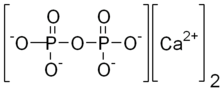
Calcium pyrophosphate
 | |
| Names | |
|---|---|
| IUPAC name
Calcium diphosphate | |
| Other names
Diphosphoric acid, calcium salt (1:2); Dicalcium diphosphate; Dicalcium pyrophosphate | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.292 |
| MeSH | Calcium+pyrophosphate |
| PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| Ca2O7P2 | |
| Molar mass | 254.053 g/mol |
| Appearance | White powder |
| Density | 3.09 g/cm3 |
| Melting point | 1,353 °C (2,467 °F; 1,626 K) |
| insoluble | |
| Solubility | soluble in HCl, nitric acids |
| Refractive index (nD) |
1.585 |
| Hazards | |
| NFPA 704 | |
| Flash point | Non-flammable |
| Related compounds | |
| Other anions |
Calcium phosphate |
| Other cations |
Magnesium pyrophosphate Sodium pyrophosphate |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Calcium pyrophosphate (Ca2P2O7) is a chemical compound, an insoluble calcium salt containing the pyrophosphate anion. There are a number of forms reported: an anhydrous form, a dihydrate, Ca2P2O7·2H2O and a tetrahydrate, Ca2P2O7·4H2O. Deposition of dihydrate crystals in cartilage are responsible for the severe joint pain in cases of calcium pyrophosphate deposition disease (pseudo gout) whose symptoms are similar to those of gout.[1] Ca2P2O7 is commonly used as a mild abrasive agent in toothpastes.[2]
Preparation
Crystals of the tetrahydrate can be prepared by reacting sodium pyrophosphate, Na4P2O7 with calcium nitrate, Ca(NO3)2, at carefully controlled pH and temperature:[3]
- Na4P2O7(aq) +2 Ca(NO3)2(aq) → Ca2P2O7·4 H2O + 2 Na2NO3
The dihydrate, sometimes termed CPPD, can be formed by the reaction of pyrophosphoric acid with calcium chloride:[2]
- CaCl2 + H4P2O7(aq) → Ca2P2O7·2 H2O + HCl.
The anhydrous forms can be prepared by heating dicalcium phosphate:[2]
- 2 CaHPO4 → Ca2P2O7 + H2O
At 240-500 °C an amorphous phase is formed, heating to 750 °C forms β-Ca2P2O7, heating to 1140 - 1350 °C forms the α-Ca2P2O7.
Structure of anhydrous and hydrated forms
The stable tetrahydrate was originally reported to be rhombohedral but is now believed to be monoclinic. Additionally there is an unstable monoclinic form.[3]
The dihydrate is triclinic, with hydrogen bonding between the two water molecules and hydrogen bonds to the O atoms on the anion.[2] An hexagonal dihydrate has also been reported.[4]
The anhydrous form has 3 polymorphs, α-, β-, and a metastable γ. The high temperature form α- is monoclinic, with 8 coordinate calcium, the lower temperature form β- is tetragonal, with calcium in four different coordination environments, 2 that are 7 coordinate, one eight and one 9. In both the pyrophosphates are essentially eclipsed.[5][6]
References
- ↑ "Calcium Pyrophosphate Deposition Disease". Medscape.
- 1 2 3 4 Ropp, R.C. (2013). "Chapter 4 - Group 15 (N, P, As, Sb and Bi) Alkaline Earth Compounds". Encyclopedia of the Alkaline Earth Compounds. 1. Elsevier. ISBN 978-0-444-59550-8. doi:10.1016/B978-0-444-59550-8.00004-1. – via ScienceDirect (Subscription may be required or content may be available in libraries.)
- 1 2 Christoffersen, Margaret R.; Balic-Zunic, Tonci; Pehrson, Søren; Christoffersen, Jørgen (May 2000). "Growth and precipitation of a monoclinic calcium pyrophosphate tetrahydrate indicating auto-inhibition at pH7". Journal of Crystal Growth. 212 (3–4): 500–506. ISSN 0022-0248. doi:10.1016/S0022-0248(00)00231-1. – via ScienceDirect (Subscription may be required or content may be available in libraries.)
- ↑ Mandel, Gretchen S.; Renne, Kathleen M.; Kolbach, Ann M. Kaplan; Wayne D., Miller; Jay D., Mandel; Neil S. (March 1983). "Calcium pyrophosphate crystal deposition disease: Preparation and characterization of crystals". Journal of Crystal Growth. 87 (4): 453–462. ISSN 0022-0248. doi:10.1016/0022-0248(88)90093-0. – via ScienceDirect (Subscription may be required or content may be available in libraries.)
- ↑ Parodi, J. A.; Hickok, R. L.; Segelken, W. G.; Cooper, J. R. (1965). "Electronic Paramagnetic Resonance Study of the Thermal Decomposition of Dibasic Calcium Orthophosphate". Journal of The Electrochemical Society. 112 (7): 688. ISSN 0013-4651. doi:10.1149/1.2423665.
- ↑ Webb, N. C. (1966). "The crystal structure of β-Ca2P2O". Acta Crystallographica. 21 (6): 942–948. ISSN 0365-110X. doi:10.1107/S0365110X66004225.
