Macular degeneration
| Macular degeneration | |
|---|---|
 Picture of the back of the eye showing intermediate age-related macular degeneration | |
| Classification and external resources | |
| Specialty | Ophthalmology |
| ICD-10 | H35.3 |
| ICD-9-CM | 362.50 |
| DiseasesDB | 11948 |
| MedlinePlus | 001000 |
| eMedicine | article/1223154 |
| Patient UK | Macular degeneration |
| MeSH | D008268 |
Macular degeneration, also known as age-related macular degeneration (AMD or ARMD), is a medical condition which may result in blurred or no vision in the center of the visual field.[1] Early on there are often no symptoms. Over time, however, some people experience a gradual worsening of vision that may affect one or both eyes. While it does not result in complete blindness, loss of central vision can make it hard to recognize faces, drive, read, or perform other activities of daily life. Visual hallucinations may also occur but these are not serious and do not represent a mental illness.[1]
Macular degeneration typically occur in older people. Genetic factors and smoking also play a role. It is due to damage to the macula of the retina. Diagnosis is by a complete eye exam. The severity is divided into early, intermediate, and late types.[1] The late type is additionally divided into "dry" and "wet" forms with the dry form making up 90% of cases.[1][2]
Prevention includes not smoking, exercising, and eating well.[1] Vitamin supplements do not appear to be useful for prevention.[3] There is no cure or treatment that returns vision already lost. In the wet form, anti-VEGF medication injected into the eye or less commonly laser coagulation or photodynamic therapy may slow worsening.[1] Supplements in those who already have the disease may slow progression.[4]
In 2010 it affected 23.5 million people globally.[5] In 2013 moderate to severe disease affected 13.4 million and it is the fourth most common cause of blindness after cataracts, preterm birth, and glaucoma.[6] It most commonly occurs in people over the age of fifty and in the United States is the most common cause of vision loss in this age group.[1][2] About 0.4% of people between 50 and 60 have the disease, while it occurs in 0.7% of people 60 to 70, 2.3% of those 70 to 80, and nearly 12% of people over 80 years old.[2]
Signs and symptoms


Signs and symptoms of macular degeneration include:
Visual symptoms
- Distorted vision in the form of metamorphopsia, in which a grid of straight lines appears wavy and parts of the grid may appear blank: Patients often first notice this when looking at things like miniblinds in their home or telephone poles while driving. There may also be central scotomas, shadows or missing areas of vision
- Slow recovery of visual function after exposure to bright light (photostress test)
- Visual acuity drastically decreasing (two levels or more), e.g.: 20/20 to 20/80
- Blurred vision: Those with nonexudative macular degeneration may be asymptomatic or notice a gradual loss of central vision, whereas those with exudative macular degeneration often notice a rapid onset of vision loss (often caused by leakage and bleeding of abnormal blood vessels).
- Trouble discerning colors, specifically dark ones from dark ones and light ones from light ones
- A loss in contrast sensitivity
Macular degeneration by itself will not lead to total blindness. For that matter, only a very small number of people with visual impairment are totally blind. In almost all cases, some vision remains, mainly peripheral. Other complicating conditions may possibly lead to such an acute condition (severe stroke or trauma, untreated glaucoma, etc.), but few macular degeneration patients experience total visual loss.[7]
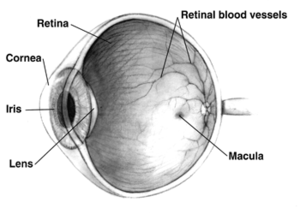
The area of the macula comprises only about 2.1% of the retina, and the remaining 97.9% (the peripheral field) remains unaffected by the disease. Even though the macula provides such a small fraction of the visual field, almost half of the visual cortex is devoted to processing macular information.[8]
The loss of central vision profoundly affects visual functioning. It is quite difficult, for example, to read without central vision. Pictures that attempt to depict the central visual loss of macular degeneration with a black spot do not really do justice to the devastating nature of the visual loss. This can be demonstrated by printing letters six inches high on a piece of paper and attempting to identify them while looking straight ahead and holding the paper slightly to the side. Most people find this difficult to do.
Risk factors
- Aging: Advanced age is the strongest predictor of AMD. .
- Family history:
Environment and lifestyle
- Smoking: Smoking tobacco increases the risk of AMD by two to three times that of someone who has never smoked, and may be the most important modifiable factor in its prevention. A review of previous studies found "a strong association between current smoking and AMD. ... Cigarette smoking is likely to have toxic effects on the retina."[9]
- Hypertension (high blood pressure): In the ALIENOR study 2013, early and late AMD were not significantly associated with systolic or diastolic BP, hypertension, or use of antihypertensive medications, but elevated pulse pressure ((PP) systolic BP minus diastolic BP) was significantly associated with an increased risk of late AMD.[10]
- Atherosclerosis:
- High cholesterol: Elevated cholesterol may increase the risk of AMD[11]
- Obesity: Abdominal obesity is a risk factor, especially among men[12]
- Fat intake: Consuming high amounts of certain fats including saturated fats, trans fats and omega-6 fatty acids likely contributes to AMD, while monounsaturated fats are potentially protective.[13] In particular, ω-3 fatty acids may decrease the risk of AMD.[14]
- Exposure to sunlight, especially blue light: Evidence is conflicting as to whether exposure to sunlight contributes to the development of macular degeneration. A recent study on 446 subjects found it does not.[15] Other research, however, has shown high-energy visible light may contribute to AMD.[16][17]
Genetics
Recurrence ratios for siblings of an affected individual are three- to sixfold higher than in the general population.[18] Genetic linkage analysis has identified 5 sets of gene variants at three locations on different chromosomes (1, 6 and 10) as explaining at least 50% of the risk. These genes have roles regulating immune response, inflammatory processes and homeostasis of the retina. Variants of these genes give rise to different kinds of dysfunction in these processes. Over time, this results in accumulation of intracellular and extracellular metabolic debris. This can cause scarring of the retina or breakdown of its vascularization.
