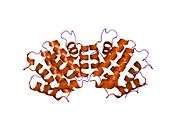
GGA1
ADP-ribosylation factor-binding protein GGA1 is a protein that in humans is encoded by the GGA1 gene.[3][4][5]
This gene encodes a member of the Golgi-localized, gamma adaptin ear-containing, ARF-binding (GGA) protein family. Members of this family are ubiquitous coat proteins that regulate the trafficking of proteins between the trans-Golgi network and the lysosome. These proteins share an amino-terminal VHS domain which mediates sorting of the mannose 6-phosphate receptors at the trans-Golgi network. They also contain a carboxy-terminal region with homology to the ear domain of gamma-adaptins. Multiple alternatively spliced transcript variants encoding different isoforms have been found for this gene.[6]
Interactions
GGA1 has been shown to interact with Sortilin 1,[7] BACE2,[8] RABEP1[9] and ARF3.[10][11]
References
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ Hirst J, Lui WW, Bright NA, Totty N, Seaman MN, Robinson MS (May 2000). "A family of proteins with gamma-adaptin and VHS domains that facilitate trafficking between the trans-Golgi network and the vacuole/lysosome". J Cell Biol. 149 (1): 67–80. PMC 2175106
 . PMID 10747088. doi:10.1083/jcb.149.1.67.
. PMID 10747088. doi:10.1083/jcb.149.1.67. - ↑ Dell'Angelica EC, Puertollano R, Mullins C, Aguilar RC, Vargas JD, Hartnell LM, Bonifacino JS (May 2000). "GGAs: a family of ADP ribosylation factor-binding proteins related to adaptors and associated with the Golgi complex". J Cell Biol. 149 (1): 81–94. PMC 2175099
 . PMID 10747089. doi:10.1083/jcb.149.1.81.
. PMID 10747089. doi:10.1083/jcb.149.1.81. - ↑ Xie L, Boyle D, Sanford D, Scherer PE, Pessin JE, Mora S (March 2006). "Intracellular trafficking and secretion of adiponectin is dependent on GGA-coated vesicles". J Biol Chem. 281 (11): 7253–9. PMID 16407204. doi:10.1074/jbc.M511313200.
- ↑ "Entrez Gene: GGA1 golgi associated, gamma adaptin ear containing, ARF binding protein 1".
- ↑ Jacobsen, Linda; Madsen Peder; Nielsen Morten S; Geraerts Wijnand P M; Gliemann Jørgen; Smit August B; Petersen Claus M (January 2002). "The sorLA cytoplasmic domain interacts with GGA1 and -2 and defines minimum requirements for GGA binding". FEBS Lett. Netherlands. 511 (1-3): 155–8. ISSN 0014-5793. PMID 11821067. doi:10.1016/S0014-5793(01)03299-9.
- ↑ He, Xiangyuan; Chang Wan-Pin; Koelsch Gerald; Tang Jordan (July 2002). "Memapsin 2 (beta-secretase) cytosolic domain binds to the VHS domains of GGA1 and GGA2: implications on the endocytosis mechanism of memapsin 2". FEBS Lett. Netherlands. 524 (1-3): 183–7. ISSN 0014-5793. PMID 12135764. doi:10.1016/S0014-5793(02)03052-1.
- ↑ Mattera, Rafael; Arighi Cecilia N; Lodge Robert; Zerial Marino; Bonifacino Juan S (January 2003). "Divalent interaction of the GGAs with the Rabaptin-5-Rabex-5 complex". EMBO J. England. 22 (1): 78–88. ISSN 0261-4189. PMC 140067
 . PMID 12505986. doi:10.1093/emboj/cdg015.
. PMID 12505986. doi:10.1093/emboj/cdg015. - ↑ Boman, Annette L; Salo Paul D; Hauglund Melissa J; Strand Nicole L; Rensink Shelly J; Zhdankina Olga (September 2002). "ADP-ribosylation factor (ARF) interaction is not sufficient for yeast GGA protein function or localization". Mol. Biol. Cell. United States. 13 (9): 3078–95. ISSN 1059-1524. PMC 124144
 . PMID 12221117. doi:10.1091/mbc.E02-02-0078.
. PMID 12221117. doi:10.1091/mbc.E02-02-0078. - ↑ Boman, A L; Zhang C j; Zhu X; Kahn R A (April 2000). "A family of ADP-ribosylation factor effectors that can alter membrane transport through the trans-Golgi". Mol. Biol. Cell. UNITED STATES. 11 (4): 1241–55. ISSN 1059-1524. PMC 14844
 . PMID 10749927. doi:10.1091/mbc.11.4.1241.
. PMID 10749927. doi:10.1091/mbc.11.4.1241.
Further reading
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides.". Gene. 138 (1-2): 171–4. PMID 8125298. doi:10.1016/0378-1119(94)90802-8.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, et al. (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library.". Gene. 200 (1-2): 149–56. PMID 9373149. doi:10.1016/S0378-1119(97)00411-3.
- Dunham I, Shimizu N, Roe BA, et al. (1999). "The DNA sequence of human chromosome 22.". Nature. 402 (6761): 489–95. PMID 10591208. doi:10.1038/990031.
