Triclocarban
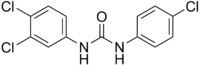 | |
| Names | |
|---|---|
| IUPAC name
3-(4-Chlorophenyl)-1-(3,4-dichlorophenyl)urea | |
| Other names
Trichlorocarbanilide, TCC, Solubacter | |
| Identifiers | |
| 101-20-2 | |
| ChEBI | CHEBI:48347 |
| ChEMBL | ChEMBL1076347 |
| ChemSpider | 7266 |
| Jmol interactive 3D | Image |
| PubChem | 7547 |
| UNII | BGG1Y1ED0Y |
| |
| |
| Properties | |
| C13H9Cl3N2O | |
| Molar mass | 315.58 g·mol−1 |
| Density | 1.53 g/cm3 |
| Melting point | 254 to 256 °C (489 to 493 °F; 527 to 529 K) |
| Hazards | |
| NFPA 704 | |
| Flash point | > 150 °C (302 °F; 423 K) |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose) |
>5000 mg/kg (oral, mouse)[1] 2100 mg/kg (i.p., mouse)[1] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Triclocarban is an antibacterial agent common in personal care products like soaps and lotions as well as in the medical field, for which it was originally developed.[2] Studies on its antibacterial qualities and mechanisms are growing. Research suggests that it is similar in its mechanism to triclosan and is effective in fighting infections by targeting the growth of bacteria such as Staphylococcus aureus. Additional research seeks to understand its potential for causing antibacterial resistance and its effects on organismal and environmental health.
Uses
Triclocarban has been used as an antimicrobial and antifungal compound since the 1960s.[3] It is commonly found in personal care products as antimicrobials such as in soaps, lotions, deodorants, toothpaste, and plastic.[4] About 80% of all antimicrobial bar soap sold in the United States contains triclocarban.[3] Additionally, the United States spends nearly 1 billion dollars annually on products containing triclocarban and triclosan.[5]
In December 2013, the Food and Drug Administration required all companies to prove within the next year, that triclocarban is not harmful to consumers. In recent years, companies like Johnson & Johnson, Procter & Gamble, Colgate-Palmolive, and Avon have begun phasing out chemical use due to health concerns.[6]
Chemical structure and properties
Triclocarban, 3-(4-chlorophenyl)-1-(3,4-dichlorophenyl)urea, is a white powder that is insoluble in water. While triclocarban has two chlorinated phenyl rings, it is structurally similar to carbanilide compounds often found in pesticides (such as diuron) and some drugs. Chlorination of ring structures is often associated with hydrophobicity, persistence in the environment, and bioaccumulation in fatty tissues of living organisms. For this reason, chlorine is also a common component of persistent organic pollutants.[7] Triclocarban is incompatible with strong oxidizing reagents and strong bases, reaction with which could result in safety concerns such as explosion, toxicity, gas, and heat.[8]
Synthesis of triclocarban
There are two commercial routes used for the production of triclocarban, using the reaction of isocyanates with nucleophiles such as amines to form ureas:[9]
- 4-chlorophenylisocyanate is reacted with 3,4-dichloroaniline
- 3,4-dichlorophenylisocyanate is reacted with 4-chloroaniline
The purity specification in the draft USP monograph for triclocarban is: not less than 97.0% w/w. The purity of commercial production is greater 98% w/w.[10]
Mechanism of action
Bacteria
Triclocarban is predominantly active against gram positive bacteria (bacteria with a thick peptidoglycan wall). The precise mechanism of action of triclocarban is unknown, but it is shown to be bacteriostatic, which prevents bacterial proliferation. Unlike other antibacterial compounds, triclocarban does not interfere with the membrane. As a result, it is hypothesized that triclocarban’s molecular mechanism resembles that of triclosan, which inhibits bacterial fatty acid synthesis.[3][11] By mimicking the natural substrate of the enoyl-acyl-carrier protein reductase (ENR) enzyme, triclosan acts as a site-directed inhibitor and disrupts lipid, phospholipid, lipopolysaccharide and lipoprotein synthesis. ENR is a highly conserved enzyme of lipid biosynthetic pathways in bacteria, notably Gram-negative, Gram-positive and mycobacterial species and thus is absent in humans.[12]