Genetic tests are available for some of these gene variations. However, pathogenesis of macular degeneration is a complex interaction between genetics, environment and lifestyle, and presence of unfavorable genetic factors doesn't necessarily predict progression to disease. The three loci where identified gene variants are found are designated:
- Complement Factor H (CFH) on chromosome 1 at location 1q31.3
- HTRA serine peptidase 1/Age Related Maculopathy Susceptibility 2 (HTRA1/ARMS2) on chromosome 10 at location 10q26
- Complement Factor B/Complement Component 2 (CFB/CC2) on chromosome 6 at 6p21.3
Specific genes
- Polymorphisms in genes for complement system proteins: The genes for the complement system proteins factor H (CFH), factor B (CFB) and factor 3 (C3) are strongly associated with a person's risk for developing AMD. CFH is involved in inhibiting the inflammatory response. The mutation in CFH (Y402H) results in reduced ability of CFH to regulate complement on critical surfaces such as the retina and leads to increased inflammatory response within the macula. Absence of the complement factor H-related genes R3 and R1 protects against AMD.[19][20] Two independent studies in 2007 showed a certain common mutation Arg80Gly in the C3 gene, which is a central protein of the complement system, is strongly associated with the occurrence of AMD.[21][22] The authors of both papers consider their study to underscore the influence of the complement pathway in the pathogenesis of this disease.
- In two 2006 studies, another gene that has implications for the disease, called HTRA1 (encoding a secreted serine protease), was identified.[23][24]
- Six mutations of the gene SERPING1 (Serpin Peptidase Inhibitor, Clade G (C1 Inhibitor), Member 1) are associated with AMD. Mutations in this gene can also cause hereditary angioedema.[25]
- Fibulin-5 mutation: Rare forms of the disease are caused by genetic defects in fibulin-5, in an autosomal dominant manner. In 2004, Stone et al. performed a screen on 402 AMD patients and revealed a statistically significant correlation between mutations in fibulin-5 and incidence of the disease.
Mitochondrial related gene polymorphisms
such as that in the MT-ND2 molecule, predicts wet AMD.[26][27]
Pathophysiology
The pathogenesis of age-related macular degeneration is not well known, although a number of theories have been put forward, including oxidative stress, mitochondrial dysfunction, and inflammatory processes.
The imbalance between production of damaged cellular components and degradation leads to the accumulation of detrimental products, for example, intracellular lipofuscin and extracellular drusen. Incipient atrophy is demarcated by areas of RPE thinning or depigmentation that precede geographic atrophy in the early stages of AMD. In advanced stages of AMD, atrophy of the RPE (geographic atrophy) and/or development of new blood vessels (neovascularization) result in death of photoreceptors and central vision loss.
In the dry (nonexudative) form, cellular debris called drusen accumulates between the retina and the choroid, causing atrophy and scarring to the retina. In the wet (exudative) form, which is more severe, blood vessels grow up from the choroid (neovascularization) behind the retina which can leak exudate and fluid and also cause hemorrhaging.
Early work demonstrated a family of immune mediators was plentiful in drusen.[28] Complement factor H (CFH) is an important inhibitor of this inflammatory cascade, and a disease-associated polymorphism in the CFH gene strongly associates with AMD.[29][30][31][32][33] Thus an AMD pathophysiological model of chronic low grade complement activation and inflammation in the macula has been advanced.[34][35] Lending credibility to this has been the discovery of disease-associated genetic polymorphisms in other elements of the complement cascade including complement component 3 (C3).[36]
A powerful predictor of AMD is found on chromosome 10q26 at LOC 387715. An insertion/deletion polymorphism at this site reduces expression of the ARMS2 gene though destabilization of its mRNA through deletion of the polyadenylation signal.[37] ARMS2 protein may localize to the mitochondria and participate in energy metabolism, though much remains to be discovered about its function.
Other gene markers of progression risk includes tissue inhibitor of metalloproteinase 3 (TIMP3), suggesting a role for intracellular matrix metabolism in AMD progression.[38] Variations in cholesterol metabolising genes such as the hepatic lipase, cholesterol ester transferase, lipoprotein lipase and the ABC-binding cassette A1 correlate with disease progression. The early stigmata of disease, drusen, are rich in cholesterol, offering face validity to the results of genome-wide association studies.[39]
Dry AMD
Dry AMD begins with characteristic yellow deposits (drusen) in the macula, between the retinal pigment epithelium and the underlying choroid. Most people with these early changes (referred to as age-related maculopathy) still have good vision. People with drusen may or may not develop AMD, in fact the majority of people over age 55 have drusen with no negative effects. The risk of developing symptoms is higher when the drusen are large and numerous and associated with disturbance in the pigmented cell layer under the macula. Large and soft drusen are thought to be related to elevated cholesterol deposits. [progression to geographic atrophy] Central geographic atrophy, the "dry" form of advanced AMD, results from atrophy of the retinal pigment epithelial layer below the retina, which causes vision loss through loss of photoreceptors (rods and cones) in the central part of the eye.
