Ferrocenium tetrafluoroborate
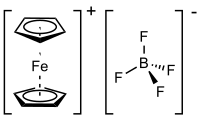 | |
| Names | |
|---|---|
| Other names
dicyclopentadienyl iron tetrafluoroborate | |
| Identifiers | |
| ECHA InfoCard | 100.156.161 |
| Properties | |
| C10H10BFeF4 | |
| Molar mass | 272.84 g/mol |
| Appearance | dark blue powder |
| Melting point | 178 °C (352 °F; 451 K) (decomposes) |
| Solubility in acetonitrile | Soluble |
| Hazards | |
| Safety data sheet | External MSDS |
| EU classification (DSD) (outdated) |
|
| Related compounds | |
| Related compounds |
Ferrocene |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Ferrocenium tetrafluoroborate is an organometallic compound with the formula [Fe(C5H5)2]BF4. This salt is composed of the cation [Fe(C5H5)2]+ and the tetrafluoroborate anion (BF−
4). The related hexafluorophosphate is also a popular reagent with similar properties. The cation is often abbreviated Fc+ or Cp2Fe+. The salt is deep blue in color and paramagnetic.
Ferrocenium salts are sometimes used as one-electron oxidizing agents, and the reduced product, ferrocene, is inert and readily separated from ionic products. The ferrocene–ferrocenium couple is often used as a reference in electrochemistry. The standard potential of ferrocene-ferrocenium is 0.400 V vs. the normal hydrogen electrode (NHE)[1] and is often assumed to be invariant between different solvents.
Preparation
Commercially available, this compound may be prepared by oxidizing ferrocene typically with ferric salts followed by addition of fluoroboric acid.[2] A variety of other oxidants work well also, such as nitrosyl tetrafluoroborate.[3] Many analogous ferrocenium salts are known.[4]
References
- ↑ Bard, A.J.; Parsons, R.; Jordan, J. (1985). Standard Potentials in Aqueous Solution. New York: IUPAC.
- ↑ Connelly, N. G.; Geiger, W. E. (1996). "Chemical Redox Agents for Organometallic Chemistry". Chem. Rev. 96 (2): 877–910. PMID 11848774. doi:10.1021/cr940053x.
- ↑ Nielson, Roger M.; McManis, George E.; Safford, Lance K.; Weaver, Michael J. (1989). "Solvent and electrolyte effects on the kinetics of ferrocenium-ferrocene self-exchange. A reevaluation". J. Phys. Chem. 93 (5): 2152. doi:10.1021/j100342a086.
- ↑ Le Bras, J.; Jiao, H.; Meyer, W. E.; Hampel, F.; Gladysz, J. A. (2000). "Synthesis, Crystal Structure, and Reactions of the 17-Valence-Electron Rhenium Methyl Complex [(η5-C5Me5)Re(NO)(P(4-C6H4CH3)3)(CH3)]+ B(3,5-C
6H
3(CF
3)
2)−
4: Experimental and Computational Bonding Comparisons with 18-Electron Methyl and Methylidene Complexes". J. Organomet. Chem. 616: 54–66. doi:10.1016/S0022-328X(00)00531-3.