Thalamus
| Thalamus | |
|---|---|
 thalamus marked (MRI cross-section) | |
 anterolateral view | |
| Details | |
| Part of | Diencephalon |
| Components | See List of thalamic nuclei |
| Artery | Posterior cerebral artery and branches |
| Identifiers | |
| Latin | thalamus dorsalis |
| MeSH | A08.186.211.730.385.826 |
| NeuroNames | hier-283 |
| NeuroLex ID | Thalamus |
| TA |
A14.1.08.101 A14.1.08.601 |
| FMA | 62007 |
The thalamus (from Greek θάλαμος, "chamber")[1] is the large mass of gray matter in the dorsal part of the diencephalon of the brain with several functions such as relaying of sensory and motor signals to the cerebral cortex,[2][3] and the regulation of consciousness, sleep, and alertness.
It is a midline symmetrical structure of two halves, within the vertebrate brain, situated between the cerebral cortex and the midbrain. The medial surface of the two halves constitute the upper lateral wall of the third ventricle.
It is the main product of the embryonic diencephalon, as first assigned by Wilhelm His, Sr. in 1893.[4]
Anatomy

The thalamus is located in the hindbrain which is superior to the midbrain, near the center of the brain, with nerve fibers projecting out to the cerebral cortex in all directions. The medial surface of the thalamus constitutes the upper part of the lateral wall of the third ventricle, and is connected to the corresponding surface of the opposite thalamus by a flattened gray band, the interthalamic adhesion.
Blood supply
The thalamus derives its blood supply from a number of arteries: the polar artery (posterior communicating artery), paramedian thalamic-subthalamic arteries, inferolateral (thalamogeniculate) arteries, and posterior (medial and lateral) choroidal arteries.[5] These are all branches of the posterior cerebral artery.[6]
Some people have the artery of Percheron, which is a rare anatomic variation in which a single arterial trunk arises from the posterior cerebral artery to supply both parts of the thalamus.
Thalamic nuclei

The thalamus is part of a nuclear complex structured of four parts, the hypothalamus, epithalamus, prethalamus (formerly called ventral thalamus), and dorsal thalamus.[7]
Derivatives of the diencephalon also include the dorsally-located epithalamus (essentially the habenula and annexes) and the perithalamus (prethalamus) containing the zona incerta and the thalamic reticular nucleus. Due to their different ontogenetic origins, the epithalamus and the perithalamus are formally distinguished from the thalamus proper.
The thalamus comprises a system of lamellae (made up of myelinated fibers) separating different thalamic subparts. Other areas are defined by distinct clusters of neurons, such as the periventricular nucleus, the intralaminar elements, the "nucleus limitans", and others.[8] These latter structures, different in structure from the major part of the thalamus, have been grouped together into the allothalamus as opposed to the isothalamus.[9] This distinction simplifies the global description of the thalamus.
Connections

The thalamus is manifoldly connected to the hippocampus via the mammillo-thalamic tract, this tract comprises the mammillary bodies and fornix.[10]
The thalamus is connected to the cerebral cortex via the thalamocortical radiations.[11]
The spinothalamic tract is a sensory pathway originating in the spinal cord. It transmits information to the thalamus about pain, temperature, itch and crude touch. There are two main parts: the lateral spinothalamic tract, which transmits pain and temperature, and the anterior (or ventral) spinothalamic tract, which transmits crude touch and pressure.
Function
The thalamus has multiple functions. It may be thought of as a kind of hub of information. It is generally believed to act as a relay between different subcortical areas and the cerebral cortex.[12] In particular, every sensory system (with the exception of the olfactory system) includes a thalamic nucleus that receives sensory signals and sends them to the associated primary cortical area. For the visual system, for example, inputs from the retina are sent to the lateral geniculate nucleus of the thalamus, which in turn projects to the visual cortex in the occipital lobe. The thalamus is believed to both process sensory information as well as relay it—each of the primary sensory relay areas receives strong feedback connections from the cerebral cortex. Similarly the medial geniculate nucleus acts as a key auditory relay between the inferior colliculus of the midbrain and the primary auditory cortex. The ventral posterior nucleus is a key somatosensory relay, which sends touch and proprioceptive information to the primary somatosensory cortex.