Wet AMD
Neovascular or exudative AMD, the "wet" form of advanced AMD, causes vision loss due to abnormal blood vessel growth (choroidal neovascularization) in the choriocapillaris, through Bruch's membrane. The proliferation of abnormal blood vessels in the retina is stimulated by vascular endothelial growth factor (VEGF). Unfortunately, these new vessels are fragile, ultimately leading to blood and protein leakage below the macula. Bleeding, leaking, and scarring from these blood vessels eventually cause irreversible damage to the photoreceptors and rapid vision loss if left untreated.
Oxidative stress
Age-related accumulation of low-molecular-weight, phototoxic, pro-oxidant melanin oligomers within lysosomes in the retinal pigment epithelium (RPE) may be partly responsible for decreasing the digestive rate of photoreceptor outer rod segments (POS) by the RPE - autophagy. A decrease in the digestive rate of POS has been shown to be associated with lipofuscin formation - a classic sign associated with AMD.[40][41]
The role of retinal oxidative stress in the etiology of AMD by causing further inflammation of the macula is suggested by the enhanced rate of disease in smokers and those exposed to UV irradiation.[42][43][44]
Mitochondrial dysfunction may play a role.[45]
Diagnosis

Diagnosis of age-related macular degeneration rests on signs in the macula, irrespective of visual acuity. Diagnosis of AMD may include the following procedures and tests:
- There is a loss of contrast sensitivity, so that contours, shadows, and color vision are less vivid. The loss in contrast sensitivity can be quickly and easily measured by a contrast sensitivity test like Pelli Robson performed either at home or by an eye specialist.
- When viewing an Amsler grid, some straight lines appear wavy and some patches appear blank
- When viewing a Snellen chart, at least 2 lines decline
- Preferential hyperacuity perimetry changes (for wet AMD)[46][47]
- In dry macular degeneration, which occurs in 85-90 percent of AMD cases, drusen spots can be seen in Fundus photography
- In wet macular degeneration, using angiography we can see leakage of bloodstream behind the macula. Fluorescein angiography allows for the identification and localization of abnormal vascular processes.
- Using an electroretinogram, points in the macula with a weak or absent response compared to a normal eye may be found
- Farnsworth-Munsell 100 hue test and Maximum Color Contrast Sensitivity test (MCCS) for assessing color acuity and color contrast sensitivity
- Optical coherence tomography is now used by most ophthalmologists in the diagnosis and the follow-up evaluation of the response to treatment with antiangiogenic drugs.
Histology
- Pigmentary changes in the retina - In addition to the pigmented cells in the iris (the colored part of the eye), there are pigmented cells beneath the retina. As these cells break down and release their pigment, dark clumps of released pigment and later, areas that are less pigmented may appear
- Exudative changes: hemorrhages in the eye, hard exudates, subretinal/sub-RPE/intraretinal fluid
- Drusen, tiny accumulations of extracellular material that build up on the retina. While there is a tendency for drusen to be blamed for the progressive loss of vision, drusen deposits can be present in the retina without vision loss. Some patients with large deposits of drusen have normal visual acuity. If normal retinal reception and image transmission are sometimes possible in a retina when high concentrations of drusen are present, then, even if drusen can be implicated in the loss of visual function, there must be at least one other factor that accounts for the loss of vision.
Prevention
A 2012 Cochrane review found the use of vitamin and mineral supplements, alone or in combination, by the general population had no effect on whether or not AMD started.[3]
Management
Supplements may slow down the worsening of AMD.[4] There is not enough evidence to determine if statins have a role in preventing or slowing the progression of AMD.[48] Antiangiogenic steroids such as anecortave acetate and triamcinolone acetonide have shown no evidence in preventing visual loss in people with neovascular AMD.[49]
Dry AMD
No medical or surgical treatment is available for this condition.
Wet AMD
It can be treated with laser coagulation, and more commonly with medication that stops and sometimes reverses the growth of blood vessels.[50][51] There is strong evidence that laser coagulation will result in the disappearance of drusens but does not affect choroidal neovascularisation.[52]
A randomized control trial found that bevacizumab and ranibizumab had similar efficacy, and reported no significant increase in adverse events with bevacizumab.[53] A 2014 Cochrane review found that the systemic safety of bevacizumab and ranibizumab are similar when used to treat neovascular AMD, except for gastrointestinal disorders.[54] Bevacizumab however is not FDA approved for treatment of macular degeneration. A controversy in the UK involved the off-label use of cheaper bevacizumab over the approved, but expensive, ranibizumab.[55] Ranibizumab is a smaller fragment, Fab fragment, of the parent bevacizumab molecule specifically designed for eye injections. Other approved antiangiogenic drugs for the treatment of neo-vascular AMD include pegaptanib[56] and aflibercept.[57]
Laser coagulation is feasible in only limited cases. Only about 15 out of 100 cases can be effectively treated with laser photocoagulation surgery. The surgery works best when the abnormal blood vessels (choroidal neovascularization) are clustered close together in a specific area. Blood vessels that are scattered over a wider area are much harder to treat. Surgery is also less helpful after the abnormal blood vessels reach the center of the macula (fovea). The procedure cannot restore lost vision, only slow progression of the disease. Laser treatment almost always causes some immediate, permanent central vision loss (a central blind spot). And laser treatment does not always prevent future growth of abnormal blood vessels.