Humans
The specific mechanism of action for triclocarban's health effects on humans, like in bacteria, is unclear. Generally, triclocarban enhances the gene expression of other steroid hormones, including androgens, estrogens, and cortisol. It is hypothesized that the compound acts similar to cofactors or coactivators that modulate the activity of estrogen receptors and androgen receptors.[13][14] Experiments show that triclocarban activates constitutive androstane receptor and estrogen receptor alpha both in vivo and in vitro and might have the potential to alter normal physiological homeostasis. Activation of these receptors amplifies gene expression and, in doing so, may be the mechanistic base of triclocarban's health impact on humans. However, further investigation is needed to determine whether triclocarban increases the activity of sex steroid hormones by binding to the receptors or by binding to and sensitizing the receptor coactivators.[15][16]
Antibacterial properties
Triclocarban acts to effectively treat both initial bacterial skin and mucosal infections as well as those infections at risk for superinfection. In vitro, triclocarban has been found to be effective against various strains of staphylococcus, streptococcus, and enterococcus bacteria. It has been shown to be effective as an antibacterial even at very low levels. Triclocarban’s minimum inhibitory concentration has been found to range from 0.5 to 8 mg/L for these various strains.[17] Triclocarban is only unquestionably bacteriostatic for gram-positive bacteria such as Staphylococcus aureus, which suggests that the mechanism of triclocarban’s antibacterial activity is through its destabilization of bacterial cell walls.[3]
Resistance
Exposure of organisms like fish, algae, and humans to low levels of triclocarban and other antibacterial chemicals kills weak microbes and allows the stronger, resistant strains to proliferate. As microbes share genes, an increase in resistant strains increases the probability that weak microbes acquire these resistance genes. The consequence is a new colony of drug resistant microbes.[18]
When resistant microbes are exposed to antimicrobials, they increase their expression of genes that confer this resistance. The risk of bacterial antibiotic resistance has been studied by quantitatively monitoring the abundance of the tetQ gene in wastewater microcosms. As tetQ is the most common resistance gene in the environment and encodes for ribosomal protection proteins, the amount that it expresses correlates with the amount of resistance in a microbial population. The addition of triclocarban was shown to increase the expression of this tetQ gene.[18]
TetQ gene expression in bacteria was also found to be significantly increased when multiple antimicrobials such as tetracycline, triclosan, and triclocarban were added to an experimental system at the same time. Combining these compounds affects resistance by creating a situation where co-selection (or natural selection by more than one reagent) for resistance genes occurs. The complex nature of microbial communities and the multitude of antibiotics present in aquatic environments often leads to this sort of dynamic selection event and the multiple resistance patterns seen in naturally-occurring bacteria.[18]
Breakdown of product
When triclocarban is manufactured, 139 toxic, carcinogenic byproducts, such as 4-chloroaniline and 3,4-dichloroaniline, are released. More of these carcinogens can be released upon chemical, physical and biological attack of triclocarban.[19] The duration of triclocarban chemical in personal product use is relatively short. Upon disposal, the triclocarban is washed down the drain to municipal wastewater treatment plants, where about 97-98% of triclocarban is removed from the water.