The thalamus also plays an important role in regulating states of sleep and wakefulness.[13] Thalamic nuclei have strong reciprocal connections with the cerebral cortex, forming thalamo-cortico-thalamic circuits that are believed to be involved with consciousness. The thalamus plays a major role in regulating arousal, the level of awareness, and activity. Damage to the thalamus can lead to permanent coma.
The role of the thalamus in the more anterior pallidal and nigral territories in the basal ganglia system disturbances is recognized but still poorly understood. The contribution of the thalamus to vestibular or to tectal functions is almost ignored. The thalamus has been thought of as a "relay" that simply forwards signals to the cerebral cortex. Newer research suggests that thalamic function is more selective.[14] Many different functions are linked to various regions of the thalamus. This is the case for many of the sensory systems (except for the olfactory system), such as the auditory, somatic, visceral, gustatory and visual systems where localized lesions provoke specific sensory deficits. A major role of the thalamus is support of motor and language systems, and much of the circuitry implicated for these systems is shared. The thalamus is functionally connected to the hippocampus[15] as part of the extended hippocampal system at the thalamic anterior nuclei[16] with respect to spatial memory and spatial sensory datum they are crucial for human episodic memory and rodent event memory.[17][18] There is support for the hypothesis that thalamic regions connection to particular parts of the mesio-temporal lobe provide differentiation of the functioning of recollective and familiarity memory.[10]
The neuronal information processes necessary for motor control were proposed as a network involving the thalamus as a subcortical motor center.[19] Through investigations of the anatomy of the brains of primates[20] the nature of the interconnected tissues of the cerebellum to the multiple motor cortices suggested that the thalamus fulfills a key function in providing the specific channels from the basal ganglia and cerebellum to the cortical motor areas.[21][22] In an investigation of the saccade and antisaccade[23] motor response in three monkeys, the thalamic regions were found to be involved in the generation of antisaccade eye-movement.[24]
Development
The thalamic complex is composed of the perithalamus (or prethalamus, previously also known as ventral thalamus), the mid-diencephalic organiser (which forms later the zona limitans intrathalamica (ZLI) ) and the thalamus (dorsal thalamus).[25][26] The development of the thalamus can be subdivided into three steps.[27] The thalamus is the largest structure deriving from the embryonic diencephalon, the posterior part of the forebrain situated between the midbrain and the cerebrum.
Early brain development
After neurulation the anlage of the prethalamus and the thalamus is induced within the neural tube. Data from different vertebrate model organisms support a model in which the interaction between two transcription factors, Fez and Otx, are of decisive importance. Fez is expressed in the prethalamus, and functional experiments show that Fez is required for prethalamus formation.[28][29] Posteriorly, Otx1 and Otx2 abut the expression domain of Fez and are required for proper development of the thalamus.[30][31]
Formation of progenitor domains
Early in thalamic development two progenitor domains form, a caudal domain (TH-C) and a rostral domain (TH-R). The caudal domain gives rise to all of the glutamatergic neurons in the adult thalamus while the rostral domain gives rise to all of the GABAergic neurons in the adult thalamus.[32]
The formation of the mid-diencephalic organiser (MDO)
At the interface between the expression domains of Fez and Otx, the mid-diencephalic organizer (MDO, also called the ZLI organiser) is induced within the thalamic anlage. The MDO is the central signalling organizer in the thalamus. A lack of the organizer leads to the absence of the thalamus. The MDO matures from ventral to dorsal during development. Members of the SHH family and of the Wnt family are the main principal signals emitted by the MDO.
Besides its importance as signalling center, the organizer matures into the morphological structure of the zona limitans intrathalamica (ZLI).
