Autophagy
Autophagy (or autophagocytosis) (from the Ancient Greek αὐτόφαγος autóphagos, meaning "self-devouring"[1] and κύτος kýtos, meaning "hollow"[2]) is the natural, regulated, destructive mechanism of the cell that disassembles unnecessary or dysfunctional components.[3]
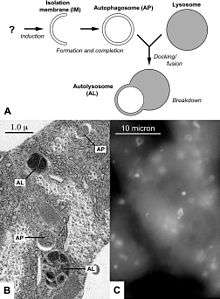
Autophagy allows the orderly degradation and recycling of cellular components.[4][5] In macroautophagy, targeted cytoplasmic constituents are isolated from the rest of the cell within a double-membraned vesicle known as an autophagosome.[6][7] The autophagosome eventually fuses with lysosomes and the contents are degraded and recycled. Three forms of autophagy are commonly described: macroautophagy, microautophagy, and chaperone-mediated autophagy (CMA), along with mitophagy. In disease, autophagy has been seen as an adaptive response to stress, which promotes survival, whereas in other cases it appears to promote cell death and morbidity. In the extreme case of starvation, the breakdown of cellular components promotes cellular survival by maintaining cellular energy levels.
The name "autophagy" was coined by Belgian biochemist Christian de Duve in 1963.[3] The identification of autophagy-related genes in yeast in the 1990s let researchers figure out the mechanisms of autophagy,[8][9][10][11][12] and led to the award of the 2016 Nobel Prize in Physiology or Medicine to Japanese autophagy researcher Yoshinori Ohsumi.[13]
History
Autophagy was first observed by Keith R. Porter and his student Thomas Ashford at the Rockefeller Institute. In January 1962 they reported an increased number of lysosomes in rat liver cells after addition of glucagon, and that some displaced lysosomes towards the centre of the cell contained other cell organelles such as mitochondria. They called this autolysis after Christian de Duve and Alex B. Novikoff. However Porter and Ashford wrongly interpreted their data as lysosome formation (ignoring the pre-existing organelles). Lysosomes could not be cell organelles, but part of cytoplasm such as mitochondria, and that hydrolytic enzymes were produced by microbodies.[14] In 1963 researchers published a detailed ultrastructural description of "focal cytoplasmic degradation," which referenced a 1955 German study of injury-induced sequestration. The study recognized three continuous stages of maturation of the sequestered cytoplasm to lysosomes, and that the process was not limited to injury states that functioned under physiological conditions for "reutilization of cellular materials," and the "disposal of organelles" during differentiation.[15] Inspired by this discovery, de Duve christened the phenomena "autophagy". Unlike Porter and Ashford, de Duve conceived the term as a part of lysosomal function while describing the role of glucagon as a major inducer of cell degradation in the liver. With his student Peter, he established that lysosomes are responsible for glucagon-induced autophagy.[16][17] This was the first time the fact that lysosomes were established as the sites of intracellular autophagy.[3][18][19]
A new era of autophagy research began in 1990s when several groups of scientists independently discovered autophagy-related genes using the budding yeast. Notably, Yoshinori Ohsumi and Michael Thumm examined starvation-induced non-selective autophagy;[9][10][11] in the meantime, Daniel J Klionsky discovered the Cytoplasm-to-Vacuole Targeting (CVT) pathway, which is a form of selective autophagy.[8][12] They soon found that they were in fact looking at essentially the same pathway, just from different angles.[20][21] Initially, the genes discovered by these and other yeast groups were given different names (APG, AUT, CVT, GSA, PAG, PAZ, and PDD). A unified nomenclature was advocated in 2003 by the yeast researchers to use ATG to denote autophagy genes.[22] The 2016 Nobel Prize in Physiology or Medicine was awarded to Yoshinori Ohsumi.[13] Dr. Ohsumi’s contribution to autophagy research is well recognized. However, some have pointed out that the award could have been more inclusive.[23]
The field of autophagy research experienced accelerated growth at the turn of the century. Knowledge of ATG genes provided scientists more convenient tools to dissect functions of autophagy in human health and disease. In 1999, a landmark discovery connecting autophagy with cancer was published by Beth Levine’s group.[24] To this date, relationship between cancer and autophagy continues to be a main theme of autophagy research. The roles of autophagy in neurodegeneration and immune defense also received considerable attention. In 2003, the first Gordon conference on autophagy was held at Waterville.[25] In 2005, Daniel J Klionsky launched “Autophagy”, a scientific journal dedicated to this field. The first Keystone Symposia on autophagy was held in 2007 at Monterey.[26]
Process and pathways
There are four pathways of autophagy and these are mediated by the autophagy-related genes and their associated enzymes.[6][7][27][28][29]
Macroautophagy is the main pathway, used primarily to eradicate damaged cell organelles or unused proteins.[30] This involves the formation of a double membrane known as an autophagosome, around the organelle marked for destruction.[28][31] The autophagosome then travels through the cytoplasm of the cell to a lysosome, and the two organelles fuse.[28] Within the lysosome, the contents of the autophagosome are degraded via acidic lysosomal hydrolases.[32]
Microautophagy, on the other hand, involves the direct engulfment of cytoplasmic material into the lysosome.[33] This occurs by invagination, meaning the inward folding of the lysosomal membrane, or cellular protrusion.[31]
Chaperone-mediated autophagy, or CMA, is a very complex and specific pathway, which involves the recognition by the hsc70-containing complex.[31][34] This means that a protein must contain the recognition site for this hsc70 complex which will allow it to bind to this chaperone, forming the CMA- substrate/chaperone complex.[32] This complex then moves to the lysosomal membrane-bound protein that will recognise and bind with the CMA receptor, allowing it to enter the cell.[35] Upon recognition, the substrate protein gets unfolded and it is translocated across the lysosome membrane with the assistance of the lysosomal hsc70 chaperone.[27][28][35] CMA is significantly different from other types of autophagy because it translocates protein material in a one by one manner, and it is extremely selective about what material crosses the lysosomal barrier.[30][35]
Mitophagy is the selective degradation of mitochondria by autophagy. It often occurs to defective mitochondria following damage or stress. Mitophagy promotes turnover of mitochondria and prevents accumulation of dysfunctional mitochondria which can lead to cellular degeneration. It is mediated by Atg32 (in yeast) and NIX and its regulator BNIP3 in mammals. Mitophagy is regulated by PINK1 and parkin proteins. The occurrence of mitophagy is not limited to the damaged mitochondria but also involves undamaged ones.[29]
Molecular biology
