Breast cancer
| Breast cancer | |
|---|---|
 | |
| Mammograms showing a normal breast (left) and a breast with cancer (right, white arrows). | |
| Specialty | Oncology |
| Symptoms | Lump in a breast, change in breast shape, dimpling of the skin, fluid coming from the nipple, red scaly patch of skin[1] |
| Risk factors | Female, obesity, lack of exercise, alcohol, hormone replacement therapy during menopause, ionizing radiation, early age at first menstruation, having children late or not at all, older age, family history, Klinefelter syndrome[1][2][3] |
| Diagnostic method | Tissue biopsy[1] |
| Treatment | Surgery, radiation therapy, chemotherapy, hormonal therapy, targeted therapy[1] |
| Prognosis | Five-year survival rate ~85% (US, UK)[4][5] |
| Frequency | 2.1 million affected as of 2015[6] |
| Deaths | 533,600 (2015)[7] |
Breast cancer is cancer that develops from breast tissue.[8] Signs of breast cancer may include a lump in the breast, a change in breast shape, dimpling of the skin, fluid coming from the nipple, or a red scaly patch of skin.[1] In those with distant spread of the disease, there may be bone pain, swollen lymph nodes, shortness of breath, or yellow skin.[9]
Risk factors for developing breast cancer include being female, obesity, lack of physical exercise, drinking alcohol, hormone replacement therapy during menopause, ionizing radiation, early age at first menstruation, having children late or not at all, older age, and family history.[1][2] About 5–10% of cases are due to genes inherited from a person's parents, including BRCA1 and BRCA2 among others. Breast cancer most commonly develops in cells from the lining of milk ducts and the lobules that supply the ducts with milk. Cancers developing from the ducts are known as ductal carcinomas, while those developing from lobules are known as lobular carcinomas.[1] In addition, there are more than 18 other sub-types of breast cancer. Some cancers, such as ductal carcinoma in situ, develop from pre-invasive lesions.[2] The diagnosis of breast cancer is confirmed by taking a biopsy of the concerning lump. Once the diagnosis is made, further tests are done to determine if the cancer has spread beyond the breast and which treatments it may respond to.[1]
The balance of benefits versus harms of breast cancer screening is controversial. A 2013 Cochrane review stated that it is unclear if mammographic screening does more good or harm.[10] A 2009 review for the US Preventive Services Task Force found evidence of benefit in those 40 to 70 years of age,[11] and the organization recommends screening every two years in women 50 to 74 years old.[12] The medications tamoxifen or raloxifene may be used in an effort to prevent breast cancer in those who are at high risk of developing it.[2] Surgical removal of both breasts is another preventative measure in some high risk women.[2] In those who have been diagnosed with cancer, a number of treatments may be used, including surgery, radiation therapy, chemotherapy, hormonal therapy and targeted therapy.[1] Types of surgery vary from breast-conserving surgery to mastectomy.[13][14] Breast reconstruction may take place at the time of surgery or at a later date. In those in whom the cancer has spread to other parts of the body, treatments are mostly aimed at improving quality of life and comfort.[14]
Outcomes for breast cancer vary depending on the cancer type, extent of disease, and person's age.[14] Survival rates in the developed world are high,[15] with between 80% and 90% of those in England and the United States alive for at least 5 years.[5][4] In developing countries survival rates are poorer.[2] Worldwide, breast cancer is the leading type of cancer in women, accounting for 25% of all cases.[16] In 2012 it resulted in 1.68 million new cases and 522,000 deaths.[16] It is more common in developed countries[2] and is more than 100 times more common in women than in men.[15][17]
Signs and symptoms
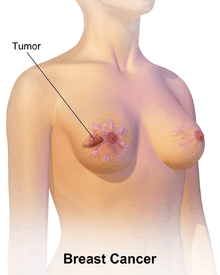

The first noticeable symptom of breast cancer is typically a lump that feels different from the rest of the breast tissue. More than 80% of breast cancer cases are discovered when the woman feels a lump.[18] The earliest breast cancers are detected by a mammogram.[19] Lumps found in lymph nodes located in the armpits[18] can also indicate breast cancer.
Indications of breast cancer other than a lump may include thickening different from the other breast tissue, one breast becoming larger or lower, a nipple changing position or shape or becoming inverted, skin puckering or dimpling, a rash on or around a nipple, discharge from nipple/s, constant pain in part of the breast or armpit, and swelling beneath the armpit or around the collarbone.[20] Pain ("mastodynia") is an unreliable tool in determining the presence or absence of breast cancer, but may be indicative of other breast health issues.[18][19][21]
Inflammatory breast cancer is a particular type of breast cancer which can pose a substantial diagnostic challenge. Symptoms may resemble a breast inflammation and may include itching, pain, swelling, nipple inversion, warmth and redness throughout the breast, as well as an orange-peel texture to the skin referred to as peau d'orange.[18] As inflammatory breast cancer does not present as a lump there can sometimes be a delay in diagnosis.
Another reported symptom complex of breast cancer is Paget's disease of the breast. This syndrome presents as skin changes resembling eczema, such as redness, discoloration, or mild flaking of the nipple skin. As Paget's disease of the breast advances, symptoms may include tingling, itching, increased sensitivity, burning, and pain. There may also be discharge from the nipple. Approximately half of women diagnosed with Paget's disease of the breast also have a lump in the breast.[22]
In rare cases, what initially appears as a fibroadenoma (hard, movable non-cancerous lump) could in fact be a phyllodes tumor. Phyllodes tumors are formed within the stroma (connective tissue) of the breast and contain glandular as well as stromal tissue. Phyllodes tumors are not staged in the usual sense; they are classified on the basis of their appearance under the microscope as benign, borderline, or malignant.[23]
Occasionally, breast cancer presents as metastatic disease—that is, cancer that has spread beyond the original organ. The symptoms caused by metastatic breast cancer will depend on the location of metastasis. Common sites of metastasis include bone, liver, lung and brain.[24] Unexplained weight loss can occasionally signal breast cancer, as can symptoms of fevers or chills. Bone or joint pains can sometimes be manifestations of metastatic breast cancer, as can jaundice or neurological symptoms. These symptoms are called non-specific, meaning they could be manifestations of many other illnesses.[25]
Most symptoms of breast disorders, including most lumps, do not turn out to represent underlying breast cancer. Fewer than 20% of lumps, for example, are cancerous,[26] and benign breast diseases such as mastitis and fibroadenoma of the breast are more common causes of breast disorder symptoms. Nevertheless, the appearance of a new symptom should be taken seriously by both patients and their doctors, because of the possibility of an underlying breast cancer at almost any age.[27]
Risk factors
Risk factors can be divided into two categories:
- modifiable risk factors (things that people can change themselves, such as consumption of alcoholic beverages), and
- fixed risk factors (things that cannot be changed, such as age and biological sex).[28]
The primary risk factors for breast cancer are being female and older age.[29] Other potential risk factors include genetics,[30] lack of childbearing or lack of breastfeeding,[31] higher levels of certain hormones,[32][33] certain dietary patterns, and obesity. Recent studies have indicated that exposure to light pollution is a risk factor for the development of breast cancer.[34]
Lifestyle
Smoking tobacco appears to increase the risk of breast cancer, with the greater the amount smoked and the earlier in life that smoking began, the higher the risk.[35] In those who are long-term smokers, the risk is increased 35% to 50%.[35] A lack of physical activity has been linked to about 10% of cases.[36] Sitting regularly for prolonged periods is associated with higher mortality from breast cancer. The risk is not negated by regular exercise, though it is lowered.[37]
There is an association between use of hormonal birth control and the development of premenopausal breast cancer,[28][38] but whether oral contraceptives use may actually cause premenopausal breast cancer is a matter of debate.[39] If there is indeed a link, the absolute effect is small.[39][40] Additionally, it is not clear if the association exists with newer hormonal birth controls.[40] In those with mutations in the breast cancer susceptibility genes BRCA1 or BRCA2, or who have a family history of breast cancer, use of modern oral contraceptives does not appear to affect the risk of breast cancer.[41][42]
The association between breast feeding and breast cancer has not been clearly determined; some studies have found support for an association while others have not.[43] In the 1980s, the abortion–breast cancer hypothesis posited that induced abortion increased the risk of developing breast cancer.[44] This hypothesis was the subject of extensive scientific inquiry, which concluded that neither miscarriages nor abortions are associated with a heightened risk for breast cancer.[45]
A number of dietary factors have been linked to the risk for breast cancer. Dietary factors which may increase risk include a high fat diet,[46] high alcohol intake,[47] and obesity-related high cholesterol levels.[48][49] Dietary iodine deficiency may also play a role.[50] Evidence for fiber is unclear. A 2015 review found that studies trying to link fiber intake with breast cancer produced mixed results.[51] In 2016 a tentative association between low fiber intake during adolescence and breast cancer was observed.[52]
Other risk factors include radiation[53] and shift-work.[54] A number of chemicals have also been linked, including polychlorinated biphenyls, polycyclic aromatic hydrocarbons, and organic solvents[55] Although the radiation from mammography is a low dose, it is estimated that yearly screening from 40 to 80 years of age will cause approximately 225 cases of fatal breast cancer per million women screened.[56]
Genetics
Some genetic susceptibility may play a minor role in most cases.[57] Overall, however, genetics is believed to be the primary cause of 5–10% of all cases.[58] Women whose mother was diagnosed before 50 have an increased risk of 1.7 and those whose mother was diagnosed at age 50 or after has an increased risk of 1.4.[59] In those with zero, one or two affected relatives, the risk of breast cancer before the age of 80 is 7.8%, 13.3%, and 21.1% with a subsequent mortality from the disease of 2.3%, 4.2%, and 7.6% respectively.[60] In those with a first degree relative with the disease the risk of breast cancer between the age of 40 and 50 is double that of the general population.[61]
In less than 5% of cases, genetics plays a more significant role by causing a hereditary breast–ovarian cancer syndrome.[57] This includes those who carry the BRCA1 and BRCA2 gene mutation.[57] These mutations account for up to 90% of the total genetic influence with a risk of breast cancer of 60–80% in those affected.[58] Other significant mutations include p53 (Li–Fraumeni syndrome), PTEN (Cowden syndrome), and STK11 (Peutz–Jeghers syndrome), CHEK2, ATM, BRIP1, and PALB2.[58] In 2012, researchers said that there are four genetically distinct types of the breast cancer and that in each type, hallmark genetic changes lead to many cancers.[62]
Medical conditions
Breast changes like atypical ductal hyperplasia[63] and lobular carcinoma in situ,[64][65][66] found in benign breast conditions such as fibrocystic breast changes, are correlated with an increased breast cancer risk. Diabetes mellitus might also increase the risk of breast cancer.[67] Autoimmune diseases such as lupus erythematosus seem also to increase the risk for the acquisition of breast cancer.[68]
Pathophysiology

Breast cancer, like other cancers, occurs because of an interaction between an environmental (external) factor and a genetically susceptible host. Normal cells divide as many times as needed and stop. They attach to other cells and stay in place in tissues. Cells become cancerous when they lose their ability to stop dividing, to attach to other cells, to stay where they belong, and to die at the proper time.
Normal cells will commit cell suicide (programmed cell death) when they are no longer needed. Until then, they are protected from cell suicide by several protein clusters and pathways. One of the protective pathways is the PI3K/AKT pathway; another is the RAS/MEK/ERK pathway. Sometimes the genes along these protective pathways are mutated in a way that turns them permanently "on", rendering the cell incapable of committing suicide when it is no longer needed. This is one of the steps that causes cancer in combination with other mutations. Normally, the PTEN protein turns off the PI3K/AKT pathway when the cell is ready for programmed cell death. In some breast cancers, the gene for the PTEN protein is mutated, so the PI3K/AKT pathway is stuck in the "on" position, and the cancer cell does not commit suicide.[69]
Mutations that can lead to breast cancer have been experimentally linked to estrogen exposure.[70]
Abnormal growth factor signaling in the interaction between stromal cells and epithelial cells can facilitate malignant cell growth.[71][72] In breast adipose tissue, overexpression of leptin leads to increased cell proliferation and cancer.[73]
In the United States, 10 to 20 percent of people with breast cancer and people with ovarian cancer have a first- or second-degree relative with one of these diseases. The familial tendency to develop these cancers is called hereditary breast–ovarian cancer syndrome. The best known of these, the BRCA mutations, confer a lifetime risk of breast cancer of between 60 and 85 percent and a lifetime risk of ovarian cancer of between 15 and 40 percent. Some mutations associated with cancer, such as p53, BRCA1 and BRCA2, occur in mechanisms to correct errors in DNA. These mutations are either inherited or acquired after birth. Presumably, they allow further mutations, which allow uncontrolled division, lack of attachment, and metastasis to distant organs.[53][74] However, there is strong evidence of residual risk variation that goes well beyond hereditary BRCA gene mutations between carrier families. This is caused by unobserved risk factors.[75] This implicates environmental and other causes as triggers for breast cancers. The inherited mutation in BRCA1 or BRCA2 genes can interfere with repair of DNA cross links and DNA double strand breaks (known functions of the encoded protein).[76] These carcinogens cause DNA damage such as DNA cross links and double strand breaks that often require repairs by pathways containing BRCA1 and BRCA2.[77][78] However, mutations in BRCA genes account for only 2 to 3 percent of all breast cancers.[79] Levin et al. say that cancer may not be inevitable for all carriers of BRCA1 and BRCA2 mutations.[80] About half of hereditary breast–ovarian cancer syndromes involve unknown genes.