Maturation and parcellation of the thalamus
After its induction, the MDO starts to orchestrate the development of the thalamic anlage by release of signalling molecules such as SHH.[33] In mice, the function of signaling at the MDO has not been addressed directly due to a complete absence of the diencephalon in SHH mutants.[34]
Studies in chicks have shown that SHH is both necessary and sufficient for thalamic gene induction.[35] In zebrafish, it was shown that the expression of two SHH genes, SHH-a and SHH-b (formerly described as twhh) mark the MDO territory, and that SHH signaling is sufficient for the molecular differentiation of both the prethalamus and the thalamus but is not required for their maintenance and SHH signaling from the MDO/alar plate is sufficient for the maturation of prethalamic and thalamic territory while ventral Shh signals are dispensable.[36]
The exposure to SHH leads to differentiation of thalamic neurons. SHH signaling from the MDO induces a posterior-to-anterior wave of expression the proneural gene Neurogenin1 in the major (caudal) part of the thalamus, and Ascl1 (formerly Mash1) in the remaining narrow stripe of rostral thalamic cells immediately adjacent to the MDO, and in the prethalamus.[37][38]
This zonation of proneural gene expression leads to the differentiation of glutamatergic relay neurons from the Neurogenin1+ precursors and of GABAergic inhibitory neurons from the Ascl1+ precursors. In fish, selection of these alternative neurotransmitter fates is controlled by the dynamic expression of Her6 the homolog of HES1. Expression of this hairy-like bHLH transcription factor, which represses Neurogenin but is required for Ascl1, is progressively lost from the caudal thalamus but maintained in the prethalamus and in the stripe of rostral thalamic cells. In addition, studies on chick and mice have shown that blocking the Shh pathway leads to absence of the rostral thalamus and substantial decrease of the caudal thalamus. The rostral thalamus will give rise to the reticular nucleus mainly whereby the caudal thalamus will form the relay thalamus and will be further subdivided in the thalamic nuclei.[27]
In humans, a common genetic variation in the promotor region of the serotonin transporter (the SERT-long and -short allele: 5-HTTLPR) has been shown to affect the development of several regions of the thalamus in adults. People who inherit two short alleles (SERT-ss) have more neurons and a larger volume in the pulvinar and possibly the limbic regions of the thalamus. Enlargement of the thalamus provides an anatomical basis for why people who inherit two SERT-ss alleles are more vulnerable to major depression, posttraumatic stress disorder, and suicide.[39]
Clinical significance
A cerebrovascular accident (stroke) can lead to the thalamic syndrome,[40] which involves a one-sided burning or aching sensation often accompanied by mood swings. Bilateral ischemia of the area supplied by the paramedian artery can cause serious problems including akinetic mutism, and be accompanied by oculomotor problems. A related concept is thalamocortical dysrhythmia. The occlusion of the artery of Percheron can lead to a bilateral thalamus infarction.
Korsakoff's syndrome stems from damage to the mammillary body, the mammillothalamic fasciculus or the thalamus.
Fatal familial insomnia is a hereditary prion disease in which degeneration of the thalamus occurs, causing the patient to gradually lose his ability to sleep and progressing to a state of total insomnia, which invariably leads to death. In contrast, damage to the thalamus can result in coma.
Additional images
Drawings are by Gray and Carter (1858).[41]
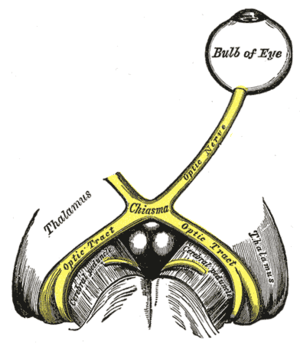 The left optic nerve and the optic tracts.
The left optic nerve and the optic tracts. Coronal section of lateral and 3rd ventricles.
Coronal section of lateral and 3rd ventricles. Dissection showing the ventricles of the brain.
Dissection showing the ventricles of the brain. Section of brain showing upper surface of temporal lobe.
Section of brain showing upper surface of temporal lobe. Horizontal section of right cerebral hemisphere.
Horizontal section of right cerebral hemisphere. Mesal aspect of a brain sectioned in the median sagittal plane.
Mesal aspect of a brain sectioned in the median sagittal plane. Schematic representation of the chief ganglionic categories (I to V).