Autophagy is executed by autophagy-related (Atg) genes. The first autophagy genes were identified by genetic screens conducted in the budding yeast Saccharomyces cerevisiae.[8][9][10][11][12] Following their identification those genes were functionally characterized and their orthologs in a variety of different organisms were identified and studied.[6][36]
In mammals, amino acid sensing and additional signals such as growth factors and reactive oxygen species regulate the activity of the protein kinases mTOR and AMPK.[36][37] These two kinases regulate autophagy through inhibitory phosphorylation of the Unc-51-like kinases ULK1 and ULK2 (mammalian homologues of Atg1).[38] Induction of autophagy results in the dephosphorylation and activation of the ULK kinases. ULK is part of a protein complex containing Atg13, Atg101 and FIP200. ULK phosphorylates and activates Beclin-1 (mammalian homologue of Atg6),[39] which is also part of a protein complex. The autophagy-inducible Beclin-1 complex[40] contains the proteins p150, Atg14L and the class III phosphatidylinositol 3-phosphate kinase (PI(3)K) VPS34.[41] The active ULK and Beclin-1 complexes re-localize to the site of autophagosome initiation, the phagophore, where they both contribute to the activation of downstream autophagy components.[42][43]
Once active, VPS34 phosphorylates the lipid phosphatidylinositol to generate phosphatidylinositol 3-phosphate (PtdIns(3)P) on the surface of the phagophore. The generated PtdIns(3)P is used as a docking point for proteins harboring a PtdIns(3)P binding motif. WIPI2, a PtdIns(3)P binding protein of the WIPI (WD-repeat protein interacting with phosphoinositides) protein family, was recently shown to physically bind Atg16L1.[44] Atg16L1 is a member of an E3-like protein complex involved in one of two ubiquitin-like conjugation systems essential for autophagosome formation. Its binding by WIPI2 recruits it to the phagophore and mediates its activity.[45]
The first of the two ubiquitin-like conjugation systems involved in autophagy covalently binds the ubiquitin-like protein Atg12 to Atg5. The resulting conjugate protein then binds Atg16L1 to form an E3-like complex which functions as part of the second ubiquitin-like conjugation system.[46] This complex binds and activates Atg3, which covalently attaches mammalian homologues of the ubiquitin-like yeast protein Atg8 (LC3A-C, GATE16, and GABARAPL1-3), the most studied being LC3 proteins, to the lipid phosphatidylethanolamine (PE) on the surface of autophagosomes.[47] Lipidated LC3 contributes to the closure of autophagosomes,[48] and enables the docking of specific cargos and adaptor proteins such as Sequestosome-1/p62.[49] The completed autophagosome then fuses with a lysosome through the actions of multiple proteins, including SNAREs[50][51] and UVRAG.[52][53] Following the fusion LC3 is retained on the vesicle's inner side and degraded along with the cargo, while the LC3 molecules attached to the outer side are cleaved off by Atg4 and recycled.[54] The contents of the autolysosome are subsequently degraded and their building blocks are released from the vesicle through the action of permeases.[55]
Functions
Nutrient starvation
Autophagy has roles in various cellular functions. One particular example is in yeasts, where the nutrient starvation induces a high level of autophagy. This allows unneeded proteins to be degraded and the amino acids recycled for the synthesis of proteins that are essential for survival.[56][57][58] In higher eukaryotes, autophagy is induced in response to the nutrient depletion that occurs in animals at birth after severing off the trans-placental food supply, as well as that of nutrient starved cultured cells and tissues.[59][60] Mutant yeast cells that have a reduced autophagic capability rapidly perish in nutrition-deficient conditions.[61] Studies on the apg mutants suggest that autophagy via autophagic bodies is indispensable for protein degradation in the vacuoles under starvation conditions, and that at least 15 APG genes are involved in autophagy in yeast.[61] A gene known as ATG7 has been implicated in nutrient-mediated autophagy, as mice studies have shown that starvation-induced autophagy was impaired in atg7-deficient mice.[60]
Xenophagy
In microbiology, xenophagy is the autophagic degradation of infectious particles. Cellular autophagic machinery also play an important role in innate immunity. Intracellular pathogens, such as Mycobacterium tuberculosis (the bacterium which is responsible for tuberculosis) are targeted for degradation by the same cellular machinery and regulatory mechanisms that target host mitochondria for degradation. Incidentally, this is further evidence for the endosymbiotic hypothesis. This process generally leads to the destruction of the invasive organism, although some bacteria can block the maturation of phagosomes into degradative organelles called phagolysosomes.[62] Stimulation of autophagy in infected cells can help overcome this phenomenon, restoring pathogen degradation.
Infection
Vesicular stomatitis virus is believed to be taken up by the autophagosome from the cytosol and translocated to the endosomes where detection takes place by a member of the PRRs called toll-like receptor 7, detecting single stranded RNA. Following activation of the toll-like receptor, intracellular signaling cascades are initiated, leading to induction of interferon and other antiviral cytokines. A subset of viruses and bacteria subvert the autophagic pathway to promote their own replication.[63] Galectin-8 has recently been identified as an intracellular "danger receptor", able to initiate autophagy against intracellular pathogens. When galectin-8 binds to a damaged vacuole, it recruits autophagy adaptor such as NDP52 leading to the formation of an autophagosome and bacterial degradation.[64]
Repair mechanism
Autophagy degrades damaged organelles, cell membranes and proteins, and the failure of autophagy is thought to be one of the main reasons for the accumulation of cell damage and aging.[65]
Programmed cell death
One of the mechanisms of programmed cell death (PCD) is associated with the appearance of autophagosomes and depends on autophagy proteins. This form of cell death most likely corresponds to a process that has been morphologically defined as autophagic PCD. One question that constantly arises, however, is whether autophagic activity in dying cells is the cause of death or is actually an attempt to prevent it. Morphological and histochemical studies so far did not prove a causative relationship between the autophagic process and cell death. In fact, there have recently been strong arguments that autophagic activity in dying cells might actually be a survival mechanism.[66][67] Studies of the metamorphosis of insects have shown cells undergoing a form of PCD that appears distinct from other forms; these have been proposed as examples of autophagic cell death.[68] Recent pharmacological and biochemical studies have proposed that survival and lethal autophagy can be distinguished by the type and degree of regulatory signaling during stress particularly after viral infection.[69] Although promising, these findings have not been examined in non-viral systems.