Discharge of effluent from these treatment plants and disposal of sludge on land is the primary route of environmental exposure to triclocarban. Research shows that triclocarban and triclosan have been detected in sewage effluents and sludge (biosolids) due to their incomplete removal during wastewater treatment.[20] Due to their hydrophobic nature, significant amounts of them in wastewater streams partition into sludge, with concentrations at mg/kg levels. The volume of triclocarban reentering the environment in sewage sludge after initial successful capture from wastewater is s 127,000 ± 194,000 kg/yr. This is equivalent to a 4.8 – 48.2% of its total U.S consumption volume. Crops shown to take up antimicrobials from soil include barley, meadow fescue, carrots and pinto beans.[19]Studies show that substantial quantities of triclocarban (227,000 – 454,000 kg/y) can break through wastewater treatment plants and damage algae on surface waters.[19]
Environmental concerns
Waste water
High concentrations of triclocarban may be found in wastewater. It is among the top ten most commonly detected organic wastewater compounds in terms of frequency and concentration. Triclocarban has been found in increasing concentrations over the past five years and is now more frequently detected than triclosan.[4]
Wildlife toxicity
Triclocarban has a hazard quotient rating of greater than one. Hazard quotients greater than one indicate the potential for adverse effects on organisms due to toxicity.[4] As triclocarban is found in high concentrations in aquatic environments, there are concerns regarding its toxicity to aquatic species. Specifically, triclocarban has been shown to be toxic to amphibians, fish, invertebrates, and aquatic plants, and traces of the compound have been found in Atlantic dolphins.[4][21] The antibacterial components of triclocarban may disrupt hormones critical to the developmental and endocrine processes in exposed animal wildlife. The neurological and reproductive systems are particularly affected through contact with this compound. Triclocarban may also affect animal wildlife behavior.[21] For example, triclosan and triclocarban are 100-1,000 times more effective in inhibiting and killing algae, crustaceans and fish than they are in killing microbes. Triclocarban and triclosan have been observed in multiple organisms, including algae, aquatic blackworms, fish, and dolphins.[19]
Bioaccumulation
Triclocarban bioaccumulation is possible in a number of organisms. Earthworms are known to store this chemical in their bodies and, because of their ecological role as a food source, they have the potential to move triclocarban up the food chain.[22] Microbial species found in soils also bioaccumulate triclocarban. However, the health of these microbes has not been found to be affected by the presence of the chemical.[23] Triclocarban is rapidly accumulated in both algae and adult caged snails.[24] Moreover, triclocarban is more likely than triclosan to bioaccumulate in aquatic organisms.[25]
Bioaccumulation does occur in plants treated with water containing triclocarban. However, it is estimated that less than 0.5% of the acceptable daily intake of triclocarban for humans is represented by vegetable consumption. Thus, the concentration of triclocarban in edible portions of plants is a negligible exposure pathway for humans.
The potential for triclocarban to bioaccumulate in plants has been exploited in the construction of wetlands meant to help remove triclocarban from wastewater. These constructed wetlands are considered a cost-effective treatment option for the removal of PPCPs, including triclocarban and triclosan, from domestic water effluent. Such compounds tend to concentrate in the roots of wetland plants. Potential ecological risks associated with this method are the decrease of root systems in wetland plants, reduced nutrient uptake, decreased competitive ability, and increased potential for uprooting. Due to these risks, the long term exposure of wetland ecosystems to wastewater containing triclocarban as a major solution to wastewater pollution is still under discussion.[26]
Health concerns
Personal care
One study has investigated how triclocarban remains in the human system after using a bar of soap with traces of triclocarban. Analysis of urine samples from human test subjects shows that, after triclocarban has undergone glucuronidation, its oxidative metabolites are less readily excreted than triclocarban itself. This same study performed topical treatments of triclocarban on rats and, by analyzing urine and plasma levels, demonstrated that triclocarban does remain in the organism's system.[27]
Endocrine disorders
Triclocarban induces weak responses mediated by aryl hydrocarbon, estrogen, and androgen receptors in vitro. This has yet to be confirmed in vivo.[28] In vitro, the dihydrotestosterone-dependent activation of androgen receptor-responsive gene expression is enhanced by triclocarban by up to 130%.[29] Triclocarban is also a potent inhibitor of the enzyme soluble epoxide hydrolase (sEH) in vitro.[27] Additionally, triclocarban amplifies the bioactivity of testosterone and other androgens. This increased activity may have adverse implications for reproductive health.[5][22] Triclocarban studies on rats exhibited increased size of the specimens' prostate glands.[30] The amplification of sex hormones could promote the growth of breast and prostate cancer.
While triclocarban causes endocrine disruption, its chemical toxicity with respect to lethality is low (LD50 >5000 mg/kg). Its rate of skin absorption is also low.[31] Repeated low-dose exposure, however, can cause endocrine disruption over time.