GATA-3 directly controls the expression of estrogen receptor (ER) and other genes associated with epithelial differentiation, and the loss of GATA-3 leads to loss of differentiation and poor prognosis due to cancer cell invasion and metastasis.[81]
Diagnosis

Most types of breast cancer are easy to diagnose by microscopic analysis of a sample—or biopsy—of the affected area of the breast. Also, there are types of breast cancer that require specialized lab exams.
The two most commonly used screening methods, physical examination of the breasts by a healthcare provider and mammography, can offer an approximate likelihood that a lump is cancer, and may also detect some other lesions, such as a simple cyst.[82] When these examinations are inconclusive, a healthcare provider can remove a sample of the fluid in the lump for microscopic analysis (a procedure known as fine needle aspiration, or fine needle aspiration and cytology—FNAC) to help establish the diagnosis. The needle aspiration may be performed in a healthcare provider's office or clinic using local anaesthetic if required. A finding of clear fluid makes the lump highly unlikely to be cancerous, but bloody fluid may be sent off for inspection under a microscope for cancerous cells. Together, physical examination of the breasts, mammography, and FNAC can be used to diagnose breast cancer with a good degree of accuracy.
Other options for biopsy include a core biopsy or vacuum-assisted breast biopsy,[83] which are procedures in which a section of the breast lump is removed; or an excisional biopsy, in which the entire lump is removed. Very often the results of physical examination by a healthcare provider, mammography, and additional tests that may be performed in special circumstances (such as imaging by ultrasound or MRI) are sufficient to warrant excisional biopsy as the definitive diagnostic and primary treatment method.
 MRI showing breast cancer
MRI showing breast cancer High-grade invasive ductal carcinoma, with minimal tubule formation, marked pleomorphism, and prominent mitoses, 40x field.
High-grade invasive ductal carcinoma, with minimal tubule formation, marked pleomorphism, and prominent mitoses, 40x field. Micrograph showing a lymph node invaded by ductal breast carcinoma, with an extension of the tumor beyond the lymph node.
Micrograph showing a lymph node invaded by ductal breast carcinoma, with an extension of the tumor beyond the lymph node._expression_in_normal_breast_and_breast_carcinoma_tissue.jpg) Neuropilin-2 expression in normal breast and breast carcinoma tissue.
Neuropilin-2 expression in normal breast and breast carcinoma tissue.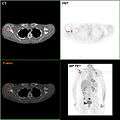 F-18 FDG PET/CT: A breast cancer metastasis to the right scapula
F-18 FDG PET/CT: A breast cancer metastasis to the right scapula Needle breast biopsy.
Needle breast biopsy. Elastography shows stiff cancer tissue on ultrasound imaging.
Elastography shows stiff cancer tissue on ultrasound imaging. Ultrasound image shows irregularly shaped mass of breast cancer.
Ultrasound image shows irregularly shaped mass of breast cancer. Infiltrating (Invasive) breast carcinoma.
Infiltrating (Invasive) breast carcinoma.
Classification
Breast cancers are classified by several grading systems. Each of these influences the prognosis and can affect treatment response. Description of a breast cancer optimally includes all of these factors.
- Histopathology. Breast cancer is usually classified primarily by its histological appearance. Most breast cancers are derived from the epithelium lining the ducts or lobules, and these cancers are classified as ductal or lobular carcinoma. Carcinoma in situ is growth of low-grade cancerous or precancerous cells within a particular tissue compartment such as the mammary duct without invasion of the surrounding tissue. In contrast, invasive carcinoma does not confine itself to the initial tissue compartment.[84]
- Grade. Grading compares the appearance of the breast cancer cells to the appearance of normal breast tissue. Normal cells in an organ like the breast become differentiated, meaning that they take on specific shapes and forms that reflect their function as part of that organ. Cancerous cells lose that differentiation. In cancer, the cells that would normally line up in an orderly way to make up the milk ducts become disorganized. Cell division becomes uncontrolled. Cell nuclei become less uniform. Pathologists describe cells as well differentiated (low grade), moderately differentiated (intermediate grade), and poorly differentiated (high grade) as the cells progressively lose the features seen in normal breast cells. Poorly differentiated cancers (the ones whose tissue is least like normal breast tissue) have a worse prognosis.
- Stage. Breast cancer staging using the TNM system is based on the size of the tumor (T), whether or not the tumor has spread to the lymph nodes (N) in the armpits, and whether the tumor has metastasized (M) (i.e. spread to a more distant part of the body). Larger size, nodal spread, and metastasis have a larger stage number and a worse prognosis.
The main stages are:- Stage 0 is a pre-cancerous or marker condition, either ductal carcinoma in situ (DCIS) or lobular carcinoma in situ (LCIS).
- Stages 1–3 are within the breast or regional lymph nodes.
- Stage 4 is 'metastatic' cancer that has a less favorable prognosis since it has spread beyond the breast and regional lymph nodes.
 Stage T1 breast cancer
Stage T1 breast cancer Stage T2 breast cancer
Stage T2 breast cancer Stage T3 breast cancer
Stage T3 breast cancer
- Where available, imaging studies may be employed as part of the staging process in select cases to look for signs of metastatic cancer. However, in cases of breast cancer with low risk for metastasis, the risks associated with PET scans, CT scans, or bone scans outweigh the possible benefits, as these procedures expose the patient to a substantial amount of potentially dangerous ionizing radiation.[85][86]
- Receptor status. Breast cancer cells have receptors on their surface and in their cytoplasm and nucleus. Chemical messengers such as hormones bind to receptors, and this causes changes in the cell. Breast cancer cells may or may not have three important receptors: estrogen receptor (ER), progesterone receptor (PR), and HER2.
ER+ cancer cells (that is, cancer cells that have estrogen receptors) depend on estrogen for their growth, so they can be treated with drugs to block estrogen effects (e.g. tamoxifen), and generally have a better prognosis. Untreated, HER2+ breast cancers are generally more aggressive than HER2- breast cancers,[87][88] but HER2+ cancer cells respond to drugs such as the monoclonal antibody trastuzumab (in combination with conventional chemotherapy), and this has improved the prognosis significantly.[89] Cells that do not have any of these three receptor types (estrogen receptors, progesterone receptors, or HER2) are called triple-negative, although they frequently do express receptors for other hormones, such as androgen receptor and prolactin receptor. - DNA assays. DNA testing of various types including DNA microarrays have compared normal cells to breast cancer cells. The specific changes in a particular breast cancer can be used to classify the cancer in several ways, and may assist in choosing the most effective treatment for that DNA type.
 Stage 1A breast cancer
Stage 1A breast cancer Stage 1B breast cancer
Stage 1B breast cancer Stage 2A breast cancer
Stage 2A breast cancer Stage 2A breast cancer
Stage 2A breast cancer Stage 2B breast cancer
Stage 2B breast cancer Stage 2B breast cancer
Stage 2B breast cancer Stage 2B breast cancer
Stage 2B breast cancer Stage 3A breast cancer
Stage 3A breast cancer Stage 3A breast cancer
Stage 3A breast cancer Stage 3A breast cancer
Stage 3A breast cancer Stage 3B breast cancer
Stage 3B breast cancer Stage 3B breast cancer
Stage 3B breast cancer Stage 4 breast cancer
Stage 4 breast cancer
Prevention
Life-style
Women may reduce their risk of breast cancer by maintaining a healthy weight, drinking less alcohol, being physically active and breastfeeding their children.[90] These modifications might prevent 38% of breast cancers in the US, 42% in the UK, 28% in Brazil and 20% in China.[90] The benefits with moderate exercise such as brisk walking are seen at all age groups including postmenopausal women.[90][91] High levels of physical activity reduce the risk of breast cancer by about 14%.[92] Strategies that encourage regular physical activity and reduce obesity could also have other benefits, such as reduced risks of cardiovascular disease and diabetes.[28]
High intake of citrus fruit has been associated with a 10% reduction in the risk of breast cancer.[93]
Marine omega-3 polyunsaturated fatty acids appear to reduce the risk.[94] High consumption of soy-based foods may reduce risk.[95]
Pre-emptive surgery
Removal of both breasts before any cancer has been diagnosed or any suspicious lump or other lesion has appeared (a procedure known as prophylactic bilateral mastectomy) may be considered in people with BRCA1 and BRCA2 mutations, which are associated with a substantially heightened risk for an eventual diagnosis of breast cancer.[96][97] Evidence is not strong enough to support this procedure in anyone but those at the highest risk.[98] BRCA testing is recommended in those with a high family risk after genetic counseling. It is not recommended routinely.[99] This is because there are many forms of changes in BRCA genes, ranging from harmless polymorphisms to obviously dangerous frameshift mutations. The effect of most of the identifiable changes in the genes is uncertain. Testing in an average-risk person is particularly likely to return one of these indeterminate, useless results. It is unclear if removing the second breast in those who have breast cancer in one is beneficial.[98]
Medications
The selective estrogen receptor modulators (such as tamoxifen) reduce the risk of breast cancer but increase the risk of thromboembolism and endometrial cancer.[100][100] There is no overall change in the risk of death.[100][101] They are thus not recommended for the prevention of breast cancer in women at average risk but may be offered for those at high risk.[102] The benefit of breast cancer reduction continues for at least five years after stopping a course of treatment with these medications.[103]
Screening
Breast cancer screening refers to testing otherwise-healthy women for breast cancer in an attempt to achieve an earlier diagnosis under the assumption that early detection will improve outcomes. A number of screening tests have been employed including clinical and self breast exams, mammography, genetic screening, ultrasound, and magnetic resonance imaging.
A clinical or self breast exam involves feeling the breast for lumps or other abnormalities. Clinical breast exams are performed by health care providers, while self-breast exams are performed by the person themselves.[104] Evidence does not support the effectiveness of either type of breast exam, as by the time a lump is large enough to be found it is likely to have been growing for several years and thus soon be large enough to be found without an exam.[105][106] Mammographic screening for breast cancer uses X-rays to examine the breast for any uncharacteristic masses or lumps. During a screening, the breast is compressed and a technician takes photos from multiple angles. A general mammogram takes photos of the entire breast, while a diagnostic mammogram focuses on a specific lump or area of concern.[107]
A number of national bodies recommend breast cancer screening. For the average woman, the U.S. Preventive Services Task Force recommends mammography every two years in women between the ages of 50 and 74,[12] the Council of Europe recommends mammography between 50 and 69 with most programs using a 2-year frequency,[108] and in Canada screening is recommended between the ages of 50 and 74 at a frequency of 2 to 3 years.[109] These task force reports point out that in addition to unnecessary surgery and anxiety, the risks of more frequent mammograms include a small but significant increase in breast cancer induced by radiation.[110]
The Cochrane collaboration (2013) states that the best quality evidence neither demonstrates a reduction in cancer specific, nor a reduction in all cause mortality from screening mammography.[10] When less rigorous trials are added to the analysis there is a reduction in mortality due to breast cancer of 0.05% (a decrease of 1 in 2000 deaths from breast cancer over 10 years or a relative decrease of 15% from breast cancer).[10] Screening over 10 years results in a 30% increase in rates of over-diagnosis and over-treatment (3 to 14 per 1000) and more than half will have at least one falsely positive test.[10][111][112] This has resulted in the view that it is not clear whether mammography screening does more good or harm.[10] Cochrane states that, due to recent improvements in breast cancer treatment, and the risks of false positives from breast cancer screening leading to unnecessary treatment, "it therefore no longer seems beneficial to attend for breast cancer screening" at any age.[113] Whether MRI as a screening method has greater harms or benefits when compared to standard mammography is not known.[114]
Management
The management of breast cancer depends on various factors, including the stage of the cancer and the age of the patient. Increasingly aggressive treatments are employed in accordance with the poorer the patient's prognosis and the higher the risk of recurrence of the cancer following treatment.
Breast cancer is usually treated with surgery, which may be followed by chemotherapy or radiation therapy, or both. A multidisciplinary approach is preferable.[115] Hormone receptor-positive cancers are often treated with hormone-blocking therapy over courses of several years. Monoclonal antibodies, or other immune-modulating treatments, may be administered in certain cases of metastatic and other advanced stages of breast cancer.
Surgery
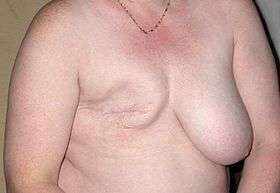
Surgery involves the physical removal of the tumor, typically along with some of the surrounding tissue. One or more lymph nodes may be biopsied during the surgery; increasingly the lymph node sampling is performed by a sentinel lymph node biopsy.
Standard surgeries include:
- Mastectomy: Removal of the whole breast.
- Quadrantectomy: Removal of one-quarter of the breast.
- Lumpectomy: Removal of a small part of the breast.