Schematic representation of the chief ganglionic categories (I to V).
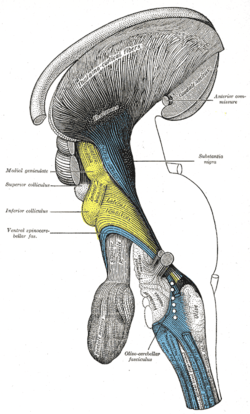 Deep dissection of brain-stem. Lateral view.
Deep dissection of brain-stem. Lateral view. Deep dissection of brain-stem. Ventral view.
Deep dissection of brain-stem. Ventral view. Coronal section of brain immediately in front of pons.
Coronal section of brain immediately in front of pons. Coronal section of brain through intermediate mass of third ventricle.
Coronal section of brain through intermediate mass of third ventricle.- Human brain frontal (coronal) section
- Thalamus
- Ventricles of brain and basal ganglia. Superior view. Horizontal section. Deep dissection
- Ventricles of brain and basal ganglia. Superior view. Horizontal section. Deep dissection
See also
- List of regions in the human brain
- List of thalamic nuclei
- Neothalamus
- Primate basal ganglia system
- Thalamic stimulator
- Thalamotomy
- 5-HT7 receptor
- Nonmotor region of the ventral nuclear group of the thalamus
References
- ↑ Harper - index & University of Washington Faculty Web Server & Search engine search page + Perseus Project tufts.edu Retrieved 2012-02-09
- ↑ Sherman, S. (2006). "Thalamus". Scholarpedia. 1 (9): 1583. doi:10.4249/scholarpedia.1583.
- ↑ Sherman, S. Murray; Guillery, R. W. (2000). Exploring the Thalamus. Academic Press. ISBN 978-0-12-305460-9.
- ↑ The Thalamus - Springer. doi:10.1007/978-1-4615-1749-8.
- ↑ Percheron, G. (1982). "The arterial supply of the thalamus". In Schaltenbrand; Walker, A. E. Stereotaxy of the human brain. Stuttgart: Thieme. pp. 218–32.
- ↑ Knipe, H Jones, J et al. Thalamus http://radiopaedia.org/articles/thalamus
- ↑ Herrero, María-Trinidad; Barcia, Carlos; Navarro, Juana (2002). "Functional anatomy of thalamus and basal ganglia". Child's Nervous System. 18 (8): 386–404. doi:10.1007/s00381-002-0604-1.
- ↑ Jones Edward G. (2007) "The Thalamus" Cambridge Uni. Press
- ↑ Percheron, G. (2003). "Thalamus". In Paxinos, G.; May, J. The human nervous system (2nd ed.). Amsterdam: Elsevier. pp. 592–675.
- 1 2 Carlesimo, GA; Lombardi, MG; Caltagirone, C (2011). "Vascular thalamic amnesia: A reappraisal". Neuropsychologia. 49 (5): 777–89. PMID 21255590. doi:10.1016/j.neuropsychologia.2011.01.026.
- ↑ University of Washington (1991). "Thalamocortical radiations". washington.edu.
- ↑ Gazzaniga; Ivry; Mangun, Michael, S.; Richard B.; George R. (2014). Cognitive Neuroscience - The Biology of The Mind. New York: W.W. Norton. p. 45. ISBN 978-0-393-91348-4.
- ↑ Steriade, Mircea; Llinás, Rodolfo R. (1988). "The Functional States of the Thalamus and the Associated Neuronal Interplay". Physiological Reviews. 68 (3): 649–742. PMID 2839857.
- ↑ Leonard, Abigail W. (August 17, 2006). "Your Brain Boots Up Like a Computer". LiveScience.
- ↑ Stein, Thor; Moritz, Chad; Quigley, Michelle; Cordes, Dietmar; Haughton, Victor; Meyerand, Elizabeth (2000). "Functional Connectivity in the Thalamus and Hippocampus Studied with Functional MR Imaging". American Journal of Neuroradiology. 21 (8): 1397–401. PMID 11003270.