- Boman AL, Zhang C, Zhu X, Kahn RA (2000). "A family of ADP-ribosylation factor effectors that can alter membrane transport through the trans-Golgi.". Mol. Biol. Cell. 11 (4): 1241–55. PMC 14844
 . PMID 10749927. doi:10.1091/mbc.11.4.1241.
. PMID 10749927. doi:10.1091/mbc.11.4.1241. - Takatsu H, Yoshino K, Nakayama K (2000). "Adaptor gamma ear homology domain conserved in gamma-adaptin and GGA proteins that interact with gamma-synergin.". Biochem. Biophys. Res. Commun. 271 (3): 719–25. PMID 10814529. doi:10.1006/bbrc.2000.2700.
- Zhdankina O, Strand NL, Redmond JM, Boman AL (2001). "Yeast GGA proteins interact with GTP-bound Arf and facilitate transport through the Golgi.". Yeast. 18 (1): 1–18. PMID 11124697. doi:10.1002/1097-0061(200101)18:1<1::AID-YEA644>3.0.CO;2-5.
- Puertollano R, Randazzo PA, Presley JF, et al. (2001). "The GGAs promote ARF-dependent recruitment of clathrin to the TGN.". Cell. 105 (1): 93–102. PMID 11301005. doi:10.1016/S0092-8674(01)00299-9.
- Puertollano R, Aguilar RC, Gorshkova I, et al. (2001). "Sorting of mannose 6-phosphate receptors mediated by the GGAs.". Science. 292 (5522): 1712–6. PMID 11387475. doi:10.1126/science.1060750.
- Takatsu H, Katoh Y, Shiba Y, Nakayama K (2001). "Golgi-localizing, gamma-adaptin ear homology domain, ADP-ribosylation factor-binding (GGA) proteins interact with acidic dileucine sequences within the cytoplasmic domains of sorting receptors through their Vps27p/Hrs/STAM (VHS) domains.". J. Biol. Chem. 276 (30): 28541–5. PMID 11390366. doi:10.1074/jbc.C100218200.
- Jacobsen L, Madsen P, Nielsen MS, et al. (2002). "The sorLA cytoplasmic domain interacts with GGA1 and -2 and defines minimum requirements for GGA binding.". FEBS Lett. 511 (1-3): 155–8. PMID 11821067. doi:10.1016/S0014-5793(01)03299-9.
- Shiba T, Takatsu H, Nogi T, et al. (2002). "Structural basis for recognition of acidic-cluster dileucine sequence by GGA1.". Nature. 415 (6874): 937–41. PMID 11859376. doi:10.1038/415937a.
- Takatsu H, Yoshino K, Toda K, Nakayama K (2002). "GGA proteins associate with Golgi membranes through interaction between their GGAH domains and ADP-ribosylation factors.". Biochem. J. 365 (Pt 2): 369–78. PMC 1222692
 . PMID 11950392. doi:10.1042/BJ20020428.
. PMID 11950392. doi:10.1042/BJ20020428. - Doray B, Bruns K, Ghosh P, Kornfeld SA (2002). "Autoinhibition of the ligand-binding site of GGA1/3 VHS domains by an internal acidic cluster-dileucine motif.". Proc. Natl. Acad. Sci. U.S.A. 99 (12): 8072–7. PMC 123022
 . PMID 12060753. doi:10.1073/pnas.082235699.
. PMID 12060753. doi:10.1073/pnas.082235699. - He X, Chang WP, Koelsch G, Tang J (2002). "Memapsin 2 (beta-secretase) cytosolic domain binds to the VHS domains of GGA1 and GGA2: implications on the endocytosis mechanism of memapsin 2.". FEBS Lett. 524 (1-3): 183–7. PMID 12135764. doi:10.1016/S0014-5793(02)03052-1.
- Boman AL, Salo PD, Hauglund MJ, et al. (2003). "ADP-ribosylation factor (ARF) interaction is not sufficient for yeast GGA protein function or localization.". Mol. Biol. Cell. 13 (9): 3078–95. PMC 124144
 . PMID 12221117. doi:10.1091/mbc.E02-02-0078.
. PMID 12221117. doi:10.1091/mbc.E02-02-0078. - Strausberg RL, Feingold EA, Grouse LH, et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences.". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. PMC 139241
 . PMID 12477932. doi:10.1073/pnas.242603899.
. PMID 12477932. doi:10.1073/pnas.242603899. - Mattera R, Arighi CN, Lodge R, et al. (2003). "Divalent interaction of the GGAs with the Rabaptin-5-Rabex-5 complex.". EMBO J. 22 (1): 78–88. PMC 140067
 . PMID 12505986. doi:10.1093/emboj/cdg015.
. PMID 12505986. doi:10.1093/emboj/cdg015. - Mills IG, Praefcke GJ, Vallis Y, et al. (2003). "EpsinR: an AP1/clathrin interacting protein involved in vesicle trafficking.". J. Cell Biol. 160 (2): 213–22. PMC 2172650
 . PMID 12538641. doi:10.1083/jcb.200208023.
. PMID 12538641. doi:10.1083/jcb.200208023.