Once the tumor has been removed, if the patient desires, breast reconstruction surgery, a type of plastic surgery, may then be performed to improve the aesthetic appearance of the treated site. Alternatively, women use breast prostheses to simulate a breast under clothing, or choose a flat chest. Nipple prosthesis can be used at any time following the mastectomy.
Medication
Drugs used after and in addition to surgery are called adjuvant therapy. Chemotherapy or other types of therapy prior to surgery are called neoadjuvant therapy. Aspirin may reduce mortality from breast cancer.[116]
There are currently three main groups of medications used for adjuvant breast cancer treatment: hormone-blocking agents, chemotherapy, and monoclonal antibodies.
- Some breast cancers require estrogen to continue growing. They can be identified by the presence of estrogen receptors (ER+) and progesterone receptors (PR+) on their surface (sometimes referred to together as hormone receptors). These ER+ cancers can be treated with drugs that either block the receptors, e.g. tamoxifen, or alternatively block the production of estrogen with an aromatase inhibitor, e.g. anastrozole[117] or letrozole. The use of tamoxifen is recommended for 10 years.[118] Letrozole is recommended for 5 years. Aromatase inhibitors are only suitable for women after menopause; however, in this group, they appear better than tamoxifen.[119] This is because the active aromatase in postmenopausal women is different from the prevalent form in premenopausal women, and therefore these agents are ineffective in inhibiting the predominant aromatase of premenopausal women.[120] Aromatase inhibitors should not be given to premenopausal women with intact ovarian function (unless they are also on treatment to stop their ovaries from working).[121]
- Chemotherapy is predominantly used for cases of breast cancer in stages 2–4, and is particularly beneficial in estrogen receptor-negative (ER-) disease. The chemotherapy medications are administered in combinations, usually for periods of 3–6 months. One of the most common regimens, known as "AC", combines cyclophosphamide with doxorubicin. Sometimes a taxane drug, such as docetaxel, is added, and the regime is then known as "CAT". Another common treatment is cyclophosphamide, methotrexate, and fluorouracil (or "CMF"). Most chemotherapy medications work by destroying fast-growing and/or fast-replicating cancer cells, either by causing DNA damage upon replication or by other mechanisms. However, the medications also damage fast-growing normal cells, which may cause serious side effects. Damage to the heart muscle is the most dangerous complication of doxorubicin, for example.
- Trastuzumab, a monoclonal antibody to HER2 (a cell receptor that is especially active in some breast cancer cells), has improved the 5-year disease free survival of stage 1–3 HER2-positive breast cancers to about 87% (overall survival 95%).[122] When stimulated by certain growth factors, HER2 causes cellular growth and division; in the absence of stimulation by the growth factor, the cell will normally stop growing. Between 25% and 30% of breast cancers overexpress the HER2 gene or its protein product,[123] and overexpression of HER2 in breast cancer is associated with increased disease recurrence and worse prognosis. When trastuzumab binds to the HER2 in breast cancer cells that overexpress the receptor, trastuzumab prevents growth factors from being able to bind to and stimulate the receptors, effectively blocking the growth of the cancer cells. Trastuzumab, however, is very expensive, and its use may cause serious side effects (approximately 2% of patients who receive it suffer significant heart damage).[124] Further, trastuzumab is only effective in patients with HER2 amplification/overexpression.
Radiation

Radiotherapy is given after surgery to the region of the tumor bed and regional lymph nodes, to destroy microscopic tumor cells that may have escaped surgery. It may also have a beneficial effect on tumor microenvironment.[125][126] Radiation therapy can be delivered as external beam radiotherapy or as brachytherapy (internal radiotherapy). Conventionally radiotherapy is given after the operation for breast cancer. Radiation can also be given at the time of operation on the breast cancer. Radiation can reduce the risk of recurrence by 50–66% (1/2 – 2/3 reduction of risk) when delivered in the correct dose[127] and is considered essential when breast cancer is treated by removing only the lump (Lumpectomy or Wide local excision).
Prognosis

Prognosis is usually given for the probability of progression-free survival (PFS) or disease-free survival (DFS). These predictions are based on experience with breast cancer patients with similar classification. A prognosis is an estimate, as patients with the same classification will survive a different amount of time, and classifications are not always precise. Survival is usually calculated as an average number of months (or years) that 50% of patients survive, or the percentage of patients that are alive after 1, 5, 15, and 20 years. Prognosis is important for treatment decisions because patients with a good prognosis are usually offered less invasive treatments, such as lumpectomy and radiation or hormone therapy, while patients with poor prognosis are usually offered more aggressive treatment, such as more extensive mastectomy and one or more chemotherapy drugs.
Prognostic factors
Prognostic factors are reflected in the classification scheme for breast cancer including stage, (i.e., tumor size, location, whether disease has spread to lymph nodes and other parts of the body), grade, recurrence of the disease, and the age and health of the patient. The Nottingham Prognostic Index is a commonly used prognostic tool.
The stage of the breast cancer is the most important component of traditional classification methods of breast cancer, because it has a greater effect on the prognosis than the other considerations. Staging takes into consideration size, local involvement, lymph node status and whether metastatic disease is present. The higher the stage at diagnosis, the poorer the prognosis. The stage is raised by the invasiveness of disease to lymph nodes, chest wall, skin or beyond, and the aggressiveness of the cancer cells. The stage is lowered by the presence of cancer-free zones and close-to-normal cell behaviour (grading). Size is not a factor in staging unless the cancer is invasive. For example, Ductal Carcinoma In Situ (DCIS) involving the entire breast will still be stage zero and consequently an excellent prognosis with a 10-year disease free survival of about 98%.[128]
- Stage 1 cancers (and DCIS, LCIS) have an excellent prognosis and are generally treated with lumpectomy and sometimes radiation.[129] HER2+ cancers should be treated with the trastuzumab (Herceptin) regime.[130] Chemotherapy is uncommon for other types of stage 1 cancers.
- Stage 2 and 3 cancers with a progressively poorer prognosis and greater risk of recurrence are generally treated with surgery (lumpectomy or mastectomy with or without lymph node removal), chemotherapy (plus trastuzumab for HER2+ cancers) and sometimes radiation (particularly following large cancers, multiple positive nodes or lumpectomy).
- Stage 4, metastatic cancer, (i.e. spread to distant sites) has poor prognosis and is managed by various combination of all treatments from surgery, radiation, chemotherapy and targeted therapies. Ten-year survival rate is 5% without treatment and 10% with optimal treatment.[131]
The breast cancer grade is assessed by comparison of the breast cancer cells to normal breast cells. The closer to normal the cancer cells are, the slower their growth and the better the prognosis. If cells are not well differentiated, they will appear immature, will divide more rapidly, and will tend to spread. Well differentiated is given a grade of 1, moderate is grade 2, while poor or undifferentiated is given a higher grade of 3 or 4 (depending upon the scale used). The most widely used grading system is the Nottingham scheme;[132] details are provided in the discussion of breast cancer grade.
The presence of estrogen and progesterone receptors in the cancer cell is important in guiding treatment. Those who do not test positive for these specific receptors will not be able to respond to hormone therapy, and this can affect their chance of survival depending upon what treatment options remain, the exact type of cancer, and how advanced the disease is.
In addition to hormone receptors, there are other cell surface proteins that may affect prognosis and treatment. HER2 status directs the course of treatment. Patients whose cancer cells are positive for HER2 have a more aggressive disease and may be treated with the 'targeted therapy', trastuzumab (Herceptin), a monoclonal antibody that targets this protein and improves the prognosis significantly.
Younger women with an age of less than 40 years or women over 80 years tend to have a poorer prognosis than post-menopausal women due to several factors. Their breasts may change with their menstrual cycles, they may be nursing infants, and they may be unaware of changes in their breasts. Therefore, younger women are usually at a more advanced stage when diagnosed. There may also be biologic factors contributing to a higher risk of disease recurrence for younger women with breast cancer.[133][134]
High mammographic breast density, which is a marker of increased risk of developing breast cancer, may not mean an increased risk of death among breast cancer patients, according to a 2012 report of a study involving 9232 women by the National Cancer Institute (NCI).[135] On the other hand, more recent research has shown that women with extremely low mammographic densities (<10%) hold a significantly worse prognosis compared to women with other densities, irrespective of all possible confounding factors.[136]
Since breast cancer in males is usually detected at later stages, outcomes are typically worse.[137]
Psychological aspects
The emotional impact of cancer diagnosis, symptoms, treatment, and related issues can be severe. Most larger hospitals are associated with cancer support groups which provide a supportive environment to help patients cope and gain perspective from cancer survivors.
Not all breast cancer patients experience their illness in the same manner. Factors such as age can have a significant impact on the way a patient copes with a breast cancer diagnosis. Premenopausal women with estrogen-receptor positive breast cancer must confront the issues of early menopause induced by many of the chemotherapy regimens used to treat their breast cancer, especially those that use hormones to counteract ovarian function.[138]
On the other hand, a small 2007 study conducted by researchers at the College of Public Health of the University of Georgia suggested a need for greater attention to promoting functioning and psychological well-being among older cancer survivors, even when they may not have obvious cancer-related medical complications.[139] The study found that older breast cancer survivors showed multiple indications of decrements in their health-related quality of life, and lower psychosocial well-being than a comparison group. Survivors reported no more depressive symptoms or anxious mood than the comparison group, however, they did score lower on measures of positive psychosocial well-being and reported more depressed mood and days affected by fatigue. As the incidence of breast cancer in women over 50 rises and survival rates increase, breast cancer is increasingly becoming a geriatric issue that warrants both further research and the expansion of specialized cancer support services tailored for specific age groups.[139]
Epidemiology

|
no data
<2
2–4
4–6
6–8
8–10
10–12
|
12–14
14–16
16–18
18–20
20–22
>22
|
Worldwide, breast cancer is the most common invasive cancer in women.[141] It affects about 12% of women worldwide.[141] (The most common form of cancer is non-invasive non-melanoma skin cancer; non-invasive cancers are generally easily cured, cause very few deaths, and are routinely excluded from cancer statistics.) Breast cancer comprises 22.9% of invasive cancers in women[142] and 16% of all female cancers.[143] In 2012, it comprised 25.2% of cancers diagnosed in women, making it the most common female cancer.[144]
In 2008, breast cancer caused 458,503 deaths worldwide (13.7% of cancer deaths in women and 6.0% of all cancer deaths for men and women together).[142] Lung cancer, the second most common cause of cancer-related death in women, caused 12.8% of cancer deaths in women (18.2% of all cancer deaths for men and women together).[142]
The incidence of breast cancer varies greatly around the world: it is lowest in less-developed countries and greatest in the more-developed countries. In the twelve world regions, the annual age-standardized incidence rates per 100,000 women are as follows: in Eastern Asia, 18; South Central Asia, 22; sub-Saharan Africa, 22; South-Eastern Asia, 26; North Africa and Western Asia, 28; South and Central America, 42; Eastern Europe, 49; Southern Europe, 56; Northern Europe, 73; Oceania, 74; Western Europe, 78; and in North America, 90.[145]
The number of cases worldwide has significantly increased since the 1970s, a phenomenon partly attributed to the modern lifestyles.[146][147] Breast cancer is strongly related to age with only 5% of all breast cancers occurring in women under 40 years old.[148] There were more than 41,000 newly diagnosed cases of breast cancer registered in England in 2011, around 80% of these cases were in women age 50 or older [149] Based on U.S. statistics in 2015 there were 2.8 million women affected by breast cancer.[141]
History
Because of its visibility, breast cancer was the form of cancer most often described in ancient documents.[150] Because autopsies were rare, cancers of the internal organs were essentially invisible to ancient medicine. Breast cancer, however, could be felt through the skin, and in its advanced state often developed into fungating lesions: the tumor would become necrotic (die from the inside, causing the tumor to appear to break up) and ulcerate through the skin, weeping fetid, dark fluid.[150]
The oldest evidence of breast cancer was discovered in Egypt in 2015 and dates back to the Sixth Dynasty.[151] The study of a woman's remains from the necropolis of Qubbet el-Hawa showed the typical destructive damage due to metastatic spread.[151] The Edwin Smith Papyrus describes 8 cases of tumors or ulcers of the breast that were treated by cauterization. The writing says about the disease, "There is no treatment."[152] For centuries, physicians described similar cases in their practices, with the same conclusion. Ancient medicine, from the time of the Greeks through the 17th century, was based on humoralism, and thus believed that breast cancer was generally caused by imbalances in the fundamental fluids that controlled the body, especially an excess of black bile.[153] Alternatively, patients often saw it as divine punishment.[154] In the 18th century, a wide variety of medical explanations were proposed, including a lack of sexual activity, too much sexual activity, physical injuries to the breast, curdled breast milk, and various forms of lymphatic blockages, either internal or due to restrictive clothing.[153][155] In the 19th century, the Scottish surgeon John Rodman said that fear of cancer caused cancer, and that this anxiety, learned by example from the mother, accounted for breast cancer's tendency to run in families.[155]
Although breast cancer was known in ancient times, it was uncommon until the 19th century, when improvements in sanitation and control of deadly infectious diseases resulted in dramatic increases in lifespan. Previously, most women had died too young to have developed breast cancer.[155] Additionally, early and frequent childbearing and breastfeeding probably reduced the rate of breast cancer development in those women who did survive to middle age.[155]
Because ancient medicine believed that the cause was systemic, rather than local, and because surgery carried a high mortality rate, the preferred treatments tended to be pharmacological rather than surgical. Herbal and mineral preparations, especially involving the poison arsenic, were relatively common.