Allergies
Triclocarban causes irritation of the lungs, eyes, and skin. Canada and Japan restrict the content of triclocarban in cosmetics.[32] Triclocarban may also cause sensitization to aeroallergens and food.[19]
Safety
Spillage may increase the risk of human, ecological, and environmental exposure to triclocarban. Immediate removal and restraint of the spill, including triclocarban as dust, is urged.[33] Although triclocarban has few to no direct detrimental effects on health aside from allergic reactions, preventing exposure to triclocarban is recommended. Since triclocarban enters the body through pores, wearing gloves, properly washing hands, and overall proper hygiene reduces the risk of skin exposure and irritation. High concentrations of triclocarban dust may remain in the lungs and inhibit lung and respiratory function. For individuals with prior respiratory conditions, triclocarban exacerbates the severity of respiratory diseases, and proper protection is recommended as a precaution. In case of exposure to triclocarban, the individual is suggested to wash the area with water or to clear the respiratory pathways.[34] In addition to its adverse effects on humans and the environment, solid triclocarban is a fire hazard. It is particularly combustible as dust. Contamination with other oxidizing agents may also result in combustion.[35]
Policy
In light of the difficulties of finding antimicrobial alternatives, the Food and Drug Administration began in the 1970s to review the safety of triclocarban and triclosan, but no enacted policy, or "drug monograph," is available to date.[19] Legal recourse by the Natural Resources Defense Council in 2010 forced the FDA to review triclocarban and triclosan.[19] However, the United States Environmental Protection Agency maintains regulatory control over triclocarban and triclosan to date.[19]
Similar in its use and its adverse health impacts as triclosan, hexachlorophene became prohibited by the FDA.[19]
Current and future research
The future of triclocarban is unknown, but scientists are searching for more sustainable antimicrobials that maintain its antibacterial properties while being minimally toxic to the environment, humans, and wildlife. This entails low degrees of bioaccumulation and rapid, clean biodegradation in existing wastewater treatment facilities. A lowered potential or no potential for resistance is also preferable.[19] These next generation chemicals should aim to act on a broad spectrum of microbes and pathogens while also being minimally toxic and bioaccumulating in non-target species.
Synthesis of these compounds could be improved upon by finding renewable sources for their production that lacks occupational hazards.[19] Research regarding the sustainability of chemical production is currently being used to help formulate green pharmaceuticals. These same principles may be applied to the development of improved antimicrobials.[19] Development in this area would benefit both people and the environment.[19]
See also
- Antibacterial soap
- Chlorine
- Dial (soap)
- Prostate cancer
- Bioaccumulation
- Breast cancer
- Triclosan
- Sludge
- Hand sanitizer
- Deodorant
- Sewage treatment
References
- 1 2 Marty, J. P.; Wepierre, J. (1979). "Toxicity evaluation of cosmetically active substances: case of trichlorocarbanilide". Labo-Pharma - Problemes et Techniques 27 (286): 306–10.
- ↑ "Anti-bacterial personal hygiene products may not be worth potential risks." UC Davis Health System Feature Story: Anti-bacterial personal hygiene products.... UC Davis Health System, n.d. Web. 12 Mar. 2014. <http://www.ucdmc.ucdavis.edu/welcome/features/20080903_anti-bacterial/>.
- 1 2 3 4 Orsi, Mario, Massimo Noro, and Jonathan Essex. "Dual-resolution molecular dynamics simulation of antimicrobials in biomembranes." Journal of The Royal Society Interface 8.59 (2010): 826-841. PubMed Central. Web. 17 Feb. 2014. http://www.ncbi.nlm.nih.gov.proxy.bc.edu/pmc/articles/PMC3104353/
- 1 2 3 4 Brausch, John, and Gary Rand. "A review of personal care products in the aquatic environment: Environmental concentrations and toxicity." Chemosphere 82.11 (2011): 1518-1532. ScienceDirect. Web. 17 Feb. 2014. http://ac.els-cdn.com/S0045653510013007/1-s2.0-S0045653510013007-main.pdf?_tid=c685833c-9812-11e3-87db-00000aab0f6c&acdnat=1392669395_a731b9c664c4e98737d93755500c43dd
- 1 2 Ahn, Ki Chang, Bin Zhao, Shirley Gee, Bruce Hammock, Jiangang Chen, Gennady Cherednichenko, Enio Sanmarti, Michael Denison, Bill Lasley, Isaac Pessah, Dietmar Kultz, and Daniel Chang. "In Vitro Biologic Activities of the Antimicrobials Triclocarban, Its Analogs, and Triclosan in Bioassay Screens: Receptor-Based Bioassay Screens." Environmental Health Perspectives 116.9 (2008): 1203-1210. PubMed Central. Web. 17 Feb. 2014. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2535623/
- ↑ Westervelt, Amy, http://www.theguardian.com/sustainable-business/avon-remove-triclosan-product-cosmetic-chemicals
- ↑ "How Do the Partitioning Properties of Polyhalogenated POPs Change When Chlorine Is Replaced with Bromine?" Environmental Science and Technology. Print. 27 April 2014. <http://pubs.acs.org/doi/abs/10.1021/es8002348.
