Triethylenetetramine
 | |
 | |
 | |
| Names | |
|---|---|
| Other names
N,N'-Bis(2-aminoethyl)ethane-1,2-diamine | |
| Identifiers | |
| 112-24-3 | |
| 605448 | |
| ChEBI | CHEBI:39501 |
| ChEMBL | ChEMBL609 |
| ChemSpider | 21106175 |
| EC Number | 203-950-6 |
| 27008 | |
| Jmol interactive 3D | Image |
| KEGG | C07166 |
| MeSH | Trientine |
| PubChem | 5565 |
| RTECS number | YE6650000 |
| UNII | SJ76Y07H5F |
| UN number | 2259 |
| |
| |
| Properties | |
| C6H18N4 | |
| Molar mass | 146.24 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Ichtyal, ammoniacal |
| Density | 982 mg mL−1 |
| Melting point | −34.6 °C; −30.4 °F; 238.5 K |
| Boiling point | 266.6 °C; 511.8 °F; 539.7 K |
| Miscible | |
| log P | 1.985 |
| Vapor pressure | <1 Pa (at 20 °C) |
| Refractive index (nD) |
1.496 |
| Thermochemistry | |
| 376 J K−1 mol−1 (at 60 °C) | |
| Hazards | |
| GHS pictograms | 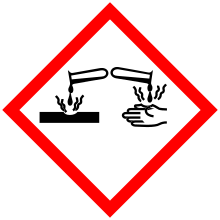  |
| GHS signal word | DANGER |
| H312, H314, H317, H412 | |
| P273, P280, P305+351+338, P310 | |
| EU classification (DSD) |
|
| R-phrases | R21, R34, R43, R52/53 |
| S-phrases | (S1/2), S26, S36/37/39, S45 |
| Flash point | 129 °C (264 °F; 402 K) |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose) |
|
| Related compounds | |
| Related amines |
|
| Related compounds |
|
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Triethylenetetramine, abbreviated TETA and trien, is an organic compound with the formula [CH2NHCH2CH2NH2]2. This oily liquid is colourless but, like many amines, assumes a yellowish color due to impurities resulting from air-oxidation. It is soluble in polar solvents and exhibits the reactivity typical for amines. The branched isomer N(CH2CH2NH2)3 and piperazine derivatives also comprise commercial samples of TETA.[1]
Production
Triethylenetetramine is prepared by heating ethylenediamine or ethanolamine/ammonia mixtures over an oxide catalyst. This process gives a variety of amines, which are separated by distillation and sublimation.[2]
Uses
The reactivity and uses of TETA are similar to those for the related polyamines ethylenediamine and diethylenetriamine. It was primarily used as a crosslinker ("hardener") in epoxy curing.[2]
The hydrochloride salt of triethylenetetramine, referred to as trientine hydrochloride, is a chelating agent, and is used to bind up and remove copper in the body to treat Wilson's disease, particularly in those who are intolerant to penicillamine. Some recommend trientine as first-line treatment, but experience with penicillamine is more extensive.[3]
Coordination chemistry
Triethylenetetramine is a tetradentate ligand in coordination chemistry, where it is referred to as trien.[4] Octahedral complexes of the type M(trien)Cl3 can adopt several diastereomeric structures, most of which are chiral.[5]
References
- ↑ http://www.huntsman.com/portal/page/portal/performance_products/Media%20Library/a_MC348531CFA3EA9A2E040EBCD2B6B7B06/Products_MC348531D0B9FA9A2E040EBCD2B6B7B06/Amines_MC348531D0BECA9A2E040EBCD2B6B7B06/Ethyleneamines_MC348531D0CD3A9A2E040EBCD2B6B7B06/files/ethyleneamines_brochure_huntsman_ethyleneamines.pdf
- 1 2 Eller, K.; Henkes, E.; Rossbacher, R.; Höke, H. (2005). "Amines, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_001.
- ↑ Roberts, E. A.; Schilsky, M. L. (2003). "A practice guideline on Wilson disease" (pdf). Hepatology 37 (6): 1475–1492. doi:10.1053/jhep.2003.50252. PMID 12774027.
- ↑ von Zelewsky, A. (1995). Stereochemistry of Coordination Compounds. Chichester: John Wiley. ISBN 047195599X.
- ↑ Utsuno, S.; Sakai, Y.; Yoshikawa, Y.; Yamatera, H. (1985). "Three Isomers of the Trans-Diammine-[N,N′-bis(2-Aminoethyl)-1,2-Ethanediamine]-Cobalt(III) Complex Cation". Inorganic Syntheses 23: 79–82. doi:10.1002/9780470132548.ch16.