Mastectomy for breast cancer was performed at least as early as AD 548, when it was proposed by the court physician Aetios of Amida to Theodora.[150] It was not until doctors achieved greater understanding of the circulatory system in the 17th century that they could link breast cancer's spread to the lymph nodes in the armpit. The French surgeon Jean Louis Petit (1674–1750) and later the Scottish surgeon Benjamin Bell (1749–1806) were the first to remove the lymph nodes, breast tissue, and underlying chest muscle.[156]
Their successful work was carried on by William Stewart Halsted who started performing radical mastectomies in 1882, helped greatly by advances in general surgical technology, such as aseptic technique and anesthesia. The Halsted radical mastectomy often involved removing both breasts, associated lymph nodes, and the underlying chest muscles. This often led to long-term pain and disability, but was seen as necessary in order to prevent the cancer from recurring.[157] Before the advent of the Halsted radical mastectomy, 20-year survival rates were only 10%; Halsted's surgery raised that rate to 50%.[158] Extending Halsted's work, Jerome Urban promoted superradical mastectomies, taking even more tissue, until 1963, when the ten-year survival rates proved equal to the less-damaging radical mastectomy.[157]
Radical mastectomies remained the standard of care in America until the 1970s, but in Europe, breast-sparing procedures, often followed radiation therapy, were generally adopted in the 1950s.[157] One reason for this striking difference in approach may be the structure of the medical professions: European surgeons, descended from the barber surgeon, were held in less esteem than physicians; in America, the surgeon was the king of the medical profession.[157] Additionally, there were far more European women surgeons: Less than one percent of American surgical oncologists were female, but some European breast cancer wards boasted a medical staff that was half female.[157] American health insurance companies also paid surgeons more to perform radical mastectomies than they did to perform more intricate breast-sparing surgeries.[157]
Breast cancer staging systems were developed in the 1920s and 1930s.[157]
During the 1970s, a new understanding of metastasis led to perceiving cancer as a systemic illness as well as a localized one, and more sparing procedures were developed that proved equally effective. Modern chemotherapy developed after World War II.[159]
The French surgeon Bernard Peyrilhe (1737–1804) realized the first experimental transmission of cancer by injecting extracts of breast cancer into an animal.
Prominent women who died of breast cancer include Anne of Austria, the mother of Louis XIV of France; Mary Washington, mother of George, and Rachel Carson, the environmentalist.[160]
The first case-controlled study on breast cancer epidemiology was done by Janet Lane-Claypon, who published a comparative study in 1926 of 500 breast cancer cases and 500 control patients of the same background and lifestyle for the British Ministry of Health.[161]
In the 1980s and 1990s, thousands of women who had successfully completed standard treatment then demanded and received high-dose bone marrow transplants, thinking this would lead to better long-term survival. However, it proved completely ineffective, and 15–20% of women died because of the brutal treatment.[162]
The 1995 reports from the Nurses' Health Study and the 2002 conclusions of the Women's Health Initiative trial conclusively proved that hormone replacement therapy significantly increased the incidence of breast cancer.[162]
Society and culture
Before the 20th century, breast cancer was feared and discussed in hushed tones, as if it were shameful. As little could be safely done with primitive surgical techniques, women tended to suffer silently rather than seeking care. When surgery advanced, and long-term survival rates improved, women began raising awareness of the disease and the possibility of successful treatment. The "Women's Field Army", run by the American Society for the Control of Cancer (later the American Cancer Society) during the 1930s and 1940s was one of the first organized campaigns. In 1952, the first peer-to-peer support group, called "Reach to Recovery", began providing post-mastectomy, in-hospital visits from women who had survived breast cancer.[163]
The breast cancer movement of the 1980s and 1990s developed out of the larger feminist movements and women's health movement of the 20th century.[164] This series of political and educational campaigns, partly inspired by the politically and socially effective AIDS awareness campaigns, resulted in the widespread acceptance of second opinions before surgery, less invasive surgical procedures, support groups, and other advances in patient care.[165]
Pink ribbon

A pink ribbon is the most prominent symbol of breast cancer awareness. Pink ribbons, which can be made inexpensively, are sometimes sold as fundraisers, much like poppies on Remembrance Day. They may be worn to honor those who have been diagnosed with breast cancer, or to identify products that the manufacturer would like to sell to consumers that are interested in breast cancer.[166]
The pink ribbon is associated with individual generosity, faith in scientific progress, and a "can-do" attitude. It encourages consumers to focus on the emotionally appealing ultimate vision of a cure for breast cancer, rather than on the fraught path between current knowledge and any future cures.[167]
Wearing or displaying a pink ribbon has been criticized by the opponents of this practice as a kind of slacktivism, because it has no practical positive effect. It has also been criticized as hypocrisy, because some people wear the pink ribbon to show good will towards women with breast cancer, but then oppose these women's practical goals, like patient rights and anti-pollution legislation.[168][169] Critics say that the feel-good nature of pink ribbons and pink consumption distracts society from the lack of progress on preventing and curing breast cancer.[170] It is also criticized for reinforcing gender stereotypes and objectifying women and their breasts.[171] Breast Cancer Action launched the "Think Before You Pink" campaign, and said that businesses have co-opted the pink campaign to promote products that cause breast cancer, such as alcoholic beverages.[172]
Breast cancer culture
Breast cancer culture, or pink ribbon culture, is the set of activities, attitudes, and values that surround and shape breast cancer in public. The dominant values are selflessness, cheerfulness, unity, and optimism. Appearing to have suffered bravely is the passport into the culture.
The woman with breast cancer is given a cultural template that constrains her emotional and social responses into a socially acceptable discourse: She is to use the emotional trauma of being diagnosed with breast cancer and the suffering of extended treatment to transform herself into a stronger, happier and more sensitive person who is grateful for the opportunity to become a better person. Breast cancer therapy becomes a rite of passage rather than a disease.[173] To fit into this mold, the woman with breast cancer needs to normalize and feminize her appearance, and minimize the disruption that her health issues cause anyone else. Anger, sadness, and negativity must be silenced.[173]
As with most cultural models, people who conform to the model are given social status, in this case as cancer survivors. Women who reject the model are shunned, punished and shamed.[173]
The culture is criticized for treating adult women like little girls, as evidenced by "baby" toys such as pink teddy bears given to adult women.[173]
The primary purposes or goals of breast cancer culture are to maintain breast cancer's dominance as the preëminent women's health issue, to promote the appearance that society is "doing something" effective about breast cancer, and to sustain and expand the social, political, and financial power of breast cancer activists.[174]
Emphasis
Compared to other diseases or other cancers, breast cancer receives a proportionately greater share of resources and attention. In 2001 MP Ian Gibson, chairman of the House of Commons of the United Kingdom all party group on cancer stated "The treatment has been skewed by the lobbying, there is no doubt about that. Breast cancer sufferers get better treatment in terms of bed spaces, facilities and doctors and nurses."[175] Breast cancer also receives significantly more media coverage than other, equally prevalent cancers, with a study by Prostate Coalition showing 2.6 breast cancer stories for each one covering cancer of the prostate.[176] Ultimately there is a concern that favoring sufferers of breast cancer with disproportionate funding and research on their behalf may well be costing lives elsewhere.[175] Partly because of its relatively high prevalence and long-term survival rates, research is biased towards breast cancer. Some subjects, such as cancer-related fatigue, have been studied little except in women with breast cancer.
One result of breast cancer's high visibility is that statistical results can sometimes be misinterpreted, such as the claim that one in eight women will be diagnosed with breast cancer during their lives—a claim that depends on the unrealistic assumption that no woman will die of any other disease before the age of 95.[177] This obscures the reality, which is that about ten times as many women will die from heart disease or stroke than from breast cancer.[178]
The emphasis on breast cancer screening may be harming women by subjecting them to unnecessary radiation, biopsies, and surgery. One-third of diagnosed breast cancers might recede on their own.[179] Screening mammography efficiently finds non-life-threatening, asymptomatic breast cancers and pre-cancers, even while overlooking serious cancers. According to H. Gilbert Welch of the Dartmouth Institute for Health Policy and Clinical Practice, research on screening mammography has taken the "brain-dead approach that says the best test is the one that finds the most cancers" rather than the one that finds dangerous cancers.[179]
Pregnancy
Breast cancers occur during pregnancy at the same rate as breast cancers in non-pregnant women of the same age. Breast cancer then becomes more common in the 5 or 10 years following pregnancy but then becomes less common than among the general population.[180] These cancers are known as postpartum breast cancer and have worse outcomes including an increased risk of distant spread of disease and mortality.[181] Other cancers found during or shortly after pregnancy appear at approximately the same rate as other cancers in women of a similar age.[182]
Diagnosing new cancer in a pregnant woman is difficult, in part because any symptoms are commonly assumed to be a normal discomfort associated with pregnancy.[182] As a result, cancer is typically discovered at a somewhat later stage than average in many pregnant or recently pregnant women. Some imaging procedures, such as MRIs (magnetic resonance imaging), CT scans, ultrasounds, and mammograms with fetal shielding are considered safe during pregnancy; some others, such as PET scans are not.[182]
Treatment is generally the same as for non-pregnant women.[182] However, radiation is normally avoided during pregnancy, especially if the fetal dose might exceed 100 cGy. In some cases, some or all treatments are postponed until after birth if the cancer is diagnosed late in the pregnancy. Early deliveries to speed the start of treatment are not uncommon. Surgery is generally considered safe during pregnancy, but some other treatments, especially certain chemotherapy drugs given during the first trimester, increase the risk of birth defects and pregnancy loss (spontaneous abortions and stillbirths).[182] Elective abortions are not required and do not improve the likelihood of the mother surviving or being cured.[182]
Radiation treatments may interfere with the mother's ability to breastfeed her baby because it reduces the ability of that breast to produce milk and increases the risk of mastitis. Also, when chemotherapy is being given after birth, many of the drugs pass through breast milk to the baby, which could harm the baby.[182]
Regarding future pregnancy among breast cancer survivors, there is often fear of cancer recurrence.[183] On the other hand, many still regard pregnancy and parenthood to represent normalcy, happiness and life fulfillment.[183]
Hormones
Birth control
In breast cancer survivors, non-hormonal birth control methods should be used as first-line options. Progestogen-based methods such as depot medroxyprogesterone acetate, IUD with progestogen or progestogen only pills have a poorly investigated but possible increased risk of cancer recurrence, but may be used if positive effects outweigh this possible risk.[184]
Menopausal hormone replacement
In breast cancer survivors, it is recommended to first consider non-hormonal options for menopausal effects, such as bisphosphonates or selective estrogen receptor modulators (SERMs) for osteoporosis, and vaginal estrogen for local symptoms. Observational studies of systemic hormone replacement therapy after breast cancer are generally reassuring. If hormone replacement is necessary after breast cancer, estrogen-only therapy or estrogen therapy with an intrauterine device with progestogen may be safer options than combined systemic therapy.[185]
Research
Treatments are being evaluated in trials. This includes individual drugs, combinations of drugs, and surgical and radiation techniques Investigations include new types of targeted therapy,[186] cancer vaccines, oncolytic virotherapy,[187] and immunotherapy.[188]
The latest research is reported annually at scientific meetings such as that of the American Society of Clinical Oncology, San Antonio Breast Cancer Symposium,[189] and the St. Gallen Oncology Conference in St. Gallen, Switzerland.[190] These studies are reviewed by professional societies and other organizations, and formulated into guidelines for specific treatment groups and risk category.
Fenretinide, a retinoid, is also being studied as a way to reduce the risk of breast cancer (retinoids are drugs related to vitamin A).[191][192]
Cryoablation
As of 2014 cryoablation is being studied to see if it could be a substitute for a lumpectomy in small cancers.[193] There is tentative evidence in those with tumors less than 2 centimeters.[194] It may also be used in those in who surgery is not possible.[194] Another review states that cryoablation looks promising for early breast cancer of small size.[195]
Breast cancer cell lines
A considerable part of the current knowledge on breast carcinomas is based on in vivo and in vitro studies performed with cell lines derived from breast cancers. These provide an unlimited source of homogenous self-replicating material, free of contaminating stromal cells, and often easily cultured in simple standard media. The first breast cancer cell line described, BT-20, was established in 1958. Since then, and despite sustained work in this area, the number of permanent lines obtained has been strikingly low (about 100). Indeed, attempts to culture breast cancer cell lines from primary tumors have been largely unsuccessful. This poor efficiency was often due to technical difficulties associated with the extraction of viable tumor cells from their surrounding stroma. Most of the available breast cancer cell lines issued from metastatic tumors, mainly from pleural effusions. Effusions provided generally large numbers of dissociated, viable tumor cells with little or no contamination by fibroblasts and other tumor stroma cells. Many of the currently used BCC lines were established in the late 1970s. A very few of them, namely MCF-7, T-47D, and MDA-MB-231, account for more than two-thirds of all abstracts reporting studies on mentioned breast cancer cell lines, as concluded from a Medline-based survey.