Caloric restriction
Research suggests that autophagy is required for the lifespan-prolonging effects of caloric restriction. A 2010 French study of nematodes, mice and flies showed that inhibition of autophagy exposed cells to metabolic stress. Resveratrol and the dietary restriction prolonged the lifespan of normal, autophagy-proficient nematodes, but not of nematodes in which autophagy had been inhibited by knocking out Beclin 1 (a known autophagic modulator).[70]
Exercise
Autophagy is essential for basal homeostasis; it is also extremely important in maintaining muscle homeostasis during physical exercise.[71][72] Autophagy at the molecular level is only partially understood. A study of mice shows that autophagy is important for the ever-changing demands of their nutritional and energy needs, particularly through the metabolic pathways of protein catabolism. In a 2012 study conducted by the University of Texas Southwestern Medical Center in Dallas, mutant mice (with a knock-in mutation of BCL2 phosphorylation sites to produce progeny that showed normal levels of basal autophagy yet were deficient in stress-induced autophagy) were tested to challenge this theory. Results showed that when compared to a control group, these mice illustrated a decrease in endurance and an altered glucose metabolism during acute exercise.[71]
Another study demonstrated that skeletal muscle fibres of collagen VI knockout mice showed signs of degeneration due to an insufficiency of autophagy which led to an accumulation of damaged mitochondria and excessive cell death.[73] Exercise-induced autophagy was unsuccessful however; but when autophagy was induced artificially post-exercise, the accumulation of damaged organelles in collagen VI deficient muscle fibres was prevented and cellular homeostasis was maintained. Both studies demonstrate that autophagy induction may contribute to the beneficial metabolic effects of exercise and that it is essential in the maintaining of muscle homeostasis during exercise, particularly in collagen VI fibres.[71][72][73]
Work at the Institute for Cell Biology, University of Bonn, showed that a certain type of autophagy, i.e., chaperone-assisted selective autophagy (CASA), is induced in contracting muscles and is required for maintaining the muscle sarcomere under mechanical tension.[74] The CASA chaperone complex recognizes mechanically damaged cytoskeleton components and directs these components through a ubiquitin-dependent autophagic sorting pathway to lysosomes for disposal. This is necessary for maintaining muscle activity.[74][75]
Osteoarthritis
Because autophagy decreases with age and age is a major risk factor for osteoarthritis, the role of autophagy in the development of this disease is suggested. Proteins involved in autophagy are reduced with age in both human and mouse articular cartilage.[76] Mechanical injury to cartilage explants in culture also reduced autophagy proteins.[77] Autophagy is constantly activated in normal cartilage but it is compromised with age and precedes cartilage cell death and structural damage.[78] These results suggest autophagy is a normal protective process (chondroprotection) in the joint.
Cancer
Oftentimes, cancer occurs when several different pathways that regulate cell differentiation are disturbed. Autophagy plays an important role in cancer – both in protecting against cancer as well as potentially contributing to the growth of cancer.[66][79] Autophagy can contribute to cancer by promoting survival of tumor cells that have been starved, or that degrade apoptotic mediators through autophagy: in such cases, use of inhibitors of the late stages of autophagy (such as chloroquine), on the cells that use autophagy to survive, increases the number of cancer cells killed by antineoplastic drugs.[80]
The role of autophagy in cancer is one that has been highly researched and reviewed. There is evidence that emphasizes the role of autophagy both as a tumor suppressor as well as a factor in tumor cell survival. However, recent research has been able to show that autophagy is more likely to be used as a tumor suppressor according to several models.[79]
Tumor suppressor
Several experiments have been done with mice and varying Beclin1, a protein that regulates autophagy. When the Beclin1 gene was altered to be heterozygous (Beclin 1+/-), the mice were found to be tumor prone.[81] However, when Beclin1 was overexpressed, tumor development was inhibited.[82] Care should be exercised when interpreting phenotypes of beclin mutants and attributing the observations to a defect in autophagy, however: Beclin1 is generally required for phosphatidylinositol 3- phosphate production and as such it affects numerous lysosomal and endosomal functions, including endocytosis and endocytic degradation of activated growth factor receptors. In support of the possibility that Beclin1 affects cancer development through an autophagy-independent pathway is the fact that core autophagy factors which are not known to affect other cellular processes and are definitely not known to affect cell proliferation and cell death, such as Atg7 or Atg5, show a much different phenotype when the respective gene is knocked out, which does not include tumor formation. In addition, full knockout of Beclin1 is embryonic lethal whereas knockout of Atg7 or Atg5 is not.