- ↑ "Triclocarban." Chemical Book. Web. 17 Feb. 2014. <http://www.chemicalbook.com/ChemicalProductProperty_EN_CB7193925.
- ↑ Christian Six, Frank Richter "Isocyanates, Organic" in Ulmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a14_611
- ↑ "High Production Volume (HPV) Chemical Challenge Program Data Availability and Screening Level Assessment for Triclocarban." EPA. Web. 17 Feb. 2014. <http://www.epa.gov/hpv/pubs/summaries/tricloca/c14186tp.pdf
- ↑ Heath RJ, Rubin JR, Holland DR, Zhang E, Snow ME, Rock CO (April 1999). "Mechanism of triclosan inhibition of bacterial fatty acid synthesis". J. Biol. Chem. 274 (16): 11110–4. doi:10.1074/jbc.274.16.11110. PMID 10196195.
- ↑ Schweizer, Herbert (August 1, 2001). "Triclosan: a widely used biocide and its link to antibiotics". FEMS Microbiology Letters. doi:10.1111/j.1574-6968.2001.tb10772.x. Retrieved February 7, 2015.
- ↑ Chang CY, McDonnell DP. "Androgen receptor-cofactor interactions as targets for new drug discovery. Trends Pharmacol Sci. 2005;26:225–228. PMID 15860367
- ↑ McDonnell DP, Norris JD. "Connections and regulation of the human estrogen receptor" Science 2002;296:1642–1644. PMID 12040178
- ↑ Yueh, Mei-Fei, Tao Li, Ronald Evans, Bruce Hammock, and Robert Tukey. "Triclocarban Mediates Induction of Xenobiotic Metabolism through Activation of the Constitutive Androstane Receptor and the Estrogen Receptor Alpha." PLoS ONE 7.6 (2012): n. pag. PlOS ONE. Web. 17 Feb. 2014. http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0037705#pone-0037705-g006
- ↑ Chen, Jiangang, et al. "Triclocarban enhances testosterone action: a new type of endocrine disruptor?." Endocrinology 149.3 (2008): 1173-1179.
- ↑ Drugeon, H.B., B. Rouveix, and A. Michaud-Nerard. "Triclocarban antibacterial activity on resistant staphylococci, streptococci, and enterococci." Medecine et Maladies Infectieuses 42.6 (2012): 276-279. PubMed. Web. 17 Feb. 2014. PMID 22626523
- 1 2 3 Son, Ahjeong, Ian Kennedy, Kate Scow, and Krassimira Hristova. "Quantitative gene monitoring of microbial tetracycline resistance using magnetic luminescent nanoparticles." Journal of Environmental Monitoring 12.6 (2010): 1362-1367. PubMed Central. Web. 17 Feb. 2014. http://www.ncbi.nlm.nih.gov.proxy.bc.edu/pmc/articles/PMC3267904/
- 1 2 3 4 5 6 7 8 9 10 11 12 13 Halden, Rolf U. "On the Need and Speed of Regulating Triclosan and Triclocarban in the United States." Environmental science & technology (2014). http://pubs.acs.org/doi/abs/10.1021/es500495p
- ↑ Chalew, Talia E., and Rolf U. Halden. "Environmental Exposure of Aquatic and Terrestrial Biota to Triclosan and Triclocarban." PubMed. J Am Water Works Assoc., 2009. Web. Feb. 2014. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2684649/>.