Molecular markers
Transcription factors
NFAT transcription factors are implicated in breast cancer, more specifically in the process of cell motility at the basis of metastasis formation. Indeed, NFAT1 (NFATC2) and NFAT5 are pro-invasive and pro-migratory in breast carcinoma[196][197] and NFAT3 (NFATc4) is an inhibitor of cell motility.[198] NFAT1 regulates the expression of the TWEAKR and its ligand TWEAK with the Lipocalin 2 to increase breast cancer cell invasion[199] and NFAT3 inhibits Lipocalin 2 expression to blunt the cell invasion.[198]
Metabolic markers
Clinically, the most useful metabolic markers in breast cancer are the estrogen and progesterone receptors that are used to predict response to hormone therapy. New or potentially new markers for breast cancer include BRCA1 and BRCA2[200] to identify patients at high risk of developing breast cancer, HER-2[201] and SCD1[202] for predicting response to therapeutic regimens, and urokinase plasminogen activator, PA1-1[203] and SCD1[204] for assessing prognosis.
Other animals
- Mammary tumor for breast cancer in other animals
- Mouse models of breast cancer metastasis
References
- 1 2 3 4 5 6 7 8 9 "Breast Cancer Treatment (PDQ®)". NCI. 23 May 2014. Retrieved 29 June 2014.
- 1 2 3 4 5 6 7 World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 5.2. ISBN 92-832-0429-8.
- ↑ "Klinefelter Syndrome". Eunice Kennedy Shriver National Institute of Child Health and Human Development. 2007-05-24. Archived from the original on November 27, 2012.
- 1 2 "SEER Stat Fact Sheets: Breast Cancer". NCI. Retrieved 18 June 2014.
- 1 2 "Cancer Survival in England: Patients Diagnosed 2007–2011 and Followed up to 2012" (PDF). Office for National Statistics. 29 October 2013. Retrieved 29 June 2014.
- ↑ GBD 2015 Disease and Injury Incidence and Prevalence, Collaborators. (8 October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015.". Lancet. 388 (10053): 1545–1602. PMC 5055577
 . PMID 27733282. doi:10.1016/S0140-6736(16)31678-6.
. PMID 27733282. doi:10.1016/S0140-6736(16)31678-6. - ↑ GBD 2015 Mortality and Causes of Death, Collaborators. (8 October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: a systematic analysis for the Global Burden of Disease Study 2015.". Lancet. 388 (10053): 1459–1544. PMC 5388903
 . PMID 27733281. doi:10.1016/s0140-6736(16)31012-1.
. PMID 27733281. doi:10.1016/s0140-6736(16)31012-1. - ↑ "Breast Cancer". NCI. Retrieved 29 June 2014.
- ↑ Saunders, Christobel; Jassal, Sunil (2009). Breast cancer (1. ed.). Oxford: Oxford University Press. p. Chapter 13. ISBN 978-0-19-955869-8.
- 1 2 3 4 5 Gøtzsche PC, Jørgensen KJ (4 June 2013). "Screening for breast cancer with mammography.". The Cochrane database of systematic reviews. 6: CD001877. PMID 23737396. doi:10.1002/14651858.CD001877.pub5.
- ↑ Nelson, HD; Tyne, K; Naik, A; Bougatsos, C; Chan, B; Nygren, P; Humphrey, L (November 2009). "Screening for Breast Cancer: Systematic Evidence Review Update for the US Preventive Services Task Force [Internet].". PMID 20722173.
- 1 2 Siu, Albert L. (12 January 2016). "Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement". Annals of Internal Medicine. 164: 279–96. PMID 26757170. doi:10.7326/M15-2886.
- ↑ American College of Surgeons (September 2013), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation, American College of Surgeons, retrieved 2 January 2013
- 1 2 3 "Breast Cancer Treatment (PDQ®)". NCI. 26 June 2014. Retrieved 29 June 2014.
- 1 2 "World Cancer Report" (PDF). International Agency for Research on Cancer. 2008. Retrieved 26 February 2011.
- 1 2 World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 1.1. ISBN 92-832-0429-8.
- ↑ "Male Breast Cancer Treatment". National Cancer Institute. 2014. Retrieved 29 June 2014.
- 1 2 3 4 Merck Manual of Diagnosis and Therapy (February 2003). "Breast Disorders: Breast Cancer". Retrieved 5 February 2008.
- 1 2 American Cancer Society (2007). "Cancer Facts & Figures 2007" (PDF). Archived from the original (PDF) on 10 April 2007. Retrieved 26 April 2007.
- ↑ Watson M (2008). "Assessment of suspected cancer". InnoAiT. 1 (2): 94–107. doi:10.1093/innovait/inn001.
- ↑ eMedicine (23 August 2006). "Breast Cancer Evaluation". Retrieved 5 February 2008.
- ↑ National Cancer Institute (27 June 2005). "Paget's Disease of the Nipple: Questions and Answers". Retrieved 6 February 2008.
- ↑ answers.com. "Oncology Encyclopedia: Cystosarcoma Phyllodes". Retrieved 10 August 2010.
- ↑ Lacroix M (December 2006). "Significance, detection and markers of disseminated breast cancer cells". Endocrine-Related Cancer. Bioscientifica. 13 (4): 1033–67. PMID 17158753. doi:10.1677/ERC-06-0001.
- ↑ National Cancer Institute (1 September 2004). "Metastatic Cancer: Questions and Answers". Retrieved 6 February 2008.
- ↑ Interpreting Signs and Symptoms. Lippincott Williams & Wilkins. 2007. pp. 99–. ISBN 978-1-58255-668-0.
- ↑ Merck Manual of Diagnosis and Therapy (February 2003). "Breast Disorders: Overview of Breast Disorders". Retrieved 5 February 2008.
- 1 2 3 Hayes,, James; Ricahrdson, Ann; Frampton, Chris (15 November 2013). "Population attributable risks for modifiable lifestyle factors and breast cancer in New Zealand women". IMJ. 43 (11): 1198–1204. PMID 23910051. doi:10.1111/imj.12256.
- ↑ Reeder JG, Vogel VG (2008). "Breast cancer prevention.". Cancer treatment and research. 141: 149–64. PMID 18274088. doi:10.1007/978-0-387-73161-2_10.
- ↑ "Am I at risk?". Breast Cancer Care. Retrieved 22 October 2013.
- ↑ Collaborative Group on Hormonal Factors in Breast Cancer (August 2002). "Breast cancer and breastfeeding: collaborative reanalysis of individual data from 47 epidemiological studies in 30 countries, including 50302 women with breast cancer and 96973 women without the disease.". Lancet. 360 (9328): 187–95. PMID 12133652. doi:10.1016/S0140-6736(02)09454-0.
- ↑ Yager JD, Davidson NE (2006). "Estrogen carcinogenesis in breast cancer". New Engl J Med. 354 (3): 270–82. PMID 16421368. doi:10.1056/NEJMra050776.
- ↑ Mazzucco A, Santoro E, DeSoto, M, Hong Lee J (February 2009). "Hormone Therapy and Menopause". National Research Center for Women & Families.
- ↑ Light Pollution as new risk factor for human Breast and Prostate Cancers- Haim, Abraham; Portnov, Biris P., 2013, ISBN 978-94-007-6220-6
- 1 2 Johnson KC, Miller AB, Collishaw NE, Palmer JR, Hammond SK, Salmon AG, Cantor KP, Miller MD, Boyd NF, Millar J, Turcotte F (Jan 2011). "Active smoking and secondhand smoke increase breast cancer risk: the report of the Canadian Expert Panel on Tobacco Smoke and Breast Cancer Risk (2009).". Tobacco control. 20 (1): e2. PMID 21148114. doi:10.1136/tc.2010.035931.
- ↑ Lee IM, Shiroma EJ, Lobelo F, Puska P, Blair SN, Katzmarzyk PT (1 July 2012). "Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy". The Lancet. 380 (9838): 219–29. PMC 3645500
 . PMID 22818936. doi:10.1016/S0140-6736(12)61031-9.
. PMID 22818936. doi:10.1016/S0140-6736(12)61031-9. - ↑ Biswas A, Oh PI, Faulkner GE, Bajaj RR, Silver MA, Mitchell MS, Alter DA (2015). "Sedentary Time and Its Association With Risk for Disease Incidence, Mortality, and Hospitalization in Adults: A Systematic Review and Meta-analysis". Annals of Internal Medicine. 162 (2): 123–32. PMID 25599350. doi:10.7326/M14-1651.
- ↑ Kahlenborn C, Modugno F, Potter DM, Severs WB (Oct 2006). "Oral contraceptive use as a risk factor for premenopausal breast cancer: a meta-analysis.". Mayo Clinic proceedings. Mayo Clinic. 81 (10): 1290–302. PMID 17036554. doi:10.4065/81.10.1290.
- 1 2 Veljković M, Veljković S (Sep 2010). "[The risk of breast cervical, endometrial and ovarian cancer in oral contraceptive users].". Medicinski pregled. 63 (9–10): 657–61. PMID 21446095. doi:10.2298/mpns1010657v.
- 1 2 Casey PM, Cerhan JR, Pruthi S (January 2008). "Oral contraceptive use and risk of breast cancer.". Mayo Clinic proceedings. Mayo Clinic. 83 (1): 86–90; quiz 90–1. PMID 18174010. doi:10.4065/83.1.86.
- ↑ Iodice S, Barile M, Rotmensz N, Feroce I, Bonanni B, Radice P, Bernard L, Maisonneuve P, Gandini S (August 2010). "Oral contraceptive use and breast or ovarian cancer risk in BRCA1/2 carriers: a meta-analysis.". European journal of cancer (Oxford, England : 1990). 46 (12): 2275–84. PMID 20537530. doi:10.1016/j.ejca.2010.04.018.
- ↑ Gaffield ME, Culwell KR, Ravi A (October 2009). "Oral contraceptives and family history of breast cancer.". Contraception. 80 (4): 372–80. PMID 19751860. doi:10.1016/j.contraception.2009.04.010.
- ↑ Yang L, Jacobsen KH (December 2008). "A systematic review of the association between breastfeeding and breast cancer.". Journal of Women's Health. 17 (10): 1635–45. PMID 19049358. doi:10.1089/jwh.2008.0917.
- ↑ Russo J, Russo IH (1980). "Susceptibility of the mammary gland to carcinogenesis. II. Pregnancy interruption as a risk factor in tumor incidence". Am J Pathol. 100 (2): 505–506. PMC 1903536
 . PMID 6773421.
. PMID 6773421. In contrast, abortion is associated with increased risk of carcinomas of the breast. The explanation for these epidemiologic findings is not known, but the parallelism between the DMBA-induced rat mammary carcinoma model and the human situation is striking. ... Abortion would interrupt this process, leaving in the gland undifferentiated structures like those observed in the rat mammary gland, which could render the gland again susceptible to carcinogenesis.
- ↑ Beral V, Bull D, Doll R, Peto R, Reeves G (27 March 2004). "Breast cancer and abortion: collaborative reanalysis of data from 53 epidemiological studies, including 83?000 women with breast cancer from 16 countries.". Lancet. 363 (9414): 1007–16. PMID 15051280. doi:10.1016/S0140-6736(04)15835-2.
- ↑ Blackburn GL, Wang KA (September 2007). "Dietary fat reduction and breast cancer outcome: results from the Women's Intervention Nutrition Study (WINS).". The American Journal of Clinical Nutrition. 86 (3): s878–81. PMID 18265482.
- ↑ Boffetta P, Hashibe M, La Vecchia C, Zatonski W, Rehm J (August 2006). "The burden of cancer attributable to alcohol drinking". International Journal of Cancer. 119 (4): 884–7. PMID 16557583. doi:10.1002/ijc.21903.
- ↑ BBC report Weight link to breast cancer risk
- ↑ Kaiser J (29 November 2013). "Cancer. Cholesterol forges link between obesity and breast cancer.". Science. 342 (6162): 1028. PMID 24288308. doi:10.1126/science.342.6162.1028.
- ↑ Aceves C, Anguiano B, Delgado G (April 2005). "Is iodine a gatekeeper of the integrity of the mammary gland?". Journal of Mammary Gland Biology and Neoplasia. 10 (2): 189–96. PMID 16025225. doi:10.1007/s10911-005-5401-5.
- ↑ Mourouti, N; Kontogianni, MD; Papavagelis, C; Panagiotakos, DB (February 2015). "Diet and breast cancer: a systematic review.". International journal of food sciences and nutrition. 66 (1): 1–42. PMID 25198160. doi:10.3109/09637486.2014.950207.
- ↑ Aubrey, Allison (1 February 2016). "A Diet High In Fiber May Help Protect Against Breast Cancer". NPR. Retrieved 1 February 2016.
- 1 2 American Cancer Society (2005). "Breast Cancer Facts & Figures 2005–2006" (PDF). Archived from the original (PDF) on 13 June 2007. Retrieved 26 April 2007.