Necrosis and chronic inflammation also has been shown to be limited through autophagy which helps protect against the formation of tumor cells. Thus these experiments show autophagy's role as a tumor suppressor.[83]
Tumor cell survival
Alternatively, autophagy has also been shown to play a huge role in tumor cell survival. In cancerous cells, autophagy is used as a way to deal with stress on the cell.[84] Once these autophagy related genes were inhibited, cell death was potentiated.[85] The increase in metabolic energy is offset by autophagy functions. These metabolic stresses include hypoxia, nutrient deprivation, and an increase in proliferation. These stresses activate autophagy in order to recycle ATP and maintain survival of the cancerous cells.[86] Autophagy has been shown to enable continued growth of tumor cells by maintaining cellular energy production. By inhibiting autophagy genes in these tumors cells, regression of the tumor and extended survival of the organs affected by the tumors were found. Furthermore, inhibition of autophagy has also been shown to enhance the effectiveness of anticancer therapies.[86]
Mechanism of cell death
Cells that undergo an extreme amount of stress experience cell death either through apoptosis or necrosis. Prolonged autophagy activation leads to a high turnover rate of proteins and organelles. A high rate above the survival threshold may kill cancer cells with a high apoptotic threshold.[86][87] This technique can be utilized as a therapeutic cancer treatment.[66]
Therapeutic target
New developments in research have found that targeted autophagy may be a viable therapeutic solution in fighting cancer. As discussed above, autophagy plays both a role in tumor suppression and tumor cell survival. Thus, the qualities of autophagy can be used as a strategy for cancer prevention. The first strategy is to induce autophagy and enhance its tumor suppression attributes. The second strategy is to inhibit autophagy and thus induce apoptosis.[85]
The first strategy has been tested by looking at dose-response anti-tumor effects during autophagy-induced therapies. These therapies have shown that autophagy increases in a dose-dependent manner. This is directly related to the growth of cancer cells in a dose-dependent manner as well.[84][87] This data supports the development of therapies that will encourage autophagy. Secondly, inhibiting the protein pathways directly known to induce autophagy may also serve as an anticancer therapy.[85][87]
The second strategy is based on the idea that autophagy is a protein degradation system used to maintain homeostasis and the findings that inhibition of autophagy often leads to apoptosis. Inhibition of autophagy is riskier as it may lead to cell survival instead of the desired cell death.[84]
Parkinson disease
Parkinson disease is a neurodegenerative disorder partially caused by the cell death of brain and brain stem cells in many nuclei like the substantia nigra. There are several genetic mutations implicated in the disease, including loss of function PINK1 [88] and Parkin.[89] Loss of function in these genes can lead to damaged mitochondrial accumulation and protein aggregates than can lead to cellular degeneration. Mitochondria is involved in Parkinson's disease. In idiopathic Parkinson's disease, the disease is commonly caused by dysfunctional mitochondria, cellular oxidative stress, autophagic alterations and the aggregation of proteins. These can lead to mitochondrial swelling and depolarization.[90]
Olive oil
Recent research has found that extra virgin olive oil can activate autophagy and is likely to be one of the primary reasons a Mediterranean diet (that involves high consumption of olive oil) leads to higher life expectancy and lower incidence of diseases related to aging such as Alzheimers. The study in mice fed with olive oil resulted in an increase in nerve cell autophagy activation compared to controls that had the same diet but without olive oil.[91]
See also
References
- ↑ Henry George Liddell; Robert Scott; Henry Stuart Jones. "A Greek–English Lexicon". tufts.edu. Retrieved 5 October 2016.
- ↑ Henry George Liddell; Robert Scott; Henry Stuart Jones. "A Greek–English Lexicon". tufts.edu. Retrieved 5 October 2016.
- 1 2 3 Klionsky, DJ (2008). "Autophagy revisited: A conversation with Christian de Duve". Autophagy. 4 (6): 740–3. PMID 18567941. doi:10.4161/auto.6398.
- ↑ Mizushima, N; Komatsu, M (11 November 2011). "Autophagy: renovation of cells and tissues.". Cell. 147 (4): 728–41. PMID 22078875. doi:10.1016/j.cell.2011.10.026.
- ↑ Kobayashi S (2015). "Choose Delicately and Reuse Adequately: The Newly Revealed Process of Autophagy". Biological & Pharmaceutical Bulletin. 38 (8): 1098–103. PMID 26235572. doi:10.1248/bpb.b15-00096.
- 1 2 3 Mizushima, Noboru; Yoshimori, Tamotsu; Ohsumi, Yoshinori (10 November 2011). "The Role of Atg Proteins in Autophagosome Formation". Annual Review of Cell and Developmental Biology. 27 (1): 107–132. doi:10.1146/annurev-cellbio-092910-154005.
- 1 2 Xie, Z; Klionsky, DJ (October 2007). "Autophagosome formation: core machinery and adaptations.". Nature Cell Biology. 9 (10): 1102–9. PMID 17909521. doi:10.1038/ncb1007-1102.
- 1 2 3 Klionsky, DJ; Cueva, R; Yaver, DS (October 1992). "Aminopeptidase I of Saccharomyces cerevisiae is localized to the vacuole independent of the secretory pathway.". The Journal of Cell Biology. 119 (2): 287–99. PMC 2289658
 . PMID 1400574. doi:10.1083/jcb.119.2.287.
. PMID 1400574. doi:10.1083/jcb.119.2.287. - 1 2 3 Takeshige, K; Baba, M; Tsuboi, S; Noda, T; Ohsumi, Y (October 1992). "Autophagy in yeast demonstrated with proteinase-deficient mutants and conditions for its induction.". The Journal of Cell Biology. 119 (2): 301–11. PMC 2289660
 . PMID 1400575. doi:10.1083/jcb.119.2.301.
. PMID 1400575. doi:10.1083/jcb.119.2.301. - 1 2 3 Thumm, M; Egner, R; Koch, B; Schlumpberger, M; Straub, M; Veenhuis, M; Wolf, DH (1 August 1994). "Isolation of autophagocytosis mutants of Saccharomyces cerevisiae.". FEBS Letters. 349 (2): 275–80. PMID 8050581. doi:10.1016/0014-5793(94)00672-5.
- 1 2 3 Tsukada, M; Ohsumi, Y (25 October 1993). "Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae.". FEBS Letters. 333 (1–2): 169–74. PMID 8224160. doi:10.1016/0014-5793(93)80398-e.
- 1 2 3 Harding, TM; Morano, KA; Scott, SV; Klionsky, DJ (November 1995). "Isolation and characterization of yeast mutants in the cytoplasm to vacuole protein targeting pathway.". The Journal of Cell Biology. 131 (3): 591–602. PMC 2120622
 . PMID 7593182. doi:10.1083/jcb.131.3.591.
. PMID 7593182. doi:10.1083/jcb.131.3.591. - 1 2 "The Nobel Prize in Physiology or Medicine 2016". The Nobel Foundation. 3 October 2016. Retrieved 3 October 2016.
- ↑ Ashford, TP; Porter, KR (1962). "Cytoplasmic components in hepatic cell lysosomes". The Journal of Cell Biology. 12 (1): 198–202. PMC 2106008
 . PMID 13862833. doi:10.1083/jcb.12.1.198.