Photodynamic therapy has also been used to treat wet AMD.[58] The drug verteporfin is administered intravenously; light of a certain wavelength is then applied to the abnormal blood vessels. This activates the verteporfin destroying the vessels.
Cataract surgery could possibly improve visual outcomes for people with AMD, though there have been concerns of surgery increasing the progression of AMD. A randomized controlled trial found that people who underwent immediate cataract surgery (within 2 weeks) had improved visual acuity and better quality of life outcomes than those who underwent delayed cataract surgery (6 months).[59]
Adaptive devices
Because peripheral vision is not affected, people with macular degeneration can learn to use their remaining vision to partially compensate.[60] Assistance and resources are available in many countries and every state in the U.S.[61] Classes for "independent living" are given and some technology can be obtained from a state department of rehabilitation.
Adaptive devices can help people read. These include magnifying glasses, special eyeglass lenses, computer screen readers, and TV systems that enlarge reading material.
Computer screen readers such as JAWS or Thunder work with standard Windows computers. Also Apple devices provide wide range of features (voice over,screen readers, Braille etc.,
Video cameras can be fed into standard or special-purpose computer monitors, and the image can be zoomed in and magnified. These systems often include a movable table to move the written material.
Accessible publishing provides larger fonts for printed books, patterns to make tracking easier, audiobooks and DAISY books with both text and audio.
Epidemiology
_world_map_-_DALY_-_WHO2004.svg.png)
Age-related macular degeneration accounts for more than 54% of all vision loss in the white population in the USA.1 An estimated 8 million Americans are affected with early age-related macular degeneration, of whom over 1 million will develop advanced age-related macular degeneration within the next 5 years.14 In the UK, age-related macular degeneration is the cause of blindness in almost 42% of those who go blind aged 65–74 years, almost two-thirds of those aged 75–84 years, and almost three-quarters of those aged 85 years or older.15
Data pooled from three population-based studies—the Beaver Dam Eye Study, the Rotterdam Study, and the Blue Mountains Eye Study—have estimated the prevalence of advanced age-related macular degeneration to be 0·2% in those aged 55–64 years, increasing to 13% in those older than 85 years.16 These three large population-based studies have found that the incidence of advanced age-related macular degeneration increases with age, as does the development of large drusen and pigmentary changes. The Blue Mountains Eye Study estimated the overall 5-year incidence of advanced age-related macular degeneration to be 1·1%; increasing from 0% in individuals under 60 years of age to 5·4% in those aged 80 years or older. The overall 5-year incidence of the development of large drusen or pigmentary changes was 8·7%, ranging from 3·2% in those aged less than 60 years to 18·3% in those aged 70–79 years and 14·8% in those older than 80 years.17,18 The 15-year incidence of neovascular macular degeneration in the Beaver Dam Eye Study ranged from 0·4% in individuals under 55 years of age to 4·4% in those older than 75 years; the 15-year incidence of soft indistinct drusen ranged from 4·7% to 16·3% in the same age-groups.19 The 5-year progression rate to advanced age-related macular degeneration in individuals who had intermediate age-related macular degeneration and who were 80 years or older was 42% in the Rotterdam Study.20 The 5-year progression rate to advanced age-related macular degeneration in another population-based study—the Visual Impairment Project from Melbourne, Australia—ranged from 0% in people aged 60 years and younger to 6·3% in those 80 years and older.21
Macular degeneration is more likely to be found in Caucasians than in people of African descent.[63][64]
Research directions
Association with other age-related diseases
Studies indicate drusen associated with AMD are similar in molecular composition to Beta-Amyloid (βA) plaques and deposits in other age-related diseases such as Alzheimer's disease and atherosclerosis. This suggests that similar pathways may be involved in the etiologies of AMD and other age-related diseases.[65]
Genetic testing
A practical application of AMD-associated genetic markers is in the prediction of progression of AMD from early stages of the disease to neovascularization.[66][67]
Stem cell transplant
Cell based therapies using bone marrow stem cells as well as Retinal pigment epithelial transplantation are being studied.[68] Recent advancements within the field of stem cell research in the United States have led to the first human embryonic stem cell trial for dry AMD, which reports positive results.[69]
Other types
There are a few other (rare) kinds of macular degeneration with similar symptoms but unrelated in etiology to Wet or Dry age-related macular degeneration. They are all genetic disorders that may occur in childhood or middle age.
- Best's disease
- Sorsby's fundus dystrophy is an autosomal dominant, retinal disease characterized by sudden acuity loss resulting from untreatable submacular neovascularisation
- Stargardt's disease (juvenile macular degeneration, STGD) is an autosomal recessive retinal disorder characterized by a juvenile-onset macular dystrophy, alterations of the peripheral retina, and subretinal deposition of lipofuscin-like material.