- ↑ Wang XS, Armstrong ME, Cairns BJ, Key TJ, Travis RC (March 2011). "Shift work and chronic disease: the epidemiological evidence.". Occupational Medicine. 61 (2): 78–89. PMC 3045028
 . PMID 21355031. doi:10.1093/occmed/kqr001.
. PMID 21355031. doi:10.1093/occmed/kqr001. - ↑ Brody JG, Rudel RA, Michels KB, Moysich KB, Bernstein L, Attfield KR, Gray S (June 2007). "Environmental pollutants, diet, physical activity, body size, and breast cancer: where do we stand in research to identify opportunities for prevention?". Cancer. 109 (12 Suppl): 2627–34. PMID 17503444. doi:10.1002/cncr.22656.
- ↑ Hendrick RE (October 2010). "Radiation doses and cancer risks from breast imaging studies.". Radiology. 257 (1): 246–53. PMID 20736332. doi:10.1148/radiol.10100570.
- 1 2 3 Boris Pasche (2010). Cancer Genetics (Cancer Treatment and Research). Berlin: Springer. pp. 19–20. ISBN 1-4419-6032-5.
- 1 2 3 Gage M, Wattendorf D, Henry LR (1 April 2012). "Translational advances regarding hereditary breast cancer syndromes". Journal of surgical oncology. 105 (5): 444–51. PMID 22441895. doi:10.1002/jso.21856.
- ↑ Colditz, Graham A.; Kaphingst, Kimberly A.; Hankinson, Susan E.; Rosner, Bernard (19 February 2012). "Family history and risk of breast cancer: nurses' health study". Breast Cancer Research and Treatment. 133 (3): 1097–1104. ISSN 0167-6806. PMC 3387322
 . PMID 22350789. doi:10.1007/s10549-012-1985-9.
. PMID 22350789. doi:10.1007/s10549-012-1985-9. - ↑ "Familial breast cancer: collaborative reanalysis of individual data from 52 epidemiological studies including 58,209 women with breast cancer and 101,986 women without the disease.". Lancet. 358 (9291): 1389–99. 27 October 2001. PMID 11705483. doi:10.1016/S0140-6736(01)06524-2.
- ↑ Nelson HD, Zakher B, Cantor A, Fu R, Griffin J, O'Meara ES, Buist DS, Kerlikowske K, van Ravesteyn NT, Trentham-Dietz A, Mandelblatt JS, Miglioretti DL (1 May 2012). "Risk factors for breast cancer for women aged 40 to 49 years: a systematic review and meta-analysis.". Annals of Internal Medicine. 156 (9): 635–48. PMC 3561467
 . PMID 22547473. doi:10.7326/0003-4819-156-9-201205010-00006.
. PMID 22547473. doi:10.7326/0003-4819-156-9-201205010-00006. - ↑ Kolata, Gina (23 September 2012). "Genetic Study Finds 4 Distinct Variations of Breast Cancer". The New York Times. Retrieved 23 September 2012.
- ↑ "Understanding Breast Changes – National Cancer Institute". Archived from the original on 27 May 2010.
- ↑ "Lobular carcinoma in situ: Marker for breast cancer risk". MayoClinic.com.
- ↑ "Breast Cancer Treatment". National Cancer Institute.
- ↑ Afonso N, Bouwman D (August 2008). "Lobular carcinoma in situ". Eur. J. Cancer Prev. 17 (4): 312–6. PMID 18562954. doi:10.1097/CEJ.0b013e3282f75e5d.
- ↑ Anothaisintawee T, Wiratkapun C, Lerdsitthichai P, Kasamesup V, Wongwaisayawan S, Srinakarin J, Hirunpat S, Woodtichartpreecha P, Boonlikit S, Teerawattananon Y, Thakkinstian A (2013). "Risk factors of breast cancer: a systematic review and meta-analysis". Asian Pacific Journal of Public Health. 25 (5): 368–387. PMID 23709491. doi:10.1177/1010539513488795.
- ↑ Böhm I (2011). "Breast cancer in lupus". Breast. 20 (3): 288–90. PMID 21237645. doi:10.1016/j.breast.2010.12.005.
- ↑ Adrian Lee; Carlos Arteaga (14 December 2009). "32nd Annual CTRC-AACR San Antonio Breast Cancer Symposium" (PDF). Sunday Morning Year-End Review. Archived from the original (PDF) on 13 August 2013.
- ↑ Cavalieri E, Chakravarti D, Guttenplan J, Hart E, Ingle J, Jankowiak R, Muti P, Rogan E, Russo J, Santen R, Sutter T (August 2006). "Catechol estrogen quinones as initiators of breast and other human cancers: implications for biomarkers of susceptibility and cancer prevention". Biochimica et Biophysica Acta. 1766 (1): 63–78. PMID 16675129. doi:10.1016/j.bbcan.2006.03.001.
- ↑ Haslam SZ, Woodward TL (June 2003). "Host microenvironment in breast cancer development: epithelial-cell-stromal-cell interactions and steroid hormone action in normal and cancerous mammary gland.". Breast Cancer Res. 5 (4): 208–15. PMC 165024
 . PMID 12817994. doi:10.1186/bcr615.
. PMID 12817994. doi:10.1186/bcr615. - ↑ Wiseman BS, Werb Z (May 2002). "Stromal effects on mammary gland development and breast cancer". Science. 296 (5570): 1046–9. PMC 2788989
 . PMID 12004111. doi:10.1126/science.1067431.
. PMID 12004111. doi:10.1126/science.1067431. - ↑ Jardé T, Perrier S, Vasson MP, Caldefie-Chézet F (January 2011). "Molecular mechanisms of leptin and adiponectin in breast cancer". Eur. J. Cancer. 47 (1): 33–43. PMID 20889333. doi:10.1016/j.ejca.2010.09.005.
- ↑ Dunning AM, Healey CS, Pharoah PD, Teare MD, Ponder BA, Easton DF (October 1999). "A systematic review of genetic polymorphisms and breast cancer risk". Cancer Epidemiology, Biomarkers & Prevention. 8 (10): 843–54. PMID 10548311.
- ↑ Begg CB, Haile RW, Borg A, Malone KE, Concannon P, Thomas DC, Langholz B, Bernstein L, Olsen JH, Lynch CF, Anton-Culver H, Capanu M, Liang X, Hummer AJ, Sima C, Bernstein JL (January 2008). "Variation of breast cancer risk among BRCA1/2 carriers". JAMA. 299 (2): 194–201. PMC 2714486
 . PMID 18182601. doi:10.1001/jama.2007.55-a.
. PMID 18182601. doi:10.1001/jama.2007.55-a. - ↑ Patel KJ, Yu VP, Lee H, Corcoran A, Thistlethwaite FC, Evans MJ, Colledge WH, Friedman LS, Ponder BA, Venkitaraman AR (February 1998). "Involvement of Brca2 in DNA repair". Mol. Cell. 1 (3): 347–57. PMID 9660919. doi:10.1016/S1097-2765(00)80035-0.
- ↑ Marietta C, Thompson LH, Lamerdin JE, Brooks PJ (May 2009). "Acetaldehyde stimulates FANCD2 monoubiquitination, H2AX phosphorylation, and BRCA1 phosphorylation in human cells in vitro: implications for alcohol-related carcinogenesis". Mutat. Res. 664 (1–2): 77–83. PMC 2807731
 . PMID 19428384. doi:10.1016/j.mrfmmm.2009.03.011.
. PMID 19428384. doi:10.1016/j.mrfmmm.2009.03.011. - ↑ Theruvathu JA, Jaruga P, Nath RG, Dizdaroglu M, Brooks PJ (2005). "Polyamines stimulate the formation of mutagenic 1,N2-propanodeoxyguanosine adducts from acetaldehyde". Nucleic Acids Res. 33 (11): 3513–20. PMC 1156964
 . PMID 15972793. doi:10.1093/nar/gki661.
. PMID 15972793. doi:10.1093/nar/gki661. - ↑ Wooster R, Weber BL (June 2003). "Breast and ovarian cancer". N. Engl. J. Med. 348 (23): 2339–47. PMID 12788999. doi:10.1056/NEJMra012284.
- ↑ Levin B, Lech D, Friedenson B (2012). "Evidence that BRCA1- or BRCA2-associated cancers are not inevitable". Mol. Med. 18: 1327–37. PMC 3521784
 . PMID 22972572. doi:10.2119/molmed.2012.00280.
. PMID 22972572. doi:10.2119/molmed.2012.00280. - ↑ Kouros-Mehr H, Kim JW, Bechis SK, Werb Z (Apr 2008). "GATA-3 and the regulation of the mammary luminal cell fate.". Current Opinion in Cell Biology. 20 (2): 164–70. PMC 2397451
 . PMID 18358709. doi:10.1016/j.ceb.2008.02.003.
. PMID 18358709. doi:10.1016/j.ceb.2008.02.003. - ↑ Saslow D, Hannan J, Osuch J, Alciati MH, Baines C, Barton M, Bobo JK, Coleman C, Dolan M, Gaumer G, Kopans D, Kutner S, Lane DS, Lawson H, Meissner H, Moorman C, Pennypacker H, Pierce P, Sciandra E, Smith R, Coates R (2004). "Clinical breast examination: practical recommendations for optimizing performance and reporting". CA: A Cancer Journal for Clinicians. 54 (6): 327–344. PMID 15537576. doi:10.3322/canjclin.54.6.327.
- ↑ Yu YH, Liang C, Yuan XZ (2010). "Diagnostic value of vacuum-assisted breast biopsy for breast carcinoma: a meta-analysis and systematic review.". Breast cancer research and treatment. 120 (2): 469–79. PMID 20130983. doi:10.1007/s10549-010-0750-1.
- ↑ Merck Manual, Professional Edition, Ch. 253, Breast Cancer.
- ↑ American Society of Clinical Oncology, "Five Things Physicians and Patients Should Question" (PDF), Choosing Wisely: an initiative of the ABIM Foundation, American Society of Clinical Oncology, archived from the original (PDF) on 31 July 2012, retrieved 14 August 2012
- ↑ Carlson RW, Allred DC, Anderson BO, Burstein HJ, Carter WB, Edge SB, Erban JK, Farrar WB, Goldstein LJ, Gradishar WJ, Hayes DF, Hudis CA, Jahanzeb M, Kiel K, Ljung BM, Marcom PK, Mayer IA, McCormick B, Nabell LM, Pierce LJ, Reed EC, Smith ML, Somlo G, Theriault RL, Topham NS, Ward JH, Winer EP, Wolff AC (2009). "Breast cancer. Clinical practice guidelines in oncology". Journal of the National Comprehensive Cancer Network : JNCCN. 7 (2): 122–192. PMID 19200416.
- ↑ Kumar, Vinay; Abul Abbas (2010). Robbins and Cotran Pathologic Basis of Disease. Philadelphia: Saunders, an imprint of Elsevier inc. p. 1090. ISBN 978-1-4160-3121-5.
- ↑ Sotiriou C, Pusztai L (February 2009). "Gene-expression signatures in breast cancer". N. Engl. J. Med. 360 (8): 790–800. PMID 19228622. doi:10.1056/NEJMra0801289.
- ↑ Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N (October 2005). "Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer". N. Engl. J. Med. 353 (16): 1673–84. PMID 16236738. doi:10.1056/NEJMoa052122.
- 1 2 3 American Institute for Cancer Research/ World Cancer Research Fund, Food, Nutrition, Physical Activity and the Prevention of Cancer: a Global Perspective
- ↑ Eliassen AH, Hankinson SE, Rosner B, Holmes MD, Willett WC (October 2010). "Physical activity and risk of breast cancer among postmenopausal women". Arch. Intern. Med. 170 (19): 1758–64. PMC 3142573
 . PMID 20975025. doi:10.1001/archinternmed.2010.363.
. PMID 20975025. doi:10.1001/archinternmed.2010.363. - ↑ Kyu, Hmwe H; Bachman, Victoria F; Alexander, Lily T; Mumford, John Everett; Afshin, Ashkan; Estep, Kara; Veerman, J Lennert; Delwiche, Kristen; Iannarone, Marissa L; Moyer, Madeline L; Cercy, Kelly; Vos, Theo; Murray, Christopher J L; Forouzanfar, Mohammad H (9 August 2016). "Physical activity and risk of breast cancer, colon cancer, diabetes, ischemic heart disease, and ischemic stroke events: systematic review and dose-response meta-analysis for the Global Burden of Disease Study 2013". BMJ. 354: i3857. PMC 4979358
 . PMID 27510511. doi:10.1136/bmj.i3857.
. PMID 27510511. doi:10.1136/bmj.i3857. - ↑ Song, Jung-Kook; Bae, Jong-Myon (2013-03-01). "Citrus fruit intake and breast cancer risk: a quantitative systematic review". Journal of Breast Cancer. 16 (1): 72–76. ISSN 1738-6756. PMC 3625773
 . PMID 23593085. doi:10.4048/jbc.2013.16.1.72.
. PMID 23593085. doi:10.4048/jbc.2013.16.1.72. - ↑ Zheng JS, Hu XJ, Zhao YM, Yang J, Li D (2013). "Intake of fish and marine n-3 polyunsaturated fatty acids and risk of breast cancer: meta-analysis of data from 21 independent prospective cohort studies". BMJ. 346: f3706. PMID 23814120. doi:10.1136/bmj.f3706.