. PMID 13862833. doi:10.1083/jcb.12.1.198. - ↑ Hruban, Z; Spargo B; Swift H; Wissler RW; Kleinfeld RG (1963). "Focal cytoplasmic degradation". Am J Pathol. 42 (6): 657–683. PMC 1949709
 . PMID 13955261.
. PMID 13955261. - ↑ Deter, RL; Baudhuin, P; De Duve, C (1967). "Participation of lysosomes in cellular autophagy induced in rat liver by glucagon". The Journal of Cell Biology. 35 (2): C11–6. PMC 2107130
 . PMID 6055998. doi:10.1083/jcb.35.2.c11.
. PMID 6055998. doi:10.1083/jcb.35.2.c11. - ↑ Deter, RL; De Duve, C (1967). "Influence of glucagon, an inducer of cellular autophagy, on some physical properties of rat liver lysosomes". The Journal of Cell Biology. 33 (2): 437–49. PMC 2108350
 . PMID 4292315. doi:10.1083/jcb.33.2.437.
. PMID 4292315. doi:10.1083/jcb.33.2.437. - ↑ De Duve, C (1983). "Lysosomes revisited". European Journal of Biochemistry / FEBS. 137 (3): 391–7. PMID 6319122. doi:10.1111/j.1432-1033.1983.tb07841.x.
- ↑ William A. Dunn Jr.; Laura A. Schroder; John P. Aris (2013). "Historical overview of autophagy". In Hong-Gang Wang. Autophagy and Cancer. Springer. pp. 3–4. ISBN 9781461465614.
- ↑ Harding, TM; Hefner-Gravink, A; Thumm, M; Klionsky, DJ (26 July 1996). "Genetic and phenotypic overlap between autophagy and the cytoplasm to vacuole protein targeting pathway.". The Journal of Biological Chemistry. 271 (30): 17621–4. PMID 8663607. doi:10.1074/jbc.271.30.17621.
- ↑ Scott, SV; Hefner-Gravink, A; Morano, KA; Noda, T; Ohsumi, Y; Klionsky, DJ (29 October 1996). "Cytoplasm-to-vacuole targeting and autophagy employ the same machinery to deliver proteins to the yeast vacuole.". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12304–8. PMC 37986
 . PMID 8901576. doi:10.1073/pnas.93.22.12304.
. PMID 8901576. doi:10.1073/pnas.93.22.12304. - ↑ Klionsky, DJ; Cregg, JM; Dunn, WA Jr; Emr, SD; Sakai, Y; Sandoval, IV; Sibirny, A; Subramani, S; Thumm, M; Veenhuis, M; Ohsumi, Y (October 2003). "A unified nomenclature for yeast autophagy-related genes.". Developmental Cell. 5 (4): 539–45. PMID 14536056. doi:10.1016/s1534-5807(03)00296-x.
- ↑ "Medicine Nobel for research on how cells 'eat themselves'". Nature News. Retrieved 4 October 2016.
- ↑ Liang, XH; Jackson, S; Seaman, M; Brown, K; Kempkes, B; Hibshoosh, H; Levine, B (9 December 1999). "Induction of autophagy and inhibition of tumorigenesis by beclin 1.". Nature. 402 (6762): 672–6. PMID 10604474. doi:10.1038/45257.
- ↑ "Autophagy in Stress, Development & Disease, 2003, Gordon Research Conference".
- ↑ "Autophagy in Health and Disease (Z3), 2007, Keystone Symposia on Molecular and Cellular Biology".
- 1 2 Lee J, Giordano S, Zhang J (January 2012). "Autophagy, mitochondria and oxidative stress: cross-talk and redox signalling". Biochem. J. 441 (2): 523–40. PMC 3258656
 . PMID 22187934. doi:10.1042/BJ20111451.
. PMID 22187934. doi:10.1042/BJ20111451. - 1 2 3 4 Mizushima N, Ohsumi Y, Yoshimori T (December 2002). "Autophagosome formation in mammalian cells". Cell Struct. Funct. 27 (6): 421–9. PMID 12576635. doi:10.1247/csf.27.421.
- 1 2 Youle, R; Narendra, D (2011). "Mechanisms of mitophagy". Nature Reviews Molecular Cell Biology. 12: 9–14. doi:10.1038/nrm3028.
- 1 2 Levine B, Mizushima N, Virgin HW (January 2011). "Autophagy in immunity and inflammation". Nature. 469 (7330): 323–35. PMC 3131688
 . PMID 21248839. doi:10.1038/nature09782.
. PMID 21248839. doi:10.1038/nature09782. - 1 2 3 Česen MH, Pegan K, Spes A, Turk B (July 2012). "Lysosomal pathways to cell death and their therapeutic applications". Exp. Cell Res. 318 (11): 1245–51. PMID 22465226. doi:10.1016/j.yexcr.2012.03.005.
- 1 2 Homma, K.S. (2011). "List of autophagy-related proteins and 3D structures". Autophagy Database. 290. Archived from the original on 2012-08-01. Retrieved 2012-10-08
- ↑ "The Discovery of Lysosomes and Autophagy". 49. 3: 49. 2010
- ↑ Bandyopadhyay U, Kaushik S, Varticovski L, Cuervo AM (September 2008). "The chaperone-mediated autophagy receptor organizes in dynamic protein complexes at the lysosomal membrane". Mol. Cell. Biol. 28 (18): 5747–63. PMC 2546938
 . PMID 18644871. doi:10.1128/MCB.02070-07.