- ↑ Aggleton, John P.; Brown, Malcolm W. (1999). "Episodic memory, amnesia, and the hippocampal–anterior thalamic axis". Behavioral and Brain Sciences. 22 (3): 425–44; discussion 444–89. PMID 11301518. doi:10.1017/S0140525X99002034.
- ↑ Aggleton, John P.; O'Mara, Shane M.; Vann, Seralynne D.; Wright, Nick F.; Tsanov, Marian; Erichsen, Jonathan T. (2010). "Hippocampal-anterior thalamic pathways for memory: Uncovering a network of direct and indirect actions". European Journal of Neuroscience. 31 (12): 2292–307. PMC 2936113
 . PMID 20550571. doi:10.1111/j.1460-9568.2010.07251.x.
. PMID 20550571. doi:10.1111/j.1460-9568.2010.07251.x. - ↑ Burgess, Neil; Maguire, Eleanor A; O'Keefe, John (2002). "The Human Hippocampus and Spatial and Episodic Memory". Neuron. 35 (4): 625–41. PMID 12194864. doi:10.1016/S0896-6273(02)00830-9.
- ↑ Evarts, E V; Thach, W T (1969). "Motor Mechanisms of the CNS: Cerebrocerebellar Interrelations". Annual Review of Physiology. 31: 451–98. PMID 4885774. doi:10.1146/annurev.ph.31.030169.002315.
- ↑ Orioli, PJ; Strick, PL (1989). "Cerebellar connections with the motor cortex and the arcuate premotor area: An analysis employing retrograde transneuronal transport of WGA-HRP". The Journal of Comparative Neurology. 288 (4): 612–26. PMID 2478593. doi:10.1002/cne.902880408.
- ↑ Asanuma C, Thach WT, Jones EG (May 1983). "Cytoarchitectonic delineation of the ventral lateral thalamic region in the monkey". Brain Research. 286 (3): 219–35. PMID 6850357. doi:10.1016/0165-0173(83)90014-0.
- ↑ Kurata, K (2005). "Activity properties and location of neurons in the motor thalamus that project to the cortical motor areas in monkeys". Journal of Neurophysiology. 94 (1): 550–66. PMID 15703228. doi:10.1152/jn.01034.2004.
- ↑ http://www.optomotorik.de/blicken/anti-rev.htm%5B%5D
- ↑ Kunimatsu, J; Tanaka, M (2010). "Roles of the primate motor thalamus in the generation of antisaccades". Journal of Neuroscience. 30 (14): 5108–17. PMID 20371831. doi:10.1523/JNEUROSCI.0406-10.2010.
- ↑ Kuhlenbeck, Hartwig (1937). "The ontogenetic development of the diencephalic centers in a bird's brain (chick) and comparison with the reptilian and mammalian diencephalon". The Journal of Comparative Neurology. 66: 23–75. doi:10.1002/cne.900660103.
- ↑ Shimamura, K; Hartigan, DJ; Martinez, S; Puelles, L; Rubenstein, JL (1995). "Longitudinal organization of the anterior neural plate and neural tube". Development. 121 (12): 3923–33. PMID 8575293.
- 1 2 Scholpp, Steffen; Lumsden, Andrew (2010). "Building a bridal chamber: Development of the thalamus". Trends in Neurosciences. 33 (8): 373–80. PMC 2954313
 . PMID 20541814. doi:10.1016/j.tins.2010.05.003.
. PMID 20541814. doi:10.1016/j.tins.2010.05.003. - ↑ Hirata, T.; Nakazawa, M; Muraoka, O; Nakayama, R; Suda, Y; Hibi, M (2006). "Zinc-finger genes Fez and Fez-like function in the establishment of diencephalon subdivisions". Development. 133 (20): 3993–4004. PMID 16971467. doi:10.1242/dev.02585.
- ↑ Jeong, J.-Y.; Einhorn, Z.; Mathur, P.; Chen, L.; Lee, S.; Kawakami, K.; Guo, S. (2007). "Patterning the zebrafish diencephalon by the conserved zinc-finger protein Fezl". Development. 134 (1): 127–36. PMID 17164418. doi:10.1242/dev.02705.