- ↑ Wu, AH; Yu, MC; Tseng, CC; Pike, MC (15 January 2008). "Epidemiology of soy exposures and breast cancer risk.". British Journal of Cancer. 98 (1): 9–14. PMC 2359677
 . PMID 18182974. doi:10.1038/sj.bjc.6604145.
. PMID 18182974. doi:10.1038/sj.bjc.6604145. - ↑ Hartmann LC, Schaid DJ, Woods JE, Crotty TP, Myers JL, Arnold PG, Petty PM, Sellers TA, Johnson JL, McDonnell SK, Frost MH, Jenkins RB (1999). "Efficacy of bilateral prophylactic mastectomy in women with a family history of breast cancer". N Engl J Med. 340 (2): 77–84. PMID 9887158. doi:10.1056/NEJM199901143400201.
- ↑ Meijers-Heijboer H, van Geel B, van Putten WL, Henzen-Logmans SC, Seynaeve C, Menke-Pluymers MB, Bartels CC, Verhoog LC, van den Ouweland AM, Niermeijer MF, Brekelmans CT, Klijn JG (2001). "Breast cancer after prophylactic bilateral mastectomy in women with BRCA1 and BRCA2 mutations". N Engl J Med. 345 (3): 159–164. PMID 11463009. doi:10.1056/NEJM200107193450301.
- 1 2 Lostumbo, L; Carbine, NE; Wallace, J (10 November 2010). "Prophylactic mastectomy for the prevention of breast cancer.". The Cochrane database of systematic reviews (11): CD002748. PMID 21069671. doi:10.1002/14651858.CD002748.pub3.
- ↑ "Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer in Women: U.S. Preventive Services Task Force Recommendation Statement". Annals of Internal Medicine. 160: 271–281. 24 December 2013. doi:10.7326/M13-2747.
- 1 2 3 Nelson HD, Smith ME, Griffin JC, Fu R (16 April 2013). "Use of medications to reduce risk for primary breast cancer: a systematic review for the U.S. Preventive Services Task Force.". Annals of Internal Medicine. 158 (8): 604–14. PMID 23588749. doi:10.7326/0003-4819-158-8-201304160-00005.
- ↑ Cuzick J, Sestak I, Bonanni B, Costantino JP, Cummings S, DeCensi A, Dowsett M, Forbes JF, Ford L, LaCroix AZ, Mershon J, Mitlak BH, Powles T, Veronesi U, Vogel V, Wickerham DL (25 May 2013). "Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data.". Lancet. 381 (9880): 1827–34. PMC 3671272
 . PMID 23639488. doi:10.1016/S0140-6736(13)60140-3.
. PMID 23639488. doi:10.1016/S0140-6736(13)60140-3. - ↑ Moyer VA (24 September 2013). "Medications for Risk Reduction of Primary Breast Cancer in Women: U.S. Preventive Services Task Force Recommendation Statement.". Annals of Internal Medicine. 159: 698–708. PMID 24061412. doi:10.7326/0003-4819-159-10-201311190-00718.
- ↑ Cuzick J, Sestak I, Bonanni B, Costantino JP, Cummings S, DeCensi A, Dowsett M, Forbes JF, Ford L, LaCroix AZ, Mershon J, Mitlak BH, Powles T, Veronesi U, Vogel V, Wickerham DL (31 March 2013). "Selective oestrogen receptor modulators in prevention of breast cancer: an updated meta-analysis of individual participant data". The Lancet. 381 (9880): 1827–34. PMC 3671272
 . PMID 23639488. doi:10.1016/S0140-6736(13)60140-3.
. PMID 23639488. doi:10.1016/S0140-6736(13)60140-3. - ↑ "Screening". Centers for Disease Control and Prevention. Retrieved 17 November 2015.
- ↑ "Screening for Breast Cancer". US Preventative Services Task Force. December 2009. Retrieved 24 December 2012.
- ↑ Kösters JP, Gøtzsche PC (2003). "Cochrane Database of Systematic Reviews". Cochrane Database Syst Rev (2): CD003373. PMID 12804462. doi:10.1002/14651858.CD003373.
- ↑ "Breast Cancer and Mammograms". WebMD. Retrieved 24 December 2012.
- ↑ Biesheuvel C, Weigel S, Heindel W (2011). "Mammography Screening: Evidence, History and Current Practice in Germany and Other European Countries.". Breast care (Basel, Switzerland). 6 (2): 104–109. PMC 3104900
 . PMID 21673820. doi:10.1159/000327493.
. PMID 21673820. doi:10.1159/000327493. - ↑ Tonelli M, Connor Gorber S, Joffres M, Dickinson J, Singh H, Lewin G, Birtwhistle R, Fitzpatrick-Lewis D, Hodgson N, Ciliska D, Gauld M, Liu YY (22 November 2011). "Recommendations on screening for breast cancer in average-risk women aged 40–74 years.". Canadian Medical Association Journal. 183 (17): 1991–2001. PMC 3225421
 . PMID 22106103. doi:10.1503/cmaj.110334.
. PMID 22106103. doi:10.1503/cmaj.110334. - ↑ "Breast Cancer: Screening". United States Preventive Services Task Force. Archived from the original on 16 June 2013.
- ↑ Welch HG, Passow HJ (30 December 2013). "Quantifying the Benefits and Harms of Screening Mammography.". JAMA internal medicine. 174 (3): 448–54. PMID 24380095. doi:10.1001/jamainternmed.2013.13635.
- ↑ Gøtzsche PC, Nielsen M (2011). "Screening for breast cancer with mammography". Cochrane Database Syst Rev (1): CD001877. PMID 21249649. doi:10.1002/14651858.CD001877.pub4.
- ↑ "Screening for breast cancer with mammography". Cochrane Nordic. 27 August 2015. Retrieved 15 October 2015.
- ↑ "Screening for breast cancer: U.S. Preventive Services Task Force recommendation statement.". Annals of Internal Medicine. 151 (10): 716–26, W–236. 17 November 2009. PMID 19920272. doi:10.7326/0003-4819-151-10-200911170-00008.
- ↑ Saini KS, Taylor C, Ramirez AJ, Palmieri C, Gunnarsson U, Schmoll HJ, Dolci SM, Ghenne C, Metzger-Filho O, Skrzypski M, Paesmans M, Ameye L, Piccart-Gebhart MJ, de Azambuja E (August 2011). "Role of the multidisciplinary team in breast cancer management: results from a large international survey involving 39 countries". Annals of Oncology. 23 (4): 853–9. PMID 21821551. doi:10.1093/annonc/mdr352.
- ↑ Holmes MD, Chen WY, Li L, Hertzmark E, Spiegelman D, Hankinson SE (2010). "Aspirin Intake and Survival After Breast Cancer". Journal of Clinical Oncology. 28 (9): 1467–72. PMC 2849768
 . PMID 20159825. doi:10.1200/JCO.2009.22.7918.
. PMID 20159825. doi:10.1200/JCO.2009.22.7918. - ↑ Ting Bao; Michelle A Rudek (2011). "The Clinical Pharmacology of Anastrozole". European Oncology & Haematology. 7 (2): 106–8.
- ↑ Burstein, HJ; Temin, S; Anderson, H; Buchholz, TA; Davidson, NE; Gelmon, KE; Giordano, SH; Hudis, CA; Rowden, D; Solky, AJ; Stearns, V; Winer, EP; Griggs, JJ (27 May 2014). "Adjuvant Endocrine Therapy for Women With Hormone Receptor-Positive Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline Focused Update.". Journal of Clinical Oncology. 32 (21): 2255–69. PMID 24868023. doi:10.1200/JCO.2013.54.2258.
- ↑ Early Breast Cancer Trialists' Collaborative Group (23 July 2015). "Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials". The Lancet. 386: 1341–1352. PMID 26211827. doi:10.1016/S0140-6736(15)61074-1.
- ↑ Petit T, Dufour P, Tannock I (June 2011). "A critical evaluation of the role of aromatase inhibitors as adjuvant therapy for postmenopausal women with breast cancer". Endocr. Relat. Cancer. 18 (3): R79–89. PMID 21502311. doi:10.1530/ERC-10-0162.
- ↑ http://www.uptodate.com/contents/treatment-of-metastatic-breast-cancer-beyond-the-basics?source=search_result&search=letrozole&selectedTitle=4%7E6
- ↑ Jahanzeb M (August 2008). "Adjuvant trastuzumab therapy for HER2-positive breast cancer". Clin. Breast Cancer. 8 (4): 324–33. PMID 18757259. doi:10.3816/CBC.2008.n.037.
- ↑ "Entrez Gene: ERBB2 v-erb-b2 erythroblastic leukemia viral oncogene homolog 2, neuro/glioblastoma derived oncogene homolog (avian)". Retrieved 17 November 2015.
- ↑ "Herceptin (trastuzumab) Adjuvant HER2+ Breast Cancer Therapy Pivotal Studies and Efficacy Data". Herceptin.com. Retrieved 8 May 2010.
- ↑ Massarut S, Baldassare G, Belleti B, Reccanello S, D'Andrea S, Ezio C, Perin T, Roncadin M, Vaidya JS (2006). "Intraoperative radiotherapy impairs breast cancer cell motility induced by surgical wound fluid". J Clin Oncol. 24 (18S): 10611.
- ↑ Belletti B, Vaidya JS, D'Andrea S, Entschladen F, Roncadin M, Lovat F, Berton S, Perin T, Candiani E, Reccanello S, Veronesi A, Canzonieri V, Trovò MG, Zaenker KS, Colombatti A, Baldassarre G, Massarut S (March 2008). "Targeted intraoperative radiotherapy impairs the stimulation of breast cancer cell proliferation and invasion caused by surgical wounding". Clin. Cancer Res. 14 (5): 1325–32. PMID 18316551. doi:10.1158/1078-0432.CCR-07-4453.
- ↑ "Radiation Therapy". Breastcancer.org. Retrieved 17 November 2015.
- ↑ "Breast Cancer: Breast Disorders: Merck Manual Professional". Merck.com. Retrieved 8 May 2010.
- ↑ "Surgery Choices for Women with Early Stage Breast Cancer" (PDF). National Cancer Institute and the National Research Center for Women & Families. August 2004. Archived from the original (PDF) on 13 August 2013.
- ↑ Gonzalez-Angulo AM, Litton JK, Broglio KR, Meric-Bernstam F, Rakkhit R, Cardoso F, Peintinger F, Hanrahan EO, Sahin A, Guray M, Larsimont D, Feoli F, Stranzl H, Buchholz TA, Valero V, Theriault R, Piccart-Gebhart M, Ravdin PM, Berry DA, Hortobagyi GN (December 2009). "High risk of recurrence for patients with breast cancer who have human epidermal growth factor receptor 2-positive, node-negative tumors 1 cm or smaller". J. Clin. Oncol. 27 (34): 5700–6. PMC 2792998
 . PMID 19884543. doi:10.1200/JCO.2009.23.2025. Lay summary – ScienceDaily.
. PMID 19884543. doi:10.1200/JCO.2009.23.2025. Lay summary – ScienceDaily. - ↑ "Breast Cancer: Breast Disorders: Merck Manual Professional". Merck.com. Retrieved 14 November 2010.
- ↑ Elston CW, Ellis IO (November 1991). "Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up". Histopathology. 19 (5): 403–10. PMID 1757079. doi:10.1111/j.1365-2559.1991.tb00229.x.
- ↑ Peppercorn J (2009). "Breast Cancer in Women Under 40". Oncology. 23 (6).
- ↑ Brandt, Jasmine; Garne, Jens; Tengrup, Ingrid; Manjer, Jonas (2015). "Age at diagnosis in relation to survival following breast cancer: a cohort study". World Journal of Surgical Oncology. 13 (1): 33. ISSN 1477-7819. doi:10.1186/s12957-014-0429-x.
- ↑ Gierach GL, Ichikawa L, Kerlikowske K, Brinton LA, Farhat GN, Vacek PM, Weaver DL, Schairer C, Taplin SH, Sherman ME (August 2012). "Relationship between mammographic density and breast cancer death in the Breast Cancer Surveillance Consortium". Journal of National Cancer Institute. 104 (16): 1218–27. PMID 22911616. doi:10.1093/jnci/djs327.
- ↑ Amro Masarwah; Päivi Auvinen; Mazen Sudah; Suvi Rautiainen; Anna Sutela; Outi Pelkonen; Sanna Oikari; Veli-Matti Kosma; Ritva Vanninen; et al. (2015). "Very low mammographic breast density predicts poorer outcome in patients with invasive breast cancer". European Radiology. 25: 1875–1882. PMID 25735512. doi:10.1007/s00330-015-3626-2.
- ↑ Klein, Judith. "Differences in male breast cancer stage, tumor size at diagnosis, and survival rate between metropolitan and nonmetropolitan regions". Proquest. Retrieved 7 December 2012.
- ↑ Pritchard KI (2009). "Ovarian Suppression/Ablation in Premenopausal ER-Positive Breast Cancer Patients". Oncology. 23 (1).