. PMID 18644871. doi:10.1128/MCB.02070-07. - 1 2 3 Perry, A. (2010). Wiley & Sons Ltd., Chichester. 115 (11): 0 Missing or empty
|title=(help) - 1 2 C. A. Lamb, T. Yoshimori, and S. A. Tooze, 'The Autophagosome: Origins Unknown, Biogenesis Complex', Nat Rev Mol Cell Biol, 14 (2013), 759-74
- ↑ R. C. Russell, H. X. Yuan, and K. L. Guan, 'Autophagy Regulation by Nutrient Signaling', Cell Res, 24 (2014), 42-57
- ↑ E. Y. Chan, 'Regulation and Function of Uncoordinated-51 Like Kinase Proteins', Antioxid Redox Signal, 17 (2012), 775-85
- ↑ R. C. Russell, Y. Tian, H. Yuan, H. W. Park, Y. Y. Chang, J. Kim, H. Kim, T. P. Neufeld, A. Dillin, and K. L. Guan, 'Ulk1 Induces Autophagy by Phosphorylating Beclin-1 and Activating Vps34 Lipid Kinase', Nat Cell Biol, 15 (2013), 741-50
- ↑ E. Itakura, C. Kishi, K. Inoue, and N. Mizushima, 'Beclin 1 Forms Two Distinct Phosphatidylinositol 3-Kinase Complexes with Mammalian Atg14 and Uvrag', Mol Biol Cell, 19 (2008), 5360-72
- ↑ R. Kang, H. J. Zeh, M. T. Lotze, and D. Tang, 'The Beclin 1 Network Regulates Autophagy and Apoptosis', Cell Death Differ, 18 (2011), 571-80
- ↑ S. Di Bartolomeo, M. Corazzari, F. Nazio, S. Oliverio, G. Lisi, M. Antonioli, V. Pagliarini, S. Matteoni, C. Fuoco, L. Giunta, M. D'Amelio, R. Nardacci, A. Romagnoli, M. Piacentini, F. Cecconi, and G. M. Fimia, 'The Dynamic Interaction of Ambra1 with the Dynein Motor Complex Regulates Mammalian Autophagy', J Cell Biol, 191 (2010), 155-68
- ↑ T. Hara, A. Takamura, C. Kishi, S. Iemura, T. Natsume, J. L. Guan, and N. Mizushima, 'Fip200, a Ulk-Interacting Protein, Is Required for Autophagosome Formation in Mammalian Cells', J Cell Biol, 181 (2008), 497-510
- ↑ T. Proikas-Cezanne, Z. Takacs, P. Donnes, and O. Kohlbacher, 'Wipi Proteins: Essential Ptdins3p Effectors at the Nascent Autophagosome', J Cell Sci, 128 (2015), 207-17
- ↑ H. C. Dooley, M. Razi, H. E. Polson, S. E. Girardin, M. I. Wilson, and S. A. Tooze, 'Wipi2 Links Lc3 Conjugation with Pi3p, Autophagosome Formation, and Pathogen Clearance by Recruiting Atg12-5-16l1', Mol Cell, 55 (2014), 238-52
- ↑ T. Hanada, N. N. Noda, Y. Satomi, Y. Ichimura, Y. Fujioka, T. Takao, F. Inagaki, and Y. Ohsumi, 'The Atg12-Atg5 Conjugate Has a Novel E3-Like Activity for Protein Lipidation in Autophagy', J Biol Chem, 282 (2007), 37298-302
- ↑ Y. Kabeya, N. Mizushima, A. Yamamoto, S. Oshitani-Okamoto, Y. Ohsumi, and T. Yoshimori, 'Lc3, Gabarap and Gate16 Localize to Autophagosomal Membrane Depending on Form-Ii Formation', J Cell Sci, 117 (2004), 2805-12
- ↑ N. Fujita, M. Hayashi-Nishino, H. Fukumoto, H. Omori, A. Yamamoto, T. Noda, and T. Yoshimori, 'An Atg4b Mutant Hampers the Lipidation of Lc3 Paralogues and Causes Defects in Autophagosome Closure', Mol Biol Cell, 19 (2008), 4651-9
- ↑ S. Park, S. G. Choi, S. M. Yoo, J. H. Son, and Y. K. Jung, 'Choline Dehydrogenase Interacts with Sqstm1/P62 to Recruit Lc3 and Stimulate Mitophagy', Autophagy, 10 (2014), 1906-20
- ↑ C. M. Fader, D. G. Sanchez, M. B. Mestre, and M. I. Colombo, 'Ti-Vamp/Vamp7 and Vamp3/Cellubrevin: Two V-Snare Proteins Involved in Specific Steps of the Autophagy/Multivesicular Body Pathways', Biochim Biophys Acta, 1793 (2009), 1901-16
- ↑ N. Furuta, N. Fujita, T. Noda, T. Yoshimori, and A. Amano, 'Combinational Soluble N-Ethylmaleimide-Sensitive Factor Attachment Protein Receptor Proteins Vamp8 and Vti1b Mediate Fusion of Antimicrobial and Canonical Autophagosomes with Lysosomes', Mol Biol Cell, 21 (2010), 1001-10
- ↑ Y. M. Kim, C. H. Jung, M. Seo, E. K. Kim, J. M. Park, S. S. Bae, and D. H. Kim, 'Mtorc1 Phosphorylates Uvrag to Negatively Regulate Autophagosome and Endosome Maturation', Mol Cell (2014)
- ↑ C. Liang, J. S. Lee, K. S. Inn, M. U. Gack, Q. Li, E. A. Roberts, I. Vergne, V. Deretic, P. Feng, C. Akazawa, and J. U. Jung, 'Beclin1-Binding Uvrag Targets the Class C Vps Complex to Coordinate Autophagosome Maturation and Endocytic Trafficking', Nat Cell Biol, 10 (2008), 776-87
- ↑ K. Satoo, N. N. Noda, H. Kumeta, Y. Fujioka, N. Mizushima, Y. Ohsumi, and F. Inagaki, 'The Structure of Atg4b-Lc3 Complex Reveals the Mechanism of Lc3 Processing and Delipidation During Autophagy', EMBO J, 28 (2009), 1341-50
- ↑ Z. Yang, J. Huang, J. Geng, U. Nair, and D. J. Klionsky, 'Atg22 Recycles Amino Acids to Link the Degradative and Recycling Functions of Autophagy', Mol Biol Cell, 17 (2006), 5094-104
- ↑ Reggiori F, Klionsky DJ (February 2002). "Autophagy in the eukaryotic cell". Eukaryotic Cell. 1 (1): 11–21. PMC 118053
 . PMID 12455967. doi:10.1128/EC.01.1.11-21.2002.
. PMID 12455967. doi:10.1128/EC.01.1.11-21.2002. - ↑ Klionsky, Daniel J.; Emr, Scott D. (2000). "Autophagy as a regulated pathway of cellular degradation". Science. 290 (5497): 1717–1721. doi:10.1126/science.290.5497.1717
- ↑ Levine B, Klionsky DJ (April 2004). "Development by self-digestion: molecular mechanisms and biological functions of autophagy". Dev. Cell. 6 (4): 463–77. PMID 15068787. doi:10.1016/S1534-5807(04)00099-1.
