively in the context of cataract formation. Advanced glycation is known to alter fiber membrane integrity in the lens, and dicarbonyl compounds are known to cause increased aggregate formation within the lens. This effect is exacerbated by both diabetes and aging. Furthermore, it is thought that AGE-inhibiting compounds are effective in preventing cataract formation in diabetics.
Glycation in Maillard reactions may lead to destabilization of the vitreous gel structure within the eye via unnecessary cross-linking between collagen fibrils. Again, this process is more strongly observed within diabetic patients.
Within the retina, the accumulation of AGEs in the drusen and Bruch's membrane has been associated with age, and has also been observed at a higher level among patients with age-related macular degeneration (AMD). This is manifested by the thickening of the Bruch's membrane. Furthermore, it has been observed that AGE levels increase with age within the lamina cribrosa, and the products of the Maillard reaction have been observed there, as well.
A wide range of ocular diseases, particularly diabetic retinopathy, may be prevented by the inhibition of the Maillard reaction. This may be achieved in numerous ways: preventing the formation of AGEs, reducing the effectiveness of the AGE signaling pathway and the receptor-ligand interactions, or breaking the AGE crosslinks. This latter method has already been achieved to some extent by the breaker alagebrium, though its effectiveness against retinopathy is unknown. Another method is by the use of amadorins, which are able to prevent the reaction of Amadori intermediates, which form into AGEs, by scavenging the reactive carbonyls.[14]
See also
References
- 1 2 Maillard, L. C. (1912). "Action des acides amines sur les sucres; formation de melanoidines par voie méthodique (Action of Amino Acids on Sugars. Formation of Melanoidins in a Methodical Way)". Compt. Rend. 154: 66.
- ↑ Chichester, C. O., ed. (1986). Advances in Food Research. Advances in Food and Nutrition Research. 30. Boston: Academic Press. p. 79. ISBN 0-12-016430-2.
- 1 2 Tareke, E.; Rydberg, P.; Karlsson, Patrik; Eriksson, Sune; Törnqvist, Margareta (2002). "Analysis of acrylamide, a carcinogen formed in heated foodstuffs". J. Agric. Food. Chem. 50 (17): 4998–5006. PMID 12166997. doi:10.1021/jf020302f.
- ↑ Hodge, J. E. (1953). "Dehydrated Foods, Chemistry of Browning Reactions in Model Systems". Journal of Agricultural and Food Chemistry. 1 (15): 928–43. doi:10.1021/jf60015a004.
- ↑ Everts, Sarah (October 1, 2012). "The Maillard Reaction Turns 100". Chemical & Engineering News. 90 (40): 58–60.
- ↑ Harrison, T. J.; v, G. R. (2005). "An expeditious, high-yielding construction of the food aroma compounds 6-acetyl-1,2,3,4-tetrahydropyridine and 2-acetyl-1-pyrroline". J. Org. Chem. 70 (26): 10872–4. PMID 16356012. doi:10.1021/jo051940a.
- ↑ McGee, Harold (2004). On Food and Cooking: The Science and Lore of the Kitchen. New York: Scribner. pp. 778–9. ISBN 978-0-684-80001-1.
- ↑ Nursten, H. E. The Maillard Reaction: Chemistry, Biochemistry, and Implications. Royal Society of Chemistry. ISBN 978-0-85404-964-6. doi:10.1039/9781847552570.
- ↑ Stadler RH, Robert F, Riediker S, Varga N, Davidek T, Devaud S, Goldmann T, Hau J, Blank I (2004). "In-depth mechanistic study on the formation of acrylamide and other vinylogous compounds by the Maillard reaction". Journal of Agricultural and Food Chemistry. 52 (17): 5550–8. PMID 15315399. doi:10.1021/jf0495486.
- ↑ Acrylamide. Cancer.org. Retrieved on 2016-07-24.
- ↑ Virk-Baker MK, Nagy TR, Barnes S, Groopman J (2014). "Dietary Acrylamide and Human Cancer: A Systematic Review of Literature". Nutrition and Cancer. 66 (5): 774–90. PMC 4164905
 . PMID 24875401. doi:10.1080/01635581.2014.916323.
. PMID 24875401. doi:10.1080/01635581.2014.916323. - ↑ Mottram DS, Wedzicha BL, Dodson AT (2002). "Acrylamide is formed in the Maillard reaction". Nature. 419 (6906): 448–9. PMID 12368844. doi:10.1038/419448a.
- ↑ Grandhee, SK; Monnier, VM (June 25, 1991). "Mechanism of formation of the Maillard protein cross-link pentosidine. Glucose, fructose, and ascorbate as pentosidine precursors". J. Biol. Chem. 266 (18): 11649–53. PMID 1904866.
- ↑ Stitt, Alan W. (2005). "The Maillard Reaction in Eye Diseases". Annals of the New York Academy of Sciences. 1043: 582–97. PMID 16037281. doi:10.1196/annals.1338.066.
Further reading
- Van Soest, Peter J. (1982). Nutritional Ecology of the Ruminant (2nd ed.). Ithaca, NY: Cornell University Press. ISBN 9780801427725. OCLC 29909839.
External links
| Wikimedia Commons has media related to Maillard reaction. |
- Course website on Maillard reaction (archived copy)
- Diagram of the Maillard Reaction
- Food-Info.net
- Official Website of the International Maillard Reaction Society
- The Thermochemical Joy of Cooking by Rebecca Smith Hurd (Wired Magazine, Issue 12.06 – June 2004)
- The secret of fresh beer by Roland Piquepaille (Emerging Tech – June 3, 2008)
- The Maillard Reaction at Modernist Cuisine

