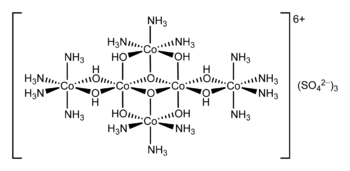
Hexol
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tris[tetrammine-μ-dihydroxocobalt(III)]cobalt (III) ion | |
| Identifiers | |
| Properties | |
| Co4H42N12O18S3 | |
| Molar mass | 830.31 g·mol−1 |
| Sparingly soluble in water [1] | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Hexol is the name for various salts of a coordination complex that has historical significance. The salts were the first synthetic non-carbon-containing chiral compounds.[2] The sulfate salt has the formula {[Co(NH3)4(OH)2]3Co}(SO4)3.
Preparation and optical resolution
Salts of hexol were first described by Jorgenson.[3] The salt is prepared by heating [Co(NH3)4(H2O)2]3+ with dilute base such as ammonia followed by precipitation of the sulfate salt:
- 4 [Co(NH3)4(H2O)2]3+ → {[Co(NH3)4(OH)2]3Co}6+ + 4 NH4+ + 2 H+ + 2 H2O
Depending on the conditions one obtains the 9-hydrate, the 6-hydrate, and the 4-hydrate. These salts exists as dark brownish-violet or black tabular crystals. The salts has low solubility in water. The cation can be quantitatively precipitated from its yellow-gray chromate and hexachloroplatinate salts. When treated with concentrated hydrochloric acid, hexol converts to cis-diaquotetramminecobalt(III) sulfate. In boiling dilute sulfuric acid, hexol degrades with evolution of oxygen and nitrogen.[1]
Optical resolution
In a historic set of experiments, Alfred Werner obtained chiral resolution by fractional crystallisation of the diastereomeric D-(+)-bromocamphorsulfonate salt. This ion has a high specific rotation of 2640°.[4] More efficient methods involve the bis(tartrato)diantimonate(III) salt.[5]
The "second hexol"
Werner also described a second achiral hexol (a minor byproduct from the production of Fremy's salt) that he incorrectly identified as a linear trimer. The second hexol is hexanuclear (contains six cobalt centres in each ion), not tetranuclear.[6]
References
- 1 2 Kauffman, George B.; Pinnell, Robert P. (1960). "Tris[Tetrammine-μ-Dihydroxo-Cobalt(III)] Cobalt(III) Sulfate 4-Hydrate". Inorganic Syntheses. 6: 176–179. ISBN 9780470132371. doi:10.1002/9780470132371.ch56.
- ↑ Miessler, G. L. and Tarr, D. A. Inorganic Chemistry, 3rd ed., Pearson/Prentice Hall publisher, ISBN 0-13-035471-6.
- ↑ Jörgensen, S. M. (1898). "Zur Konstitution der Kobalt-, Chrom- und Rhodiumbasen". Zeitschrift für anorganische Chemie. 16: 184–197. doi:10.1002/zaac.18980160116.
- ↑ Werner, A. (1907). "Über mehrkernige Metallammoniake" [Poly-nucleated Metal-amines]. Ber. Dtsch. Chem. Ges. (in German). 40 (2): 2103–2125. doi:10.1002/cber.190704002126.
- ↑ Yasui, Takaji; Ama, Tomoharu; Kauffman, George B. (1992). "Resolution of the Dodecaamminehexa-μ-Hydroxo-Tetracobalt(III) Ion". Inorganic Syntheses. 29: 169–174. ISBN 9780470132609. doi:10.1002/9780470132609.ch41.
- ↑ Jackson, W. Gregory; McKeon, Josephine A.; Zehnder, Margareta; Neuberger, Markus; Fallab, Silvio (2004). "The rediscovery of Alfred Werner's second hexol". Chemical Communications (20): 2322–2323. doi:10.1039/B408277J.
External links
- Hexol Molecule of the Month September 1997 Website
- National Pollutant Inventory – Cobalt fact sheet