Aluminium fluoride
 Crystal structure | |
| Names | |
|---|---|
| Other names
Aluminium(III) fluoride Aluminum trifluoride | |
| Identifiers | |
| |
| 3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.029.137 |
| PubChem CID |
|
| RTECS number | BD0725000 |
| |
| |
| Properties | |
| AlF3 | |
| Molar mass | 83.977 g/mol (anhydrous) 101.992 g/mol (monohydrate) 138.023 (trihydrate)[1] |
| Appearance | white, crystalline solid odorless |
| Density | 3.10 g/cm3 (anhydrous) 2.17 g/cm3 (monohydrate) 1.914 g/cm3 (trihydrate)[1] |
| Melting point | 1,290 °C (2,350 °F; 1,560 K)[2] (anhydrous) (sublimes) |
| 5.6 g/L (0 °C) 6.7 g/L (20 °C) 17.2 g/L (100 °C) | |
| -13.4·10−6 cm3/mol[3] | |
| Refractive index (nD) |
1.3767 (visible range)[4] |
| Structure | |
| Rhombohedral, hR24 | |
| R3c, No. 167[5] | |
| a = 0.49254 nm, c = 1.24477 nm | |
| Lattice volume (V) |
0.261519 |
| Formula units (Z) |
6 |
| Thermochemistry | |
| 75.1 J/mol·K[6] | |
| Std molar entropy (S |
66.5 J/mol·K[6] |
| Std enthalpy of formation (ΔfH |
−1510.4 kJ/mol[6] |
| Gibbs free energy (ΔfG˚) |
-1431.1 kJ/mol[6] |
| Hazards | |
| EU classification (DSD) (outdated) |
No classification according to EU Regulation (EC) No. 1272/2008. |
| R-phrases (outdated) | – |
| S-phrases (outdated) | – |
| NFPA 704 | |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
none[7] |
| REL (Recommended) |
2 mg/m3[7] |
| IDLH (Immediate danger) |
N.D.[7] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Aluminium fluoride (AlF3) is an inorganic compound used primarily in the production of aluminium. This colorless solid can be prepared synthetically but also occurs in nature as minerals rosenbergite and oskarssonite.
Production and occurrence
The majority of aluminium fluoride is produced by treating alumina with hydrogen fluoride gas at 700 °C:[2]
- H2SiF6 + Al2O3 → 2 AlF3 + SiO2 + H2O
Alternatively, it is manufactured by thermal decomposition of ammonium hexafluoroaluminate.[8] For small scale laboratory preparations, AlF3 can also be prepared by treating aluminium hydroxide or aluminium metal with HF.
Aluminium fluoride trihydrate is found in nature as the rare mineral rosenbergite. The non-hydrated form appears as the mineral oskarssonite.[9]
Structure
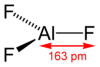
Its structure adopts the rhenium trioxide motif, featuring distorted AlF6 octahedra. Each fluoride is connected to two Al centers. Because of its 3-dimensional polymeric structure, AlF3 has a high melting point. The other trihalides of aluminium in the solid state differ, AlCl3 has a layer structure and AlBr3 and AlI3, are molecular dimers.[10] Also they have low melting points and evaporate readily to give dimers.[11] In the gas phase aluminium fluoride exists as trigonal molecules of D3h symmetry. The Al-F bond lengths of this gaseous molecule are 163 pm.
Applications
Aluminium fluoride is an important additive for the production of aluminium by electrolysis.[2] Together with cryolite, it lowers the melting point to below 1000 °C and increases the conductivity of the solution. It is into this molten salt that aluminium oxide is dissolved and then electrolyzed to give bulk Al metal.[8]
Niche uses
Together with zirconium fluoride, aluminium fluoride is an ingredient for the production of fluoroaluminate glasses.
It is also used to inhibit fermentation.
Like magnesium fluoride it used as a low-index optical thin film, particularly when far UV transparency is required. Its deposition by physical vapor deposition, particularly by evaporation, is favorable.
Safety
AlF3 has low toxicity (LD50 = 600 mg/kg). Repeated or prolonged inhalation exposure may cause asthma. The substance may have effects on the bone, nervous system , resulting in bone alterations (fluorosis), and nervous system impairment.[12]
References
- 1 2 Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.45. ISBN 1439855110.
- 1 2 3 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 233. ISBN 0-08-037941-9.
- ↑ Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.131. ISBN 1439855110.
- ↑ Lide, David R. (2003-06-19). CRC Handbook of Chemistry and Physics, 84th Edition. CRC Handbook. CRC Press. ISBN 9780849304842.
- ↑ Hoppe, R.; Kissel, D. (1984). "Zur kenntnis von AlF3 und InF3 [1]". Journal of Fluorine Chemistry. 24 (3): 327. doi:10.1016/S0022-1139(00)81321-4.
- 1 2 3 4 Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 5.5. ISBN 1439855110.
- 1 2 3 "NIOSH Pocket Guide to Chemical Hazards #0024". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 Aigueperse, J.; Mollard, P.; Devilliers, D.; Chemla, M.; Faron, R.; Romano, R.; Cuer, J. P. (2005) "Fluorine Compounds, Inorganic" in Ullmann’s Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim. doi:10.1002/14356007.a11_307
- ↑ Oskarssonite. Mindat.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ↑ "ALUMINIUM FLUORIDE (ANHYDROUS) International Chemical Safety Cards (ICSC)". CDC.gov National Institute for Occupational Safety and Health (NIOSH). July 22, 2015. Retrieved July 17, 2017.
External links
| Wikimedia Commons has media related to Aluminium fluoride. |