- 1 2 Robb C, Haley WE, Balducci L, Extermann M, Perkins EA, Small BJ, Mortimer J (April 2007). "Impact of breast cancer survivorship on quality of life in older women". Critical Reviews in Oncology/hematology. 62 (1): 84–91. PMID 17188505. doi:10.1016/j.critrevonc.2006.11.003.
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2009. Retrieved 11 November 2009.
- 1 2 3 McGuire, A; Brown, JA; Malone, C; McLaughlin, R; Kerin, MJ (22 May 2015). "Effects of age on the detection and management of breast cancer.". Cancers. 7 (2): 908–29. PMC 4491690
 . PMID 26010605. doi:10.3390/cancers7020815.
. PMID 26010605. doi:10.3390/cancers7020815. - 1 2 3 "World Cancer Report". International Agency for Research on Cancer. 2008. Retrieved 26 February 2011. (cancer statistics often exclude non-melanoma skin cancers such as basal-cell carcinoma, which are common but rarely fatal)
- ↑ "Breast cancer: prevention and control". World Health Organization. Archived from the original on 6 September 2015.
- ↑ World Cancer Report 2014. International Agency for Research on Cancer, World Health Organization. 2014. ISBN 978-92-832-0432-9.
- ↑ Stewart B. W. and Kleihues P. (Eds): World Cancer Report. IARCPress. Lyon 2003 Archived 20 October 2008 at the Wayback Machine.
- ↑ Laurance, Jeremy (29 September 2006). "Breast cancer cases rise 80% since Seventies". The Independent. London. Archived from the original on 25 April 2008. Retrieved 9 October 2006.
- ↑ "Breast Cancer: Statistics on Incidence, Survival, and Screening". Imaginis Corporation. 2006. Retrieved 9 October 2006.
- ↑ Breast Cancer: Breast Cancer in Young Women WebMD. Retrieved 9 September 2009
- ↑ Nearly 85% of women diagnosed with breast cancer now survive for 5 year or more Office for National Statistics, 2013
- 1 2 3 Olson, James Stuart (2002). Bathsheba's breast: women, cancer & history. Baltimore: The Johns Hopkins University Press. pp. 9–13. ISBN 0-8018-6936-6.
- 1 2 "Oldest evidence of breast cancer found in Egyptian skeleton". Reuters. 24 March 2015. Retrieved 25 March 2015.
- ↑ "The History of Cancer". American Cancer Society. 25 March 2002. Retrieved 9 October 2006.
- 1 2 Olson 2002, pp. 32–33
- ↑ Yalom, Marilyn (1997). A history of the breast. New York: Alfred A. Knopf. p. 234. ISBN 0-679-43459-3.
- 1 2 3 4 Aronowitz, Robert A. (2007). Unnatural history: breast cancer and American society. Cambridge, UK: Cambridge University Press. pp. 22–24. ISBN 0-521-82249-1.
- ↑ "History of Breast Cancer". Random History. 27 February 2008. Retrieved 8 May 2010.
- 1 2 3 4 5 6 7 Olson 2002, pp. 102–6
- ↑ Olson 2002, p. 1
- ↑ Marc Lacroix (2011). A Concise History of Breast Cancer. USA: Nova Science Publishers. pp. 59–68. ISBN 978-1-61122-305-7.
- ↑ Olson 2002, pp. 26,28,229
- ↑ Alfredo Morabia (2004). A History of Epidemiologic Methods and Concepts. Boston: Birkhauser. pp. 301–302. ISBN 3-7643-6818-7. Retrieved 31 December 2007.
- 1 2 Sulik, Gayle A. (2010). Pink Ribbon Blues: How Breast Cancer Culture Undermines Women's Health. USA: Oxford University Press. pp. 200–3. ISBN 0-19-974045-3. OCLC 535493589.
- ↑ Sulik 2010, pp. 37–38
- ↑ Sulik 2010, p. 4
- ↑ Bob Riter. "History of Breast Cancer Advocacy". Cancer Resource Center of the Finger Lakes. Retrieved 29 June 2013.
- ↑ Sulik 2010, pp. 27–72
- ↑ Sulik 2010, pp. 359–361
- ↑ Sulik 2010, pp. 366–8
- ↑ Landeman, Anne (11 June 2008). "Pinkwashing: Can Shopping Cure Breast Cancer?". Center for Media and Democracy.
- ↑ Sulik 2010, pp. 365–6
- ↑ Sulik 2010, pp. 372–4
- ↑ Breast cancer month overshadowed by 'pinkwashing' 9 October 2010, Angela Mulholland, CTV.ca News
- 1 2 3 4 Ehrenreich, Barbara (November 2001). "Welcome to Cancerland". Harper's Magazine. Archived from the original on 20 November 2010.
- ↑ Sulik 2010, p. 57
- 1 2 Browne, Anthony (7 October 2001). "Cancer bias puts breasts first". The Guardian. London.
- ↑ Arnst, Catherine (13 June 2007). "A Gender Gap in Cancer". Bloomberg Businessweek. ISSN 0007-7135.
- ↑ Olson 2002, pp. 199–200
- ↑ Ave, Melanie (10 October 2006). "Tampabay: All May Not Be in the Pink". St. Petersburg Times.
- 1 2 Aschwanden, Christie (17 August 2009). "The Trouble with Mammograms". The Los Angeles Times.
- ↑ Azim HA, Jr; Santoro, L; Russell-Edu, W; Pentheroudakis, G; Pavlidis, N; Peccatori, FA (November 2012). "Prognosis of pregnancy-associated breast cancer: a meta-analysis of 30 studies.". Cancer treatment reviews. 38 (7): 834–42. PMID 22785217. doi:10.1016/j.ctrv.2012.06.004.
- ↑ Schedin, P (April 2006). "Pregnancy-associated breast cancer and metastasis.". Nature Reviews. Cancer. 6 (4): 281–91. PMID 16557280. doi:10.1038/nrc1839.
- 1 2 3 4 5 6 7 Connie Henke Yarbro; Debra Wujcik; Barbara Holmes Gobel, eds. (2011). Cancer nursing: principles and practice (7 ed.). Jones & Bartlett Publishers. pp. 901–905. ISBN 978-1-4496-1829-2.
- 1 2 Gonçalves V, Sehovic I, Quinn G (2013). "Childbearing attitudes and decisions of young breast cancer survivors: A systematic review". Human Reproduction Update. 20 (2): 279–92. PMC 3922144
 . PMID 24077938. doi:10.1093/humupd/dmt039.
. PMID 24077938. doi:10.1093/humupd/dmt039. - ↑ McNaught J, Reid RL, Provencher DM, et al. (July 2006). "Progesterone-only and non-hormonal contraception in the breast cancer survivor: Joint Review and Committee Opinion of the Society of Obstetricians and Gynaecologists of Canada and the Society of Gynecologic Oncologists of Canada". J Obstet Gynaecol Can. 28 (7): 616–39. PMID 16924781.
- ↑ Management of the menopause after breast cancer, from the Royal Australian and New Zealand College of Obstetricians and Gynaecologists. College Statement C-Gyn 15. 1st Endorsed: February 2003. Current: November 2011. Review: November 2014
- ↑ Venur, Vyshak Alva; Leone, José Pablo (2016-09-13). "Targeted Therapies for Brain Metastases from Breast Cancer". International Journal of Molecular Sciences. 17 (9): 1543. PMC 5037817
 . PMID 27649142. doi:10.3390/ijms17091543.
. PMID 27649142. doi:10.3390/ijms17091543. - ↑ Suryawanshi, Yogesh R.; Zhang, Tiantian; Essani, Karim (2017-02-09). "Oncolytic viruses: emerging options for the treatment of breast cancer". Medical Oncology. 34 (3): 43. ISSN 1357-0560. doi:10.1007/s12032-017-0899-0.
- ↑ Yu, Lin-Yu; Tang, Jie; Zhang, Cong-Min; Zeng, Wen-Jing; Yan, Han; Li, Mu-Peng; Chen, Xiao-Ping (2017-01-12). "New Immunotherapy Strategies in Breast Cancer". International Journal of Environmental Research and Public Health. 14 (1): 68. PMC 5295319
 . PMID 28085094. doi:10.3390/ijerph14010068.
. PMID 28085094. doi:10.3390/ijerph14010068. - ↑ San Antonio Breast Cancer Symposium Archived 16 May 2010 at the Wayback Machine. Abstracts, newsletters, and other reports of the meeting. Archived 16 May 2010 at the Wayback Machine.
- ↑ Goldhirsch A, Ingle JN, Gelber RD, Coates AS, Thürlimann B, Senn HJ (August 2009). "Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009". Annals of Oncology. 20 (8): 1319–29. PMC 2720818
 . PMID 19535820. doi:10.1093/annonc/mdp322.
. PMID 19535820. doi:10.1093/annonc/mdp322. - ↑ "What's new in breast cancer research and treatment?". Cancer. Retrieved 17 November 2015.
- ↑ "Fenretinide (4-HPR): A Preventive Chance for Women at Genetic and Familial Risk?". hindawi. Retrieved 17 November 2015.
- ↑ Sabel, MS (July 2014). "Nonsurgical ablation of breast cancer: future options for small breast tumors.". Surgical oncology clinics of North America. 23 (3): 593–608. PMID 24882353. doi:10.1016/j.soc.2014.03.009.
- 1 2 Roubidoux, MA; Yang, W; Stafford, RJ (March 2014). "Image-guided ablation in breast cancer treatment.". Techniques in vascular and interventional radiology. 17 (1): 49–54. PMID 24636331. doi:10.1053/j.tvir.2013.12.008.
- ↑ "Current Status of Imaging-Guided Percutaneous Ablation of Breast Cancer". American Journal of Roentgenology. 203 (2): 442–448. August 2014. doi:10.2214/AJR.13.11600.
- ↑ Jauliac S, López-Rodriguez C, Shaw LM, Brown LF, Rao A, Toker A (July 2002). "The role of NFAT transcription factors in integrin-mediated carcinoma invasion.". Nature Cell Biology. 4 (7): 540–4. PMID 12080349. doi:10.1038/ncb816.
- ↑ Yoeli-Lerner M, Yiu GK, Rabinovitz I, Erhardt P, Jauliac S, Toker A (23 November 2005). "Akt blocks breast cancer cell motility and invasion through the transcription factor NFAT.". Molecular Cell. 20 (4): 539–50. PMID 16307918. doi:10.1016/j.molcel.2005.10.033.
- 1 2 Fougère M, Gaudineau B, Barbier J, Guaddachi F, Feugeas JP, Auboeuf D, Jauliac S (15 April 2010). "NFAT3 transcription factor inhibits breast cancer cell motility by targeting the Lipocalin 2 gene.". Oncogene. 29 (15): 2292–301. PMID 20101218. doi:10.1038/onc.2009.499.
- ↑ Gaudineau B, Fougère M, Guaddachi F, Lemoine F, de la Grange P, Jauliac S (1 October 2012). "Lipocalin 2 (LCN2), the TNF-like receptor TWEAKR and its ligand TWEAK act downstream of NFAT1 to regulate breast cancer cell invasion.". Journal of Cell Science. 125 (19): 4475–4486. PMID 22767506. doi:10.1242/jcs.099879.
- ↑ Duffy MJ (2001). "Biochemical markers in breast cancer: which ones are clinically useful?". Clin Biochem. 34 (5): 347–52. PMID 11522269. doi:10.1016/s0009-9120(00)00201-0.
- ↑ Goldstein NS, Decker D, Severson D, Schell S, Vicini F, Margolis J, et al. (2007). "Molecular classification system identifies invasive breast carcinoma patients who are most likely and those who are least likely to achieve a complete pathologic response after neoadjuvant chemotherapy.". Cancer. 110 (8): 1687–96. PMID 17722109. doi:10.1002/cncr.22981.
- ↑ Mohammadzadeh F, Mosayebi G, Montazeri V, Darabi M, Fayezi S, Shaaker M, et al. (2014). "Fatty Acid Composition of Tissue Cultured Breast Carcinoma and the Effect of Stearoyl-CoA Desaturase 1 Inhibition.". J Breast Cancer. 17 (2): 136–42. PMC 4090315
 . PMID 25013434. doi:10.4048/jbc.2014.17.2.136.
. PMID 25013434. doi:10.4048/jbc.2014.17.2.136. - ↑ Ranson M, Andronicos NM (2003). "Plasminogen binding and cancer: promises and pitfalls.". Front Biosci. 8: s294–304. PMID 12700073. doi:10.2741/1044.
- ↑ Holder AM, Gonzalez-Angulo AM, Chen H, Akcakanat A, Do KA, Fraser Symmans W, et al. (2013). "High stearoyl-CoA desaturase 1 expression is associated with shorter survival in breast cancer patients.". Breast Cancer Res Treat. 137 (1): 319–27. PMC 3556743
 . PMID 23208590. doi:10.1007/s10549-012-2354-4.
. PMID 23208590. doi:10.1007/s10549-012-2354-4.
External links
| Classification |
V · T · D |
|---|---|
| External resources |
| The Wikibook Sexual Health has a page on the topic of: Cancer#Breast Cancer |
| Library resources about Breast cancer |