- 1 2 http://www.nrdc.org/living/chemicalindex/triclosan.asp
- 1 2 Higgins, Christopher , Zachary Paesani, Talia Chalew, Rolf Halden, and Lakhwinder Hundal. "Persistence of triclocarban and triclosan in soils after land application of biosolids and bioaccumulation in Eisenia foetida." Environmental Toxicology and Chemistry 30.3 (2011): 556-563. PubMed. Web. 17 Feb. 2014. PMID 21128266
- ↑ Snyder, Elizabeth Hodges, George O’Connor, and Drew Mcavoy. "Toxicity and bioaccumulation of biosolids-borne triclocarban (TCC) in terrestrial organisms." Chemosphere 82.3 (2011): 460-467. PubMed. Web. 17 Feb. 2014. PMID 21035164
- ↑ Coogan, Melinda , and Thomas La Point. "Snail Bioaccumulation Of Triclocarban, Triclosan, And Methyltriclosan In A North Texas, USA, Stream Affected By Wastewater Treatment Plant Runoff." Environmental Toxicology and Chemistry 27.8 (2008): 1788-1793. PubMed. Web. 17 Feb. 2014. PMID 18380516
- ↑ Prosser, Ryan, Linda Lissemore, Edward Topp, and Paul Sibley. "Bioaccumulation of triclosan and triclocarban in plants grown in soils amended with municipal dewatered biosolids." Environmental Toxicology and Chemistry (2013) PubMed. Web. 17 Feb. 2014. PMID 24375516
- ↑ Zarate, Frederick , Sarah Schulwitz, Kevin Stevens, and Barney Venables. "Bioconcentration of triclosan, methyl-triclosan, and triclocarban in the plants and sediments of a constructed wetland." Chemosphere 88.3 (2012): 323-329. ScienceDirect. Web. 17 Feb. 2014. http://www.sciencedirect.com.proxy.bc.edu/science/article/pii/S0045653512003190
- 1 2 Schebb, Nils Helge, Bora Inceoglu, Ki Chang Ahn, Christophe Morisseau, Shirley Gee, and Bruce Hammock. "Investigation of Human Exposure to Triclocarban after Showering and Preliminary Evaluation of Its Biological Effects." Environmental Science & Technology 45.7 (2011): 3109-3115. PubMed. Web. 17 Feb. 2014. http://www.ncbi.nlm.nih.gov/pubmed/21381656
- ↑ Witorsch, Raphael, and John Thomas. "Personal Care Products And Endocrine Disruption: A Critical Review Of The Literature." Critical Reviews in Toxicology 40.S3 (2010): 1-30. PubMed. Web. 17 Feb. 2014. PMID 20932229
- ↑ Christen, Verena, Pierre Crettaz, Aurelia Oberli-Schrämmli, and Karl Fent. "Some flame retardants and the antimicrobials triclosan and triclocarban enhance the androgenic activity in vitro." Chemosphere 81.10 (2010): 1245-1252. PubMed. Web. 17 Feb. 2014. PMID 20943248
- ↑ "Triclocarban: sc-213106 Material Safety Data Sheet." Santa Cruz Biotechnology. Santa Cruz Biotechnology, Web. 17 Feb. 2014. <http://datasheets.scbt.com/sc-213106.pdf>.
- ↑ Marty, J. P.; Wepierre, J. (1979). "Toxicity evaluation of cosmetically active substances: case of trichlorocarbanilide". Labo-Pharma - Problemes et Techniques 27 (286): 306–10.
- ↑ "Triclocarban." EWG's Skin Deep® Cosmetics Database. Web. 17 Feb. 2014. <http://www.ewg.org/skindeep/ingredient/706622/TRICLOCARBAN/>.
- ↑ "Triclocarban: sc-213106 Material Safety Data Sheet." Santa Cruz Biotechnology. Santa Cruz Biotechnology, Web. 17 Feb. 2014. <http://datasheets.scbt.com/sc-213106.pdf>.
- ↑ "Triclocarban: sc-213106 Material Safety Data Sheet." Santa Cruz Biotechnology. Santa Cruz Biotechnology, Web. 17 Feb. 2014. <http://datasheets.scbt.com/sc-213106.pdf>.
- ↑ "Triclocarban: sc-213106 Material Safety Data Sheet." Santa Cruz Biotechnology. Santa Cruz Biotechnology, Web. 17 Feb. 2014. <http://datasheets.scbt.com/sc-213106.pdf>.
