Granulin
| GRN | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||
| Identifiers | |||||||||||||||||
| Aliases | GRN, CLN11, GEP, GP88, PCDGF, PEPI, PGranulin, granulin precursor | ||||||||||||||||
| External IDs | MGI: 95832 HomoloGene: 1577 GeneCards: GRN | ||||||||||||||||
| |||||||||||||||||
| RNA expression pattern | |||||||||||||||||
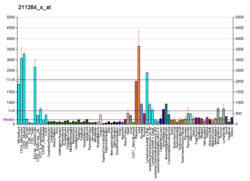 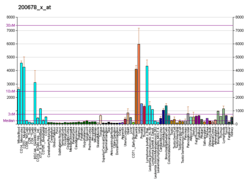 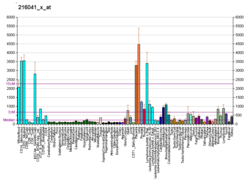 | |||||||||||||||||
| More reference expression data | |||||||||||||||||
| Orthologs | |||||||||||||||||
| Species | Human | Mouse | |||||||||||||||
| Entrez | |||||||||||||||||
| Ensembl | |||||||||||||||||
| UniProt | |||||||||||||||||
| RefSeq (mRNA) | |||||||||||||||||
| RefSeq (protein) |
| ||||||||||||||||
| Location (UCSC) | Chr 17: 44.35 – 44.35 Mb | Chr 11: 102.43 – 102.44 Mb | |||||||||||||||
| PubMed search | [1] | [2] | |||||||||||||||
| Wikidata | |||||||||||||||||
| |||||||||||||||||
| Granulin | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 the solution structure of a well-folded peptide based on the 31-residue amino-terminal subdomain of human granulin a | |||||||||
| Identifiers | |||||||||
| Symbol | Granulin | ||||||||
| Pfam | PF00396 | ||||||||
| InterPro | IPR000118 | ||||||||
| PROSITE | PDOC00634 | ||||||||
| SCOP | 1pcn | ||||||||
| SUPERFAMILY | 1pcn | ||||||||
| |||||||||
Granulin is a protein that in humans is encoded by the GRN gene.[3][4][5]
Structure
Granulins are a family of secreted, glycosylated peptides that are cleaved from a single precursor protein with 7.5 repeats of a highly conserved 12-cysteine granulin/epithelin motif. The 88 kDa precursor protein, progranulin, is also called proepithelin and prostate cancer (PC) cell-derived growth factor. Cleavage of the signal peptide produces mature granulin which can be further cleaved into a variety of active, 6 kDa peptides. These smaller cleavage products are named granulin A, granulin B, granulin C, etc. Epithelins 1 and 2 are synonymous with granulins A and B, respectively.
Function
Both the peptides and intact granulin protein regulate cell growth. However, different members of the granulin protein family may act as inhibitors, stimulators, or have dual actions on cell growth. Granulin family members are important in normal development, wound healing, and tumorigenesis.[5]
Clinical significance
The human liver fluke (Opisthorchis viverrini) contributes to the development of bile duct (liver) cancer by secreting a granulin-like growth hormone.[6]
Mutations in the GRN gene have been implicated in up to 25% of frontotemporal lobar degeneration, inherited in an autosomal dominant fashion with high penetrance.[7] Several loss-of-function mutations disease-causing mutations in GRN have been identified.[8][9]
Granulin release by macrophages has been associated with fibrotic hepatic metastasis in pancreatic cancer.[10]
Interactions
Granulin has been shown to interact with Cyclin T1[11] and TRIB3.[12]
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ Bhandari V, Bateman A (Nov 1992). "Structure and chromosomal location of the human granulin gene". Biochem Biophys Res Commun. 188 (1): 57–63. PMID 1417868. doi:10.1016/0006-291X(92)92349-3.
- ↑ Zhang H, Serrero G (Dec 1998). "Inhibition of tumorigenicity of the teratoma PC cell line by transfection with antisense cDNA for PC cell-derived growth factor (PCDGF, epithelin/granulin precursor)". Proc Natl Acad Sci U S A. 95 (24): 14202–7. Bibcode:1998PNAS...9514202Z. PMC 24351
 . PMID 9826678. doi:10.1073/pnas.95.24.14202.
. PMID 9826678. doi:10.1073/pnas.95.24.14202. - 1 2 "Entrez Gene: GRN granulin".
