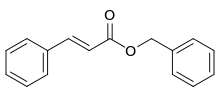
Benzyl cinnamate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Benzyl 3-phenylprop-2-enoate | |
| Other names
Benzylcinnamate; Cinnamein; Benzylcinnamoate; Benzyl 3-phenylpropenoate; 3-phenyl-2-propenoic acid phenylmethyl ester; Cinnamic acid benzyl ester | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.002.827 |
| PubChem CID |
|
| |
| |
| Properties | |
| C16H14O2 | |
| Molar mass | 238.29 g·mol−1 |
| Appearance | White to pale yellow solid[1] |
| Melting point | 34–37 °C (93–99 °F; 307–310 K)[2] |
| Boiling point | 195–200 °C (383–392 °F; 468–473 K) 5 mmHg[2] |
| Insoluble[1] | |
| Solubility in ethanol | 125 g/L |
| Solubility in glycerin | Insoluble |
| Solubility in propylene glycol | Insoluble |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Benzyl cinnamate is the chemical compound which is the ester derived from cinnamic acid and benzyl alcohol.
Natural occurrence
Benzyl cinnamate occurs in Balsam of Peru and Tolu balsam, in Sumatra and Penang benzoin, and as the main constituent of copaiba balsam.[3]
Synthesis
Benzyl cinnamate can be prepared by heating benzyl chloride and excess sodium cinnamate in water to 100–115 °C or by heating sodium cinnamate with an excess of benzyl chloride in the presence of diethylamine.[3]
Uses
Benzyl cinnamate is used in heavy oriental perfumes and as a fixative.[4] It is used as a flavoring agent.[3]
References
- 1 2 "Specifications for Flavourings". Food and Agricultural Organization.
- 1 2 "Benzyl cinnamate". Sigma-Aldrich.
- 1 2 3 George A. Burdock (2010), "BENZYL CINNAMATE", Fenaroli's Handbook of Flavor Ingredients (6th ed.), CRC Press, pp. 147–148
- ↑ Karl-Georg Fahlbusch; et al. (2007), "Flavors and Fragrances", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, p. 59
External links
- Benzyl cinnamate at National Library of Medicine's Toxicology Data Network
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.