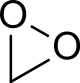
Dioxirane
| |||
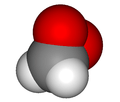 | |||
| Names | |||
|---|---|---|---|
| IUPAC name
Dioxirane | |||
| Identifiers | |||
| 3D model (JSmol) |
|||
| ChemSpider | |||
| PubChem CID |
|||
| |||
| |||
| Properties | |||
| CH2O2 | |||
| Molar mass | 46.03 g/mol | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
In chemistry dioxirane is a heterocyclic compound composed of one carbon and two oxygen atoms; it may be thought of as the smallest cyclic organic peroxide. The compound is highly unstable and has never been observed at room temperature. Compounds containing dioxirane as a functional group, called dioxiranes, often possess better stability and are used in organic synthesis as oxidizing reagents,[1] most notably as the key catalytic intermediate in the Shi epoxidation reaction. Other common derivatives employed in organic synthesis include dimethyldioxirane (DMDO) and the more reactive methyl(trifluoromethyl)dioxirane, which are prepared as dilute solutions (~0.1 M) by treatment of acetone and methyl trifluoromethyl ketone, respectively, with Oxone (2KHSO5·KHSO4·K2SO4). Difluorodioxirane, a gas (b.p ~ –80 - –90 °C), is one of the very few dioxirane derivatives that is stable in pure form at room temperature.
Synthesis
Dioxirane is highly unstable and the majority of studies of it have been computational; it has been detected during the low temperature (-196 °C) reaction of ethylene and ozone,[2] although even at these temperatures such a mixture can be explosive.[3] Its formation is thought to be radical in nature, preceding via a Criegee intermediate. Microwave analysis has indicated C-H, C-O and O-O bond lengths of 1.090, 1.388 and 1.516 Å respectively.[3] The very long and weak O-O bond (c.f. hydrogen peroxide O-O = 1.47 Å) is the origin of its instability.
See also
References
- ↑ Ruggero Curci; Anna Dinoi; Maria F. Rubino (1995). "Dioxirane oxidations: Taming the reactivity-selectivity principle" (PDF). Pure & Appl. Chem. 67 (5): 811–822. doi:10.1351/pac199567050811.
- ↑ Lovas, F.J.; Suenram, R.D. (November 1977). "Identification of dioxirane (H2) in ozone-olefin reactions via microwave spectroscopy". Chemical Physics Letters. 51 (3): 453–456. doi:10.1016/0009-2614(77)85398-0.
- 1 2 Suenram, R. D.; Lovas, F. J. (August 1978). "Dioxirane. Its synthesis, microwave spectrum, structure, and dipole moment". Journal of the American Chemical Society. 100 (16): 5117–5122. doi:10.1021/ja00484a034.