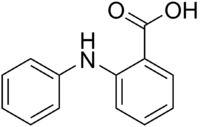
Fenamic acid
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-(phenylamino)benzoic acid | |
| Other names
N-phenylanthranilic acid | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.001.879 |
| PubChem CID |
|
| |
| |
| Properties | |
| C13H11NO2 | |
| Molar mass | 213.23 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Fenamic acid is a molecule which, especially in its ester form, fenamate,[1]:458 serves as a parent structure for several nonsteroidal anti-inflammatory drugs (NSAIDs), including mefenamic acid, tolfenamic acid, flufenamic acid, and meclofenamic acid. These drugs are commonly referred to as "anthranilic acid derivatives" or "fenamates" because fenamic acid is a derivative of anthranilic acid.[2]:235[3]:17[2]
References
- ↑ Gupta, PK. Drug NomenclatureUnited States Adopted Names. Ch 27 in Remington: The Science and Practice of Pharmacy, Vol 1. Eds. David B. Troy, Paul Beringer. Lippincott Williams & Wilkins, 2006 ISBN 9780781746731
- 1 2 Sriram D, Yogeeswari P. Medicinal Chemistry, 2nd Edition. Pearson Education India, 2010. ISBN 9788131731444
- ↑ Auburn University course material. Jack DeRuiter, Principles of Drug Action 2, Fall 2002 1: Non-Steroidal Antiinflammatory Drugs (NSAIDS)
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.