Similar symptoms with a very different etiology and different treatment can be caused by epiretinal membrane or macular pucker or any other condition affecting the macula, such as central serous retinopathy.
Notable cases
- Judi Dench[70]
- Joan Plowright[71]
- Peter Sallis,[72] notable of voices of Wallace from Wallace and Gromit until 2012
- Rosanne Barr[73]
See also
References
- 1 2 3 4 5 6 7 "Facts About Age-Related Macular Degeneration". National Eye Institute. June 2015. Retrieved 21 December 2015.
- 1 2 3 Mehta, S (September 2015). "Age-Related Macular Degeneration.". Primary care 42 (3): 377–91. doi:10.1016/j.pop.2015.05.009. PMID 26319344.
- 1 2 Evans JR, Lawrenson JG (2012). Evans, Jennifer R, ed. "Antioxidant vitamin and mineral supplements for preventing age-related macular degeneration". Cochrane Database Syst Rev 6: CD000253. doi:10.1002/14651858.CD000253.pub3. PMID 22696317.
- 1 2 Evans, JR; Lawrenson, JG (14 November 2012). "Antioxidant vitamin and mineral supplements for slowing the progression of age-related macular degeneration.". The Cochrane database of systematic reviews 11: CD000254. doi:10.1002/14651858.CD000254.pub3. PMID 23152201.
- ↑ Velez-Montoya, R; Oliver, SC; Olson, JL; Fine, SL; Quiroz-Mercado, H; Mandava, N (March 2014). "Current knowledge and trends in age-related macular degeneration: genetics, epidemiology, and prevention.". Retina (Philadelphia, Pa.) 34 (3): 423–41. doi:10.1097/iae.0000000000000036. PMID 24285245.
- ↑ Global Burden of Disease Study 2013, Collaborators (22 August 2015). "Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013.". Lancet (London, England) 386 (9995): 743–800. doi:10.1016/s0140-6736(15)60692-4. PMID 26063472.
- ↑ Roberts, DL (September 2006). "The First Year--Age Related Macular Degeneration". (Marlowe & Company): 100.
- ↑ Roberts, DL (September 2006). "The First Year--Age Related Macular Degeneration". (Marlowe & Company): 20.
- ↑ Thornton, J; Edwards, R; Mitchell, P; Harrison, RA; Buchan, I; Kelly, SP (September 2005). "Smoking and age-related macular degeneration: a review of association.". Eye (London, England) 19 (9): 935–44. doi:10.1038/sj.eye.6701978. PMID 16151432.
- ↑ Cougnard-Grégoire, A; et al. (March 2013). "Long-term blood pressure and age-related macular degeneration: the ALIENOR study". Invest Ophthalmol Vis Sci. 54 (3): 1905–12. doi:10.1167/iovs.12-10192. PMID 23404120.
- ↑ Dasari, Bhanu; Prasanthi, Jaya RP; Marwarha, Gurdeep; Singh, Brij B; Ghribi, Othman (18 August 2011). "Cholesterol-enriched diet causes age-related macular degeneration-like pathology in rabbit retina". BMC Ophthalmology 11: 22. doi:10.1186/1471-2415-11-22. PMC 3170645. PMID 21851605.

- ↑ Adams MK, Simpson JA, Aung KZ, et al., Adams MK, Simpson JA, Aung KZ, Makeyeva GA, Giles GG, English DR, Hopper J, Guymer RH, Baird PN, Robman LD. (1 June 2011). "Abdominal obesity and age-related macular degeneration". Am J Epidemiol. 173 (11): 1246–55. doi:10.1093/aje/kwr005. PMID 21422060. Retrieved 29 July 2012. Cite uses deprecated parameter
|coauthors=(help) - ↑ Parekh N, Voland RP, Moeller SM, et al., Parekh N, Voland RP, Moeller SM, Blodi BA, Ritenbaugh C, Chappell RJ, Wallace RB, Mares JA; CAREDS Research Study Group. (November 2009). "Association between dietary fat intake and age-related macular degeneration in the Carotenoids in Age-Related Eye Disease Study (CAREDS): an ancillary study of the Women's Health Initiative". Arch Ophthalmol. 127 (11): 1483–93. doi:10.1001/archophthalmol.2009.130. PMC 3144752. PMID 19901214. Cite uses deprecated parameter
|coauthors=(help) - ↑ John Paul SanGiovanni, ScD; Emily Y. Chew, MD; Traci E. Clemons, PhD; Matthew D. Davis, MD; Frederick L. Ferris III, MD; Gary R. Gensler, MS; Natalie Kurinij, PhD; Anne S. Lindblad, PhD; Roy C. Milton, PhD; Johanna M. Seddon, MD; and Robert D. Sperduto, MD (May 5, 2007). "The Relationship of Dietary Lipid Intake and Age-Related Macular Degeneration in a Case-Control Study". Archives of Ophthalmology.
