Leishman stain
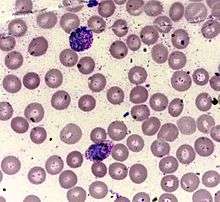
Leishman's stain (CAS:12627-53-1, EC: 235-732-1, MFCD:00131498), also Leishman stain, is used in microscopy for staining blood smears. It provides excellent stain quality. It is generally used to differentiate and identify leucocytes, malaria parasites, and trypanosomas. It is based on a methanolic mixture of "polychromed" methylene blue (i.e. demethylated into various azures) and eosin. The methanolic stock solution is stable and also serves the purpose of directly fixing the smear eliminating a prefixing step. If a working solution is made by dilution with an aqueous buffer the resulting mixture is very unstable and cannot be used for long. Leishman stain is named after its inventor, the Scottish pathologist William Boog Leishman. It is similar to and partially replaceable with Giemsa stain, Jenner's stain, and Wright's stain (See more details in Advantages disadvantages and comparisons section below). Like them, it is a version of Romanowsky stain.
Preparation[1] from Commercial source
Many companies sell good quality commercial Leishman Stain in the form of a dry powder. The following cautions should be taken while preparing.
- All glassware (the measuring cylinder and amber storage bottle) should be dried completely free of water before use.
- Both the Methanol bottle and the newly prepared Stain solution should be in containers with appropriate labels and precautions as Flammable and Toxic chemicals (Methanol if ingested can cause blindness and also is dangerous if directly contacts the eye, thus ideally, face shields, gloves, type N95 (US) or type P1 (EN143) respirator filter, are strongly recommended when a large volume of methanol is being used, as during making reagents in bulk.)
Under the above precautions the powder is to be mixed well into good quality anhydrous methanol in a proportion of 0.6g powder into 400 ml methanol. Using glass beads or a magnetic stirrer or carefully warming to 37 degrees C may help in dissolving.
'An aliquot of the stain (e.g. usually 50-100 ml) should be filtered into a dispensing unit for daily use, and the following storage conditions should be followed.'
Storage[2]
Bright light and heat oxidize the stain, especially when in aqueous solution and will cause precipitation of insoluble precipitates e.g. of methylene violet (Bernthsen). Evaporation of methanol, absorption of moisture and precipitation of Azure-Eosinate salts are also additional problems during storage that require filtering the stock while aliquotting for daily use.
For daily use, store the stain in an airtight (prevent moisture entering the stain) amber (semi-opaque) container. A closeable dropper bottle e.g. TK dropper bottle can be used that should be kept tightly closed when not in use. Keep in a cool place (not refrigerated) and never in direct sunlight.
The stock stain should be kept in a tightly stoppered light opaque (e.g. amber) container in a cool dark place. Renew every 3 months or earlier if indicated. To obtain optimum color reaction, some suggest that 3–5 days should be allowed before using freshly made stain.
Staining Methods
There are several ways of doing this stain. A common method[3] is:
1 Take an air dried blood smear on and glass slide and cover the smear with the undiluted stain. Take care not to overflow with excess stain. Preferably add just the enough number of drops to cover the smear. To standardize, count the number of drops (usually 7-10) required to cover the film (so that double the number of water can be added) and adjust the incubation time according to the result (usually 1–2 minutes, 3 minutes per WHO). The undiluted stain both acts as a fixative and also partially stains the smear. Still, since the moisture content can vary it is better to fix the slide in Methanol before staining.
2 Add twice the volume of pH 6.8 buffered water (i.e. if e.g. 7 drops of stain was used, then use 14 drops water) to dilute the stain, taking caution that the stain should not overflow (which will make the dilution inaccurate). However, the standard operating procedure published by WHO suggests adding equal (instead of twice) volume of water.[4] Mix the water with the stain underneath by gently blowing with a straw or using a plastic bulb pipette. Allow to stain for 10–12 minutes (time may require adjusting). In this method, better ionization during the dilution by the aqueous buffer in this step is necessary to complete the staining. The WHO protocol also clarifies that during this incubation
- "the appearance of a polychromatic 'scum' on the surface of the slide is merely a result of oxidation of the dye components and can be ignored."
3. Wash off the stain with clean (or filtered) tap water . If the stain is tiped off instead of washing, this will leave a fine deposit covering the film. Wipe the back of the slide clean and stand it in a draining rack to dry. The stained smear should grossly appear neither too pink nor too blue (verify final results microscopically). If the tap water is highly acidic, resulting in smear turning grossly pink too fast or highly alkaline, resulting in the smear remaining too blue, try using boiled cooled water or filtered rain water or pH 6.8 buffered water which can be used as an additional flooding step after washing in running water.
4. The slide should be air dried and can be viewed under microscope either directly or optionally (per WHO) a nonaqueous mounting medium such as DPX (Distrene 80 - a commercial polystyrene, a Plasticizer e.g. dibutyl phthalate and Xylene) may be used.
Advantages, disadvantages and comparisons
Advantages
Ease of technique
According to Sir John Vivian Dacie's famous text book of Practical Haematology:
- "Amongst the Romanowsky stains now in use, Jenner's is the simplest and Giemsa's the most complex. Leishman's stain, which occupies an intermediate position, is still widely used in the routine staining."
The WHO protocol [5] mentions:
- "There are a number of different combinations of these dyes which vary in their staining characteristics. May-Grunwald-Giemsa is a good method for routine work. Wright's stain is a simpler method, whilst Leishman's is also a simple method which is especially suitable when a stained blood film is required urgently or the routine stain is not available (e.g. at night). Field's stain is a rapid stain used primarily on thin films for malarial parasites. Whichever method is used, it is important to select dyes that are not contaminated with other dyes or metallic salts."