- ↑ Kuma A, Hatano M, Matsui M, et al. (December 2004). "The role of autophagy during the early neonatal starvation period". Nature. 432 (7020): 1032–6. PMID 15525940. doi:10.1038/nature03029.
- 1 2 Mizushima N, Yamamoto A, Matsui M, Yoshimori T, Ohsumi Y (March 2004). "In vivo analysis of autophagy in response to nutrient starvation using transgenic mice expressing a fluorescent autophagosome marker". Mol. Biol. Cell. 15 (3): 1101–11. PMC 363084
 . PMID 14699058. doi:10.1091/mbc.E03-09-0704.
. PMID 14699058. doi:10.1091/mbc.E03-09-0704. - 1 2 Tsukada M, Ohsumi Y (October 1993). "Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae". FEBS Lett. 333 (1–2): 169–74. PMID 8224160. doi:10.1016/0014-5793(93)80398-E.
- ↑ Deretic V, Delgado M, Vergne I, Master S, De Haro S, Ponpuak M, Singh S (2009). "Autophagy in immunity against mycobacterium tuberculosis: a model system to dissect immunological roles of autophagy". Curr. Top. Microbiol. Immunol. Current Topics in Microbiology and Immunology. 335: 169–88. ISBN 978-3-642-00301-1. PMC 2788935
 . PMID 19802565. doi:10.1007/978-3-642-00302-8_8.
. PMID 19802565. doi:10.1007/978-3-642-00302-8_8. - ↑ Jackson WT, Giddings TH, Taylor MP, et al. (May 2005). "Subversion of cellular autophagosomal machinery by RNA viruses". PLoS Biol. 3 (5): e156. PMC 1084330
 . PMID 15884975. doi:10.1371/journal.pbio.0030156.
. PMID 15884975. doi:10.1371/journal.pbio.0030156. 
- ↑ Thurston TL, Wandel MP, von Muhlinen N, Foeglein A, Randow F (February 2012). "Galectin 8 targets damaged vesicles for autophagy to defend cells against bacterial invasion". Nature. 482 (7385): 414–8. PMC 3343631
 . PMID 22246324. doi:10.1038/nature10744.
. PMID 22246324. doi:10.1038/nature10744. - ↑ Cuervo AM, Bergamini E, Brunk UT, Dröge W, Ffrench M, Terman A (2005). "Autophagy and aging: the importance of maintaining "clean" cells". Autophagy. 1 (3): 131–40. PMID 16874025. doi:10.4161/auto.1.3.2017.
- 1 2 3 Tavassoly, Iman (2015). Dynamics of Cell Fate Decision Mediated by the Interplay of Autophagy and Apoptosis in Cancer Cells. Springer International Publishing. ISBN 978-3-319-14962-2.
- ↑ Tsujimoto Y, Shimizu S (November 2005). "Another way to die: autophagic programmed cell death". Cell Death Differ. 12 (Suppl 2): 1528–34. PMID 16247500. doi:10.1038/sj.cdd.4401777.
- ↑ Schwartz LM, Smith SW, Jones ME, Osborne BA (February 1993). "Do all programmed cell deaths occur via apoptosis?". Proc. Natl. Acad. Sci. U.S.A. 90 (3): 980–4. PMC 45794
 . PMID 8430112. doi:10.1073/pnas.90.3.980.
. PMID 8430112. doi:10.1073/pnas.90.3.980. - ↑ Datan E, Shirazian A, Benjamin S, Matassov D, Tinari A, Malorni W, Lockshin RA, Garcia-Sastre A, Zakeri Z (2014). "mTOR/p70S6K signaling distinguishes routine, maintenance-level autophagy from autophagic cell death during influenza A infection". Virology. 452–453 (March 2014): 175–190. PMC 4005847
 . PMID 24606695. doi:10.1016/j.virol.2014.01.008.
. PMID 24606695. doi:10.1016/j.virol.2014.01.008. - ↑ Morselli E, Maiuri MC, Markaki M, et al. (2010). "Caloric restriction and resveratrol promote longevity through the Sirtuin-1-dependent induction of autophagy". Cell Death Dis. 1 (1): e10–. PMC 3032517
 . PMID 21364612. doi:10.1038/cddis.2009.8.
. PMID 21364612. doi:10.1038/cddis.2009.8. - 1 2 3 He C, Bassik MC, Moresi V, et al. (January 2012). "Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis". Nature. 481 (7382): 511–5. PMC 3518436
 . PMID 22258505. doi:10.1038/nature10758.
. PMID 22258505. doi:10.1038/nature10758. - 1 2 Nair U, Klionsky DJ (December 2011). "Activation of autophagy is required for muscle homeostasis during physical exercise". Autophagy. 7 (12): 1405–6. PMC 3288013
 . PMID 22082869. doi:10.4161/auto.7.12.18315.
. PMID 22082869. doi:10.4161/auto.7.12.18315. - 1 2 Grumati P, Coletto L, Schiavinato A, et al. (December 2011). "Physical exercise stimulates autophagy in normal skeletal muscles but is detrimental for collagen VI-deficient muscles". Autophagy. 7 (12): 1415–23. PMC 3288016
 . PMID 22024752. doi:10.4161/auto.7.12.17877.
. PMID 22024752. doi:10.4161/auto.7.12.17877. - 1 2 Arndt V, Dick N, Tawo R, Dreiseidler M, Wenzel D, Hesse M, Fürst DO, Saftig P, Saint R, Fleischmann BK, Hoch M, Höhfeld J (January 2010). "Chaperone-assisted selective autophagy is essential for muscle maintenance". Curr Biol. 20 (2): 143–8. PMID 20060297. doi:10.1016/j.cub.2009.11.022.
- ↑ Ulbricht A, Eppler FJ, Tapia VE, van der Ven PF, Hampe N, Hersch N, Vakeel P, Stadel D, Haas A, Saftig P, Behrends C, Fürst DO, Volkmer R, Hoffmann B, Kolanus W, Höhfeld J (February 2013). "Cellular Mechanotransduction Relies on Tension-Induced and Chaperone-Assisted Autophagy". Curr Biol. 23 (5): 430–5. PMID 23434281. doi:10.1016/j.cub.2013.01.064.