- ↑ Khan, JC; Shahid H, Thurlby DA, Bradley M, Clayton DG, Moore AT, Bird AC, Yates JR, Genetic Factors in AMD Study (January 2006). "Age related macular degeneration and sun exposure, iris colour, and skin sensitivity to sunlight". The British Journal of Ophthalmology 90 (1): 29–32. doi:10.1136/bjo.2005.073825. PMC 1856929. PMID 16361662. Cite uses deprecated parameter
|coauthors=(help) - ↑ Glazer-Hockstein, C; Dunaief JL (January 2006). "Could blue light-blocking lenses decrease the risk of age-related macular degeneration?". Retina 26 (1): 1–4. doi:10.1097/00006982-200601000-00001. PMID 16395131.
- ↑ Margrain, TH; Boulton M; Marshall J; Sliney DH (September 2004). "Do blue light filters confer protection against age-related macular degeneration?". Progress in Retinal and Eye Research 23 (5): 523–31. doi:10.1016/j.preteyeres.2004.05.001. PMID 15302349.
- ↑ Maller, J; et al. (September 2006). "Common variation in three genes, including a noncoding variant in CFH, strongly influences risk of age-related macular degeneration.". Nat. Genet. 38 (9): 1055–9. doi:10.1038/ng1873. PMID 16936732.
- ↑ Hughes, Anne E; Orr, Nick; Esfandiary, Hossein; Diaz-Torres, Martha; Goodship, Timothy; Chakravarthy, Usha (2006). "A common CFH haplotype, with deletion of CFHR1 and CFHR3, is associated with lower risk of age-related macular degeneration". Nature Genetics 38 (10): 1173–1177. doi:10.1038/ng1890. PMID 16998489.
- ↑ Fritsche, L. G.; Lauer, N.; Hartmann, A.; Stippa, S.; et al. (2010). "An imbalance of human complement regulatory proteins CFHR1, CFHR3 and factor H influences risk for age-related macular degeneration (AMD)". Human Molecular Genetics 19 (23): 4694–4704. doi:10.1093/hmg/ddq399. PMID 20843825.
- ↑ Yates JR, Sepp T, Matharu BK, Khan JC, Thurlby DA, Shahid H, Clayton DG, Hayward C, Morgan J, Wright AF, Armbrecht AM, Dhillon B, Deary IJ, Redmond E, Bird AC, Moore AT (2007). "Complement C3 Variant and the Risk of Age-Related Macular Degeneration". N Engl J Med. 357 (6): 553–561. doi:10.1056/NEJMoa072618. PMID 17634448.
- ↑ Maller JB, Fagerness JA, Reynolds RC, Neale BM, Daly MJ, Seddon JM (2007). "Variation in Complement Factor 3 is Associated with Risk of Age-Related Macular Degeneration". Nature Genetics 39 (10): 1200–1201. doi:10.1038/ng2131. PMID 17767156.
- ↑ Yang Z, Camp NJ, Sun H, Tong Z, Gibbs D, Cameron DJ, Chen H, Zhao Y, Pearson E; et al. (Nov 2006). "A variant of the HTRA1 gene increases susceptibility to age-related macular degeneration". Science 314 (5801): 992–3. doi:10.1126/science.1133811. PMID 17053109.
- ↑ Dewan A, Liu M, Hartman S, et al. (November 2006). "HTRA1 Promoter Polymorphism in Wet Age-Related Macular Degeneration". Science 314 (5801): 989–92. doi:10.1126/science.1133807. PMID 17053108.
- ↑ Hirschler, Ben (2008-10-07). "Gene discovery may help hunt for blindness cure". Reuters. Retrieved 2008-10-07.
- ↑ Udar N, Atilano SR, Memarzadeh M, Boyer D, Chwa M, Lu S (2009). "Mitochondrial DNA Haplogroups Associated with Age-Related Macular Degeneration". Invest Ophthalmol Vis Sci 50 (6): 2966–74. doi:10.1167/iovs.08-2646. PMID 19151382.
- ↑ Canter JA, Olson LM, Spencer K, Schnetz-Boutaud N, Anderson B, Hauser MA (2008). Nicholas Weedon, Michael, ed. "Mitochondrial DNA polymorphism A4917G is independently associated with age-related macular degeneration". PLoS ONE 3 (5): e2091. doi:10.1371/journal.pone.0002091. PMC 2330085. PMID 18461138.

- ↑ Mullins RF, Russell SR, Anderson DH, Hageman GS (2000). "Drusen associated with aging and age-related macular degeneration contain proteins common to extracellular deposits associated with atherosclerosis, elastosis, amyloidosis, and dense deposit disease". FASEB J 14 (7): 835–46. PMID 10783137.
- ↑ Hageman GS, Anderson DH, Johnson LV, Hancox LS, Taiber AJ, Hardisty LI (2005). "A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration". Proc Natl Acad Sci USA 102 (20): 7227–32. doi:10.1073/pnas.0501536102. PMC 1088171. PMID 15870199.
- ↑ Chen LJ, Liu DT, Tam PO, Chan WM, Liu K, Chong KK (2006). "Association of complement factor H polymorphisms with exudative age-related macular degeneration". Mol. Vis 12: 1536–42. PMID 17167412.
- ↑ Despriet DD, Klaver CC, Witteman JC, Bergen AA, Kardys I, de Maat MP (2006). "Complement factor H polymorphism, complement activators, and risk of age-related macular degeneration". JAMA 296 (3): 301–9. doi:10.1001/jama.296.3.301. PMID 16849663.