Sir William Boog Leishman of London and Karl Reuter of Germany independently discovered in 1901 what is considered "Perhaps the most practical modifications of Malachowski’s stain"[6] They adopted the best aspects of the stains developed by Malachowski and Jenner, i.e., they used both polychromed methylene blue (accidentally done by Romanowsky who got the name, systematically done by Ernst Malachowski even before Romanowsky, and rediscovered by Bernhard Nocht, but unknown to Jenner, May, Grunwald and many others who used simple methylene blue) and filtering the Azure Eosinate precipitate from the aqueous mixture and redissolving in an alcoholic solvent (Jenner's wisdom). Differences between Leishman's and Reuter's methods were: Leishman used methanol (like Jenner) and substituted Eosin B for Eosin Y, whereas Reuter used ethyl alcohol and rightly stressed the importance of using an absolutely pure solvent. Both methods produced a stable stain and the desired purple color.
Fairly good contrast
Leishman stain generally shows the brilliant violet color of the nucleus and the neutrophil granules for which differential count becomes convenient and makes the quality of staining better than the stains that are simple methylene blue and Eosin based which does not produce enough contrast between the cytoplasm and the nucleus. Since Leishman like other Romanowsky stains, stain cytoplasmic details and granules better hematologists generally prefer them (However see below about cytologists). Compared to the costly and toxic pure synthetic AZure B and Eosin Y based reagents used by the ICSH reference methods which are also not free from the disadvantage of getting oxidized and eventually giving grey tone instead of the optimal blue colors for cytoplasm, Leishman is a cheap easily available and easy to do technique which gives a fairly acceptable contrast.
Good sensitivity for Malaria parasite
It has been documented that Leishman staining is more sensitive than Field's stain and as good as fluorescent stains for detection of malaria parasite.[7]
Disadvantages
Difficulty controlling molar ratio
Composition of polychromed methylene blue mixed with Eosin is never as good as the directly weighed and mixed proportions in Giemsa type stains. Albert Plehn in 1890[8] had figured out that the molar ratio of basic to acidic dyes had to be increased from 2:1 to 3:1, however from Jenners time (1899) the use of the Azure Eosinate crystals brought the ratio back to the unsatisfactory 2:1 and the depth of color became less. It was only the work by Gustav Giemsa and the likes who again manually controlled the proportion of these two components that brought the depth of staining back. However Giemsa and others who artificially controlled the proportion sometimes went to the other extreme (a large molar excess of Azure up to a ratio of 16.1) which was probably unnecessary. per ICSH the optimal ratio is 6.5 to 7.3.[9]
Instability
The Leishman stain if reconstituted with buffer becomes very unstable (in contrast with Giemsa which is relatively more stable due to Glycerol, or the ICSH reference stains which use methanol+DMSO in 6:4 v/v ratio) and starts precipitating and needs repeated filtering. If not carefully supervised, water absorption, methanol evaporation from an opened container and the repeated filtering changes the composition and required frequent changing and makes this method wasteful. If not filtered, the precipitate deposits on the smear may be confused with platelets. It is important not to shake the bottle of stain before use, else settled precipitates would be resuspended and will deposit on films during staining and cause numerous artifacts and make microscopy very difficult.
Cytoplasmic contrast good but nuclear contrast not as good as H&E
Like all other Malachowski-Romanowsky-Giemsa methods, it fades with time and cannot be stably archived for long. Also its above counterparts it stains the nuclei dark purple and the nuclear feature details are not as clear as Hematoxylene and Eosin, which are thus preferred by some cytopathologists. But that way it is a complementary method (since it stains cytoplasmic details granules etc. better than H&E stain)
References
- ↑ District Laboratory Practice in Tropical Countries, Part 2 By Monica Cheesbrough, Appendix I, Preparation of Reagents and Culture Media, p396
- ↑ District Laboratory Practice in Tropical Countries, Part 2 By Monica Cheesbrough, Section 8 Hematological Tests, subsection 8.7 Blood Films, p322
- ↑ District Laboratory Practice in Tropical Countries, Part 2 By Monica Cheesbrough, Section 8 Hematological Tests, subsection 8.7 Blood Films
- ↑ Blood Safety and Clinical Technology Guidelines on Standard Operating Procedures for HAEMATOLOGY Chapter 11 - Preparation and Staining of Blood Films, (Last update: 27 April 2006)
- ↑ Blood Safety and Clinical Technology Guidelines on Standard Operating Procedures for HAEMATOLOGY Chapter 11 - Preparation and Staining of Blood Films, (Last update: 27 April 2006)
- ↑ Biotech Histochem. 2011 Feb;86(1):7-35. The Color Purple: from Royalty to Laboratory, with Apologies to Malachowski. Krafts KP, Hempelmann E, Oleksyn BJ.
- ↑ Indian Journal of Medical Microbiology, (2006) 24 (1):49-51
- ↑ Plehn F (1890b) Zur Aetiologie der Malaria. Berl. Klin. Wochenschr. 27: 292–294.
- ↑ ICSH reference method for staining of blood and bone marrow films by AZure B and Eosin Y (Romanowsky stain) INTERNATIONAL COMMITTEE FOR STANDARDIZATION IN HAEMATOLOGY Article first published online: 12 MAR 2008 DOI: 10.1111/j.1365-2141.1984.tb02949.x