- ↑ Smout MJ, Laha T, Mulvenna J, Sripa B, Suttiprapa S, Jones A, Brindley PJ, Loukas A (October 2009). "A granulin-like growth factor secreted by the carcinogenic liver fluke, Opisthorchis viverrini, promotes proliferation of host cells". PLoS Pathog. 5 (10): e1000611. PMC 2749447
 . PMID 19816559. doi:10.1371/journal.ppat.1000611.
. PMID 19816559. doi:10.1371/journal.ppat.1000611. - ↑ Mackenzie IR (2007). "The neuropathology and clinical phenotype of FTD with progranulin mutations". Acta Neuropathologica. 114 (1): 49–54. PMID 17458552. doi:10.1007/s00401-007-0223-8.
- ↑ Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M (2006). "Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17". Nature. 442 (7105): 916–919. Bibcode:2006Natur.442..916B. PMID 16862116. doi:10.1038/nature05016.
- ↑ Cruts M, Gijselinck I, van der Zee J, Engelborghs S, Wils H, Pirici D, Rademakers R, Vandenberghe R, Dermaut B, Martin JJ, van Duijn C, Peeters K, Sciot R, Santens P, De Pooter T, Mattheijssens M, Van den Broeck M, Cuijt I, Vennekens K, De Deyn PP, Kumar-Singh S, Van Broeckhoven C (2006). "Null mutations in progranulin cause ubiquitin-positive frontotemporal dementia linked to chromosome 17q21". Nature. 442 (7105): 920–924. Bibcode:2006Natur.442..920C. PMID 16862115. doi:10.1038/nature05017.
- ↑ Nielsen, Sebastian R.; Quaranta, Valeria; Linford, Andrea; Emeagi, Perpetua; Rainer, Carolyn; Santos, Almudena; Ireland, Lucy; Sakai, Takao; Sakai, Keiko; Kim, Yong-Sam; Engle, Dannielle; Campbell, Fiona; Palmer, Daniel; Ko, Jeong Heon; Tuveson, David A.; Hirsch, Emilio; Mielgo, Ainhoa; Schmid, Michael C. (18 April 2016). "Macrophage-secreted granulin supports pancreatic cancer metastasis by inducing liver fibrosis". Nature Cell Biology. 18 (5): 549–560. doi:10.1038/ncb3340.
- ↑ Hoque M, Young TM, Lee CG, Serrero G, Mathews MB, Pe'ery T (March 2003). "The growth factor granulin interacts with cyclin T1 and modulates P-TEFb-dependent transcription". Mol. Cell. Biol. 23 (5): 1688–702. PMC 151712
 . PMID 12588988. doi:10.1128/MCB.23.5.1688-1702.2003.
. PMID 12588988. doi:10.1128/MCB.23.5.1688-1702.2003. - ↑ Zhou Y, Li L, Liu Q, Xing G, Kuai X, Sun J, Yin X, Wang J, Zhang L, He F (May 2008). "E3 ubiquitin ligase SIAH1 mediates ubiquitination and degradation of TRB3". Cell. Signal. 20 (5): 942–8. PMID 18276110. doi:10.1016/j.cellsig.2008.01.010.
Further reading
- Eriksen JL, Mackenzie IR (2008). "Progranulin: normal function and role in neurodegeneration.". J Neurochem. 104 (2): 287–297. PMID 17953663. doi:10.1111/J.1471-4159.2007.04968.X.
- Serrero G (2003). "Autocrine growth factor revisited: PC-cell-derived growth factor (progranulin), a critical player in breast cancer tumorigenesis.". Biochem. Biophys. Res. Commun. 308 (3): 409–13. PMID 12914763. doi:10.1016/S0006-291X(03)01452-9.
- Ahmed Z, Mackenzie IR, Hutton ML, Dickson DW (2007). "Progranulin in frontotemporal lobar degeneration and neuroinflammation.". Journal of Neuroinflammation. 4: 7. PMC 1805428
 . PMID 17291356. doi:10.1186/1742-2094-4-7.
. PMID 17291356. doi:10.1186/1742-2094-4-7. - Mackenzie IR (2007). "The neuropathology and clinical phenotype of FTD with progranulin mutations.". Acta Neuropathol. 114 (1): 49–54. PMID 17458552. doi:10.1007/s00401-007-0223-8.