- ↑ Acampora, D; Avantaggiato, V; Tuorto, F; Simeone, A (1997). "Genetic control of brain morphogenesis through Otx gene dosage requirement". Development. 124 (18): 3639–50. PMID 9342056.
- ↑ Scholpp, S.; Foucher, I.; Staudt, N.; Peukert, D.; Lumsden, A.; Houart, C. (2007). "Otx1l, Otx2 and Irx1b establish and position the ZLI in the diencephalon". Development. 134 (17): 3167–76. PMID 17670791. doi:10.1242/dev.001461.
- ↑ Song, Hobeom; Lee, Bumwhee; Pyun, Dohoon; Guimera, Jordi; Son, Youngsook; Yoon, Jaeseung; Baek, Kwanghee; Wurst, Wolfgang; Jeong, Yongsu (2015-02-15). "Ascl1 and Helt act combinatorially to specify thalamic neuronal identity by repressing Dlxs activation". Developmental Biology. 398 (2): 280–291. ISSN 1095-564X. PMID 25512300. doi:10.1016/j.ydbio.2014.12.003.
- ↑ Puelles, L; Rubenstein, JL (2003). "Forebrain gene expression domains and the evolving prosomeric model". Trends in Neurosciences. 26 (9): 469–76. PMID 12948657. doi:10.1016/S0166-2236(03)00234-0.
- ↑ Ishibashi, M; McMahon, AP (2002). "A sonic hedgehog-dependent signaling relay regulates growth of diencephalic and mesencephalic primordia in the early mouse embryo". Development. 129 (20): 4807–19. PMID 12361972.
- ↑ Kiecker, C; Lumsden, A (2004). "Hedgehog signaling from the ZLI regulates diencephalic regional identity". Nature Neuroscience. 7 (11): 1242–9. PMID 15494730. doi:10.1038/nn1338.
- ↑ Scholpp, S.; Wolf, O; Brand, M; Lumsden, A (2006). "Hedgehog signalling from the zona limitans intrathalamica orchestrates patterning of the zebrafish diencephalon". Development. 133 (5): 855–64. PMID 16452095. doi:10.1242/dev.02248.
- ↑ Scholpp, S.; Delogu, A.; Gilthorpe, J.; Peukert, D.; Schindler, S.; Lumsden, A. (2009). "Her6 regulates the neurogenetic gradient and neuronal identity in the thalamus". Proceedings of the National Academy of Sciences. 106 (47): 19895–900. PMC 2775703
 . PMID 19903880. doi:10.1073/pnas.0910894106.
. PMID 19903880. doi:10.1073/pnas.0910894106. - ↑ Vue, Tou Yia; Bluske, Krista; Alishahi, Amin; Yang, Lin Lin; Koyano-Nakagawa, Naoko; Novitch, Bennett; Nakagawa, Yasushi (2009). "Sonic Hedgehog Signaling Controls Thalamic Progenitor Identity and Nuclei Specification in Mice". Journal of Neuroscience. 29 (14): 4484–97. PMC 2718849
 . PMID 19357274. doi:10.1523/JNEUROSCI.0656-09.2009.
. PMID 19357274. doi:10.1523/JNEUROSCI.0656-09.2009. - ↑ Young, Keith A.; Holcomb, Leigh A.; Bonkale, Willy L.; Hicks, Paul B.; Yazdani, Umar; German, Dwight C. (2007). "5HTTLPR Polymorphism and Enlargement of the Pulvinar: Unlocking the Backdoor to the Limbic System". Biological Psychiatry. 61 (6): 813–8. PMID 17083920. doi:10.1016/j.biopsych.2006.08.047.
- ↑ Dejerine, J.; Roussy, G. (1906). "Le syndrome thalamique". Revue Neurologique. 14: 521–32.
- ↑ Gray, H. & Carter, H. V. (1858), Anatomy Descriptive and Surgical, London: John W. Parker and Son, Retrieved (16 October 2011) [2012-02-10] →
External links
| Wikimedia Commons has media related to Thalamus. |