- ↑ Li M, Tmaca-Sonmez P, Othman M, Branham KE, Khanna R, Wade MS (2006). "CFH haplotypes without the Y402H coding variant show strong association with susceptibility to age-related macular degeneration". Nature Genetics 38 (9): 1049–54. doi:10.1038/ng1871. PMC 1941700. PMID 16936733.
- ↑ Haines JL, Hauser MA, Schmidt S, Scott WK, Olson LM, Gallins P (2005). "Complement factor H variant increases the risk of age-related macular degeneration". Science 308 (5720): 419–21. doi:10.1126/science.1110359. PMID 15761120.
- ↑ Rohrer B, Long Q, Coughlin B, Renner B, Huang Y, Kunchithapautham K (2010). "A targeted inhibitor of the complement alternative pathway reduces RPE injury and angiogenesis in models of age-related macular degeneration". Adv Exp Med Biol. Advances in Experimental Medicine and Biology 703: 137–49. doi:10.1007/978-1-4419-5635-4_10. ISBN 978-1-4419-5634-7. PMID 20711712.
- ↑ Kunchithapautham K, Rohrer B (May 2011). "Sublytic Membrane-Attack-Complex (MAC) Activation Alters Regulated Rather than Constitutive Vascular Endothelial Growth Factor (VEGF) Secretion in Retinal Pigment Epithelium Monolayers". J Biol Chem 286 (27): 23717–23724. doi:10.1074/jbc.M110.214593. PMC 3129152. PMID 21566137.
- ↑ Yates JR, Sepp T, Matharu BK, Khan JC, Thurlby DA, Shahid H (2007). "Complement C3 variant and the risk of age-related macular degeneration". NEJM 357 (6): 553–61. doi:10.1056/NEJMoa072618. PMID 17634448.
- ↑ Fritsche LG, Loenhardt T, Janssen A, Fisher SA, Rivera A, Keilhauer CN (2008). "Age-related macular degeneration is associated with an unstable ARMS2 (LOC387715) mRNA DNA damage and repair in age-related macular degeneration". Nat. Genet. 40 (7): 892–896. doi:10.1038/ng.170.
- ↑ Chen W, Stambolian D, Edwards AO, Branham KE, Othman M, Jakobsdottir J (2010). "Genetic variants near TIMP3 and high-density lipoprotein–associated loci influence susceptibility to age-related macular degeneration". Proc Natl Acad Sci U S A 107 (16): 7401–6. doi:10.1073/pnas.0912702107. PMC 2867722. PMID 20385819.
- ↑ Neale BM, Fagerness J, Reynolds R, Sobrin L, Parker M, Raychaudhuri S (2010). "Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC)". Proc Natl Acad Sci U S A. 107 (16): 7395–400. doi:10.1073/pnas.0912019107. PMC 2867697. PMID 20385826.
- ↑ "Melanin aggregation and polymerization: possible implications in age related macular degeneration." Ophthalmic Research, 2005; volume 37: pages 136-141.
- ↑ John Lacey, "Harvard Medical signs agreement with Merck to develop potential therapy for macular degeneration", 23-May-2006
- ↑ Thornton J, Edwards R, Mitchell P, Harrison RA, Buchan I, Kelly SP (2005). "Smoking and age-related macular degeneration: a review of association". Eye 19 (9): 935–44. doi:10.1038/sj.eye.6701978. PMID 16151432.
- ↑ Tomany SC, Cruickshanks KJ, Klein R, Klein BE, Knudtson MD (2004). "Sunlight and the 10-year incidence of age-related maculopathy: the Beaver Dam Eye Study". Arch Ophthalmol 122 (5): 750–7. doi:10.1001/archopht.122.5.750. PMID 15136324.
- ↑ Szaflik JP, Janik-Papis K, Synowiec E, Ksiazek D, Zaras M, Wozniak K (2009). "DNA damage and repair in age-related macular degeneration". Mutat Res 669 (1–2): 167–176. doi:10.1016/j.mrfmmm.2009.06.008.
- ↑ Barot, M; Gokulgandhi, MR; Mitra, AK (December 2011). "Mitochondrial dysfunction in retinal diseases". Current Eye Research 36 (12): 1069–77. doi:10.3109/02713683.2011.607536. PMC 4516173. PMID 21978133.
- ↑
- ↑ "Preferential Hyperacuity Perimetry (PHP) as an Adjunct Diagnostic Tool to Funduscopy in Age–related Macular Degeneration - Ophthalmology Technology Spotlight". Medcompare. Retrieved 2011-01-11.
- ↑ Gehlbach, P; Li, T; Hatef, E (11 February 2015). "Statins for age-related macular degeneration.". The Cochrane database of systematic reviews 2: CD006927. doi:10.1002/14651858.CD006927.pub4. PMID 25675254.
- ↑ Geltzer A, Turalba A, Vedula SS (2013). "Surgical implantation of steroids with antiangiogenic characteristics for treating neovascular age-related macular degeneration". Cochrane Database Syst Rev 1: CD005022. doi:10.1002/14651858.CD005022.pub3. PMC 4269233. PMID 23440797.
- ↑ de Jong PT (2006). "Age-related macular degeneration". N Engl J Med. 355 (14): 1474–1485. doi:10.1056/NEJMra062326. PMID 17021323.
- ↑ Ch. 25, Disorders of the Eye, Jonathan C. Horton, in Harrison's Principles of Internal Medicine, 16th ed.