- Pickering-Brown SM (2007). "Progranulin and frontotemporal lobar degeneration.". Acta Neuropathol. 114 (1): 39–47. PMID 17572900. doi:10.1007/s00401-007-0241-6.
- Bhandari V, Palfree RG, Bateman A (1992). "Isolation and sequence of the granulin precursor cDNA from human bone marrow reveals tandem cysteine-rich granulin domains.". Proc. Natl. Acad. Sci. U.S.A. 89 (5): 1715–9. Bibcode:1992PNAS...89.1715B. PMC 48523
 . PMID 1542665. doi:10.1073/pnas.89.5.1715.
. PMID 1542665. doi:10.1073/pnas.89.5.1715. - Plowman GD, Green JM, Neubauer MG, Buckley SD, McDonald VL, Todaro GJ, Shoyab M (1992). "The epithelin precursor encodes two proteins with opposing activities on epithelial cell growth.". J. Biol. Chem. 267 (18): 13073–8. PMID 1618805.
- Bateman A, Belcourt D, Bennett H, Lazure C, Solomon S (1991). "Granulins, a novel class of peptide from leukocytes.". Biochem. Biophys. Res. Commun. 173 (3): 1161–8. PMID 2268320. doi:10.1016/S0006-291X(05)80908-8.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides.". Gene. 138 (1–2): 171–4. PMID 8125298. doi:10.1016/0378-1119(94)90802-8.
- Baba T, Hoff HB, Nemoto H, Lee H, Orth J, Arai Y, Gerton GL (1993). "Acrogranin, an acrosomal cysteine-rich glycoprotein, is the precursor of the growth-modulating peptides, granulins, and epithelins, and is expressed in somatic as well as male germ cells.". Mol. Reprod. Dev. 34 (3): 233–43. PMID 8471244. doi:10.1002/mrd.1080340302.
- Kardana A, Bagshawe KD, Coles B, Read D, Taylor M (1993). "Characterisation of UGP and its relationship with beta-core fragment.". Br. J. Cancer. 67 (4): 686–92. PMC 1968365
 . PMID 8471426. doi:10.1038/bjc.1993.127.
. PMID 8471426. doi:10.1038/bjc.1993.127. - Zhou J, Gao G, Crabb JW, Serrero G (1993). "Purification of an autocrine growth factor homologous with mouse epithelin precursor from a highly tumorigenic cell line.". J. Biol. Chem. 268 (15): 10863–9. PMID 8496151.
- Andersson B, Wentland MA, Ricafrente JY, Liu W, Gibbs RA (1996). "A "double adaptor" method for improved shotgun library construction.". Anal. Biochem. 236 (1): 107–13. PMID 8619474. doi:10.1006/abio.1996.0138.
- Bhandari V, Daniel R, Lim PS, Bateman A (1996). "Structural and functional analysis of a promoter of the human granulin/epithelin gene.". Biochem. J. 319 (2): 441–7. PMC 1217788
 . PMID 8912679.
. PMID 8912679. - Yu W, Andersson B, Worley KC, Muzny DM, Ding Y, Liu W, Ricafrente JY, Wentland MA, Lennon G, Gibbs RA (1997). "Large-scale concatenation cDNA sequencing.". Genome Res. 7 (4): 353–8. PMC 139146
 . PMID 9110174. doi:10.1101/gr.7.4.353.
. PMID 9110174. doi:10.1101/gr.7.4.353. - Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library.". Gene. 200 (1–2): 149–56. PMID 9373149. doi:10.1016/S0378-1119(97)00411-3.
- Trinh DP, Brown KM, Jeang KT (1999). "Epithelin/granulin growth factors: extracellular cofactors for HIV-1 and HIV-2 Tat proteins.". Biochem. Biophys. Res. Commun. 256 (2): 299–306. PMID 10079180. doi:10.1006/bbrc.1999.0317.
- He Z, Bateman A (1999). "Progranulin gene expression regulates epithelial cell growth and promotes tumor growth in vivo.". Cancer Res. 59 (13): 3222–9. PMID 10397269.
- Thornton MA, Poncz M, Korostishevsky M, Yakobson E, Usher S, Seligsohn U, Peretz H (1999). "The human platelet alphaIIb gene is not closely linked to its integrin partner beta3.". Blood. 94 (6): 2039–47. PMID 10477733.