- ↑ Carames, B; Taniguchi, N; Otsuki, S; Blanco, FJ; Lotz, M (2010). "Autophagy is a protective mechanism in normal cartilage, and its aging related loss is linked with cell death and osteoarthritis". Arthritis Rheum. 62 (3): 791–801. PMC 2838960
 . PMID 20187128. doi:10.1002/art.27305.
. PMID 20187128. doi:10.1002/art.27305. - ↑ Carames, B; Taniguchi, N; Seino, D; Blanco, FJ; D'Lima, D; Lotz, M (2012). "Mechanical injury suppresses autophagy regulators and pharmacologic activation of autophagy results in chondroprotection". Arthritis Rheum. 64 (4): 1182–1192. PMC 3288456
 . PMID 22034068. doi:10.1002/art.33444.
. PMID 22034068. doi:10.1002/art.33444. - ↑ Carames, B; Olmer, M; Kiosses, WB; Lotz, MK (2015). "The relationship of autophagy defects to Cartilage Damage During joint aging in a mouse model". Arthritis Rheumatol. 67 (6): 1568–1576. PMC 4446178
 . PMID 25708836. doi:10.1002/art.39073.
. PMID 25708836. doi:10.1002/art.39073. - 1 2 Furuya, N., Liang, X.H., and Levin, B. 2004. Autophagy and cancer. In Autophagy. D.J. Klionsky editor. Landes Bioscience. Georgetown, Texas, USA. 244-253.
- ↑ Vlahopoulos S, Critselis E, Voutsas IF, Perez SA, Moschovi M, Baxevanis CN, Chrousos GP (2014). "New use for old drugs? Prospective targets of chloroquines in cancer therapy". Curr Drug Targets. 15 (9): 843–51. PMID 25023646. doi:10.2174/1389450115666140714121514.
- ↑ Qu X, Yu J, Bhagat G, Furuya N, Hibshoosh H, Troxel A, et al. (2003). "Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene". J Clin Invest. 112 (12): 1809–20. PMC 297002
 . PMID 14638851. doi:10.1172/JCI20039.
. PMID 14638851. doi:10.1172/JCI20039. - ↑ Liang XH, Jackson S, Seaman M, Brown K, Kempkes B, Hibshoosh H, et al. (1999). "Induction of autophagy and inhibition of tumorigenesis by beclin 1". Nature. 402 (6762): 672–6. PMID 10604474. doi:10.1038/45257.
- ↑ Duran A, Linares JF, Galvez AS, Wikenheiser K, Flores JM, Diaz-Meco MT, et al. (2008). "The signaling adaptor p62 is an important NF-kappaB mediator in tumorigenesis". Cancer Cell. 13 (4): 343–54. PMID 18394557. doi:10.1016/j.ccr.2008.02.001.
- 1 2 3 Paglin, S; Hollister, T; Delohery, T; Hackett, N; McMahill, M; Sphicas, E; Domingo, D; Yahalom, J (2001). "A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles". Cancer Res. 61: 439–44.
- 1 2 3 Jin, S; White, E (2007). "Role of autophagy in cancer: management of metabolic stress". Autophagy. 3: 28–31. PMC 2770734
 . PMID 16969128. doi:10.4161/auto.3269.
. PMID 16969128. doi:10.4161/auto.3269. - 1 2 3 Yang, ZJ; Chee, CE; Huang, S; Sinicrope, FA (2011). "The role of autophagy in cancer: therapeutic implications". Mol Cancer Ther. 10: 1533–1541. doi:10.1158/1535-7163.MCT-11-0047.
- 1 2 3 Tavassoly, I; Parmar, J; Shajahan-Haq, An; Clarke, R; Baumann, Wt; Tyson, Jj (2015-04-01). "Dynamic Modeling of the Interaction Between Autophagy and Apoptosis in Mammalian Cells". CPT: Pharmacometrics & Systems Pharmacology. 4 (4): 263–272. ISSN 2163-8306. PMC 4429580
 . PMID 26225250. doi:10.1002/psp4.29.
. PMID 26225250. doi:10.1002/psp4.29. - ↑ Valente, EM; Abou-sleiman, PM; Caputo, V; et al. (2004). "Hereditary early-onset Parkinson's disease caused by mutations in PINK1". Science. 304 (5674): 1158–60. PMID 15087508. doi:10.1126/science.1096284.
- ↑ Kitada, T; Asakawa, S; Hattori, N; et al. (1998). "Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism". Nature. 392 (6676): 605–8. PMID 9560156. doi:10.1038/33416.
- ↑ Esteves, AR; Arduíno, DM; Silva, DF; Oliveira, CR; Cardoso, SM (2011). "Mitochondrial Dysfunction: The Road to Alpha-Synuclein Oligomerization in PD.". Parkinsons Dis. 2011: 693761. doi:10.4061/2011/693761.
- ↑ "Extra-virgin olive oil preserves memory, protects brain against Alzheimer's". June 21, 2017. Retrieved July 3, 2017.
Further reading
- Liu, Y.; Bassham, D. C. (2012). "Autophagy: Pathways for Self-Eating in Plant Cells". Annual Review of Plant Biology. 63: 215–237. PMID 22242963. doi:10.1146/annurev-arplant-042811-105441.
- Starokadomskyy, P.; Dmytruk, K. (2013). "A bird's-eye view of autophagy". Autophagy. 9 (7): 41–46.
- Tavassoly Iman, Dynamics of Cell Fate Decision Mediated by the Interplay of Autophagy and Apoptosis in Cancer Cells: Mathematical Modeling and Experimental Observation, Springer, 2015.
External links
- Autophagy, a journal produced by Landes Bioscience and edited by DJ Klionsky
- LongevityMeme entry describing PubMed article on the effects of autophagy and lifespan
- Autophagolysosome on Drugs.com
- HADb, a Human Autophagy dedicated Database
- Autophagy DB, an autophagy database that covers all eukaryotes
- Self-Destructive Behavior in Cells May Hold Key to a Longer Life
- Exercise as Housecleaning for the Body