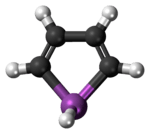
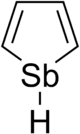Stibole
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1H-Stibole | |||
| Identifiers | |||
| 288-04-0 | |||
| Jmol interactive 3D | Image | ||
| |||
| |||
| Properties | |||
| C4H5Sb | |||
| Molar mass | 174.84 g·mol−1 | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Stibole is a theoretical heterocyclic organic compound, a five-membered ring with the formula C4H4SbH. It is classified as a metallole. It can be viewed as an structural analog of pyrrole, with antimony replacing the nitrogen atom of pyrrole. Substituted derivatives, which have been synthesized, are called stiboles.
Reactions
2,5-Dimethyl-1-phenyl-1H-stibole, for example, can be formed by the reaction of 1,1-dibutyl-2,5-dimethylstannole and dichlorophenylstibine.[1] Stiboles can be used to form ferrocene-like sandwich compounds.[2]
See also
References
- ↑ J.I.G. Cadogan, S.V. Ley, G. Pattenden, R.A. Raphael, C.W. Rees, ed. (1996), Dictionary of Organic Compounds 3 (6 ed.), Chapman & Hall, p. 2710, ISBN 978-0-412-54090-5, retrieved 2010-03-04
- ↑ A.R. Katritzky, Otto Meth-Cohn, C.W. Rees, ed. (1995), Comprehensive Organic Functional Group Transformations 4, Elsevier, pp. 1038–1040, ISBN 978-0-08-042325-8, retrieved 2010-03-04
This article is issued from Wikipedia - version of the Monday, May 18, 2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.