- ↑ Virgili G, Michelessi M, Parodi MB, Bacherini D, Evans JR (2015). "Laser treatment of drusen to prevent progression to advanced age-related macular degeneration". Cochrane Database Syst Rev 10: CD006537. doi:10.1002/14651858.CD006537.pub3. PMID 26493180.
- ↑ Chakravarthy, U; Harding, SP; Rogers, CA; Downes, SM; Lotery, AJ; Culliford, LA; Reeves, BC; on behalf of the IVAN study, investigators (Jul 18, 2013). "Alternative treatments to inhibit VEGF in age-related choroidal neovascularisation: 2-year findings of the IVAN randomised controlled trial.". Lancet 382 (9900): 1258–67. doi:10.1016/S0140-6736(13)61501-9. PMID 23870813.
- ↑ Moja, L; Lucenteforte, E; Kwag, KH; Bertele, V; Campomori, A; Chakravarthy, U; D'Amico, R; Dickersin, K; Kodjikian, L; Lindsley, K; Loke, Y; Maguire, M; Martin, DF; Mugelli, A; Mühlbauer, B; Püntmann, I; Reeves, B; Rogers, C; Schmucker, C; Subramanian, ML; Virgili, G (Sep 15, 2014). "Systemic safety of bevacizumab versus ranibizumab for neovascular age-related macular degeneration.". The Cochrane database of systematic reviews 9: CD011230. doi:10.1002/14651858.CD011230.pub2. PMC 4262120. PMID 25220133.
- ↑ Copley, Caroline; Hirschler, Ben (April 24, 2012). "Novartis challenges UK Avastin use in eye disease". Reuters.
- ↑ "FDA Approves New Drug Treatment for Age-Related Macular Degeneration". FDA.gov. U.S. Food and Drug Administration.
- ↑ FDA approves Eylea for macular degeneration
- ↑ "Clinical effectiveness and cost–utility of photodynamic therapy for wet age-related macular degeneration: a systematic review and economic evaluation". Health Technology Assessment 7 (9). 2003. doi:10.3310/hta7090.
- ↑ Casparis H, Lindsley K, Kuo IC, Sikder S, Bressler NM (2012). "Surgery for cataracts in people with age-related macular degeneration". Cochrane Database Syst Rev 6: CD006757. doi:10.1002/14651858.CD006757.pub3. PMID 22696359.
- ↑ "Low Vision Rehabilitation Delivery Model". Mdsupport.org. Retrieved 2011-01-11.
- ↑ "Agencies, Centers, Organizations, & Societies". Mdsupport.org. 2005-09-01. Retrieved 2011-01-11.
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2009. Retrieved Nov 11, 2009.
- ↑ Age-Related Eye Disease Study Research Group (Dec 2000). "Risk Factors Associated with Age-Related Macular Degeneration: A Case-control Study in the Age-Related Eye Disease Study: Age-Related Eye Disease Study Report Number 3". Ophthalmology 107 (12): 2224–32. doi:10.1016/S0161-6420(00)00409-7. PMC 1470467. PMID 11097601.
- ↑ Clemons TE, Milton RC, Klein R, Seddon JM, Ferris FL (April 2005). "Risk Factors for the Incidence of Advanced Age-Related Macular Degeneration in the Age-Related Eye Disease Study (AREDS) AREDS Report No. 19". Ophthalmology 112 (4): 533–9. doi:10.1016/j.ophtha.2004.10.047. PMC 1513667. PMID 15808240.
- ↑ Mullins, RF; et al. (May 2000). "Drusen associated with aging and age-related macular degeneration contain proteins common to extracellular deposits associated with atherosclerosis, elastosis, amyloidosis, and dense deposit disease". FASEB J 14 (7): 835–46. PMID 10783137.
- ↑ Chen W, Stambolian D, Edwards AO, Branham KE, Othman M, Jakobsdottir J (2010). "Genetic variants near TIMP3 and high-density lipoprotein–associated loci influence susceptibility to age-related macular degeneration". Proc Natl Acad Sci U S A 107 (16): 7401–7406. doi:10.1073/pnas.0912702107. PMC 2867722. PMID 20385819.
- ↑ Neale BM, Fagerness J, Reynolds R, Sobrin L, Parker M, Raychaudhuri S (2010). "Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC)". Proc Natl Acad Sci U S A 107 (16): 7395–7400. doi:10.1073/pnas.0912019107. PMC 2867697. PMID 20385826.
- ↑ John S, et al. (2013). "Choice of cell source in cell based therapies for retinal damage due to age related macular degeneration (AMD): A review". Journal of Ophthalmology 2013: 1–9. doi:10.1155/2013/465169.
- ↑ Lanza, R; Schwartz, SD (25 Feb 2012). "Embryonic stem cell trials for macular degeneration: a preliminary report". Lancet 379: 713–720. doi:10.1016/s0140-6736(12)60028-2.
- ↑ "Judi Dench 'can't read any more due to failing eye site", The Guardian, 23 February 2014
- ↑ "Joan bows out to a standing ovation", The Guardian, 13 May 2014
- ↑ "Patrons of the Macular Society", Macular Society
- ↑ "Roseanne Barr's blindness and how to prevent her diseases", CNN
External links
