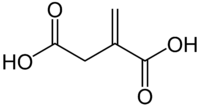
Itaconic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylidenebutanedioic acid | |
| Other names | |
| Identifiers | |
| 3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.002.364 |
| KEGG | |
| PubChem CID |
|
| |
| |
| Properties | |
| C5H6O4 | |
| Molar mass | 130.10 g·mol−1 |
| Appearance | White solid |
| Density | 1.63 g/cm3[1] |
| Melting point | 162 to 164 °C (324 to 327 °F; 435 to 437 K) (decomposes)[1] |
| 1 g/12 mL[1] | |
| Solubility in ethanol | 1 g/5 mL[1] |
| -57.57·10−6 cm3/mol | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Itaconic acid, or methylenesuccinic acid, is an organic compound. This dicarboxylic acid is a white solid that is soluble in water, ethanol, and acetone. The name itaconic was devised as an anagram of aconitic. Historically, itaconic acid was obtained by the distillation of citric acid, but currently it is produced by fermentation.
Laboratory synthesis and reactions
Dry distillation of citric acid affords itaconic anhydride, which readily undergoes hydrolysis to itaconic acid.[2] Upon heating, itaconic anhydride isomerizes to citraconic acid anhydride, which can be hydrolyzed to citraconic acid (2-methylmaleic acid).[3]
Production
Since the 1960s, it is produced industrially by the fermentation of carbohydrates such as glucose or molasses using fungi such as Aspergillus itaconicus or Aspergillus terreus.[4]
For A. terreus the itaconate pathway is mostly elucidated. The generally accepted route for itaconate is via glycolysis, tricarboxylic acid cycle, and a decarboxylation of cis-aconitate to itaconate via cis-aconitate-decarboxylase.[5]
The smut fungus Ustilago maydis uses an alternative route. Cis-aconitate is converted to the thermodynamically favoured trans-aconitate via aconitate-Δ-isomerase (Adi1).[6] trans-Aconitate is further decarboxylated to itaconate by trans-aconitate-decarboxylase (Tad1).[6]
Itaconic acid is also produced in cells of macrophage lineage and as such it has in vitro activity against bacteria expressing the enzyme isocitrate lyase such as Salmonella enterica and Mycobacterium tuberculosis.[7]
However, cells of macrophage lineage have to "pay the price" for making itaconate, and they lose the ability to perform mitochondrial substrate-level phosphorylation,[8]
Application
Itaconic acid is primarily used as a co-monomer in the production of acrylonitrile butadiene styrene and acrylate latexes with applications in the paper and architectural coating industry.
References
- 1 2 3 4 5 Merck Index, 11th Edition, 5130
- ↑ R. L. Shriner; S. G. Ford; l. J. Roll (1931). "Itaconic anhydride and itaconic acid". Org. Synth. 11: 70. doi:10.15227/orgsyn.011.0070.
- ↑ R. L. Shriner; S. G. Ford; l. J. Roll (1931). "Citraconic Anhydride and Citraconic Acid". Org. Synth. 28: 28. doi:10.15227/orgsyn.011.0028.
- ↑ Roger A. Sheldon (2014). "Green and sustainable manufacture of chemicals from biomass: state of the art". Green Chem. 16: 950–963. doi:10.1039/C3GC41935E.
- ↑ Steiger, Matthias Georg; Blumhoff, Marzena Lidia; Mattanovich, Diethard; Sauer, Michael (2013-01-01). "Biochemistry of microbial itaconic acid production". Microbial Physiology and Metabolism. 4: 23. PMC 3572532
 . PMID 23420787. doi:10.3389/fmicb.2013.00023.
. PMID 23420787. doi:10.3389/fmicb.2013.00023. - 1 2 Geiser, Elena; Przybilla, Sandra K; Friedrich, Alexandra; Buckel, Wolfgang; Wierckx, Nick; Blank, Lars M; Bölker, Michael (2016-01-01). "Ustilago maydis produces itaconic acid via the unusual intermediate trans-aconitate". Microbial Biotechnology. 9 (1): 116–126. ISSN 1751-7915. PMC 4720413
 . PMID 26639528. doi:10.1111/1751-7915.12329.
. PMID 26639528. doi:10.1111/1751-7915.12329. - ↑ Michelucci, A.; Cordes, T.; Ghelfi, J.; Pailot, A.; Reiling, N.; Goldmann, O.; Binz, T.; Wegner, A.; Tallam, A.; Rausell, A.; Buttini, M.; Linster, C. L.; Medina, E.; Balling, R.; Hiller, K. (2013). "Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production". Proceedings of the National Academy of Sciences. 110 (19): 7820–5. Bibcode:2013PNAS..110.7820M. PMC 3651434
 . PMID 23610393. doi:10.1073/pnas.1218599110.
. PMID 23610393. doi:10.1073/pnas.1218599110. - ↑ Nemeth, B.; Doczi, J.; Csete, D.; Kacso, G.; Ravasz, D.; Adams, D.; Kiss, G.; Nagy, A. M.; Horvath, G.; Tretter, L.; Mocsai, A.; Csepanyi-Komi, R.; Iordanov, I.; Adam-Vizi, V.; Chinopoulos, C. (2015). "Abolition of mitochondrial substrate-level phosphorylation by itaconic acid produced by LPS-induced Irg1 expression in cells of murine macrophage lineage". The FASEB Journal. PMID 26358042. doi:10.1096/fj.15-279398.