Steroid
A steroid is an organic compound with four rings arranged in a specific configuration. Examples include the dietary lipid cholesterol, the sex hormones estradiol and testosterone[2]:10–19 and the anti-inflammatory drug dexamethasone.[3] Steroids have two principal biological functions: certain steroids (such as cholesterol) are important components of cell membranes which alter membrane fluidity, and many steroids are signaling molecules which activate steroid hormone receptors.
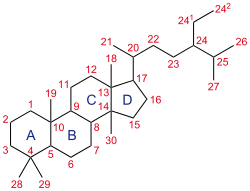
The steroid core structure is composed of seventeen carbon atoms, bonded in four "fused" rings: three six-member cyclohexane rings (rings A, B and C in the first illustration) and one five-member cyclopentane ring (the D ring). Steroids vary by the functional groups attached to this four-ring core and by the oxidation state of the rings. Sterols are forms of steroids with a hydroxyl group at position three and a skeleton derived from cholestane.[4][1]:1785f [5] They can also vary more markedly by changes to the ring structure (for example, ring scissions which produce secosteroids such as vitamin D3).
Hundreds of steroids are found in plants, animals and fungi. All steroids are manufactured in cells from the sterols lanosterol (animals and fungi) or cycloartenol (plants). Lanosterol and cycloartenol are derived from the cyclization of the triterpene squalene.[6]


Nomenclature

A gonane, the simplest steroid, is composed of seventeen carbon atoms in carbon-carbon bonds forming four fused rings in a three-dimensional shape. The three cyclohexane rings (A, B, and C in the first illustration) form the skeleton of a perhydro derivative of phenanthrene. The D ring has a cyclopentane structure. When the two methyl groups and eight carbon side chains (at C-17, as shown for cholesterol) are present, the steroid is said to have a cholestane framework. The two common 5α and 5β stereoisomeric forms of steroids exist because of differences in the side of the largely planar ring system where the hydrogen (H) atom at carbon-5 is attached, which results in a change in steroid A-ring conformation.
Examples of steroid structures are:
 |
 |
 Dexamethasone, a synthetic corticosteroid drug |
 Lanosterol, the biosynthetic precursor to animal steroids. The number of carbons (30) indicates its triterpenoid classification. |
 Progesterone, a steroid hormone involved in the female menstrual cycle, pregnancy, and embryogenesis |
 Medrogestone, a synthetic drug with effects similar to progesterone |
 β-Sitosterol, a plant or phytosterol, with a fully branched hydrocarbon side chain at C-17 and an hydroxyl group at C-3 |
In addition to the ring scissions (cleavages), expansions and contractions (cleavage and reclosing to a larger or smaller rings)—all variations in the carbon-carbon bond framework—steroids can also vary:
- in the bond orders within the rings,
- in the number of methyl groups attached to the ring (and, when present, on the prominent side chain at C17),
- in the functional groups attached to the rings and side chain, and
- in the configuration of groups attached to the rings and chain.[2]:2–9
For instance, sterols such as cholesterol and lanosterol have an hydroxyl group attached at position C-3, while testosterone and progesterone have a carbonyl (oxo substituent) at C-3; of these, lanosterol alone has two methyl groups at C-4 and cholesterol (with a C-5 to C-6 double bond) differs from testosterone and progesterone (which have a C-4 to C-5 double bond).
 Cholesterol, a prototypical animal sterol. This structural lipid and key steroid biosynthetic precursor.[1]:1785f |
 5α-cholestane, a common steroid core |
 Steroid 5α and 5β stereoisomers[1]:1786f |
Species distribution and function
The following are some common categories of steroids. In eukaryotes, steroids are found in fungi, animals, and plants. Fungal steroids include the ergosterols.
Animal steroids include compounds of vertebrate and insect origin, the latter including ecdysteroids such as ecdysterone (controlling molting in some species). Vertebrate examples include the steroid hormones and cholesterol; the latter is a structural component of cell membranes which helps determine the fluidity of cell membranes and is a principal constituent of plaque (implicated in atherosclerosis). Steroid hormones include:
- Sex hormones, which influence sex differences and support reproduction. These include androgens, estrogens, and progestagens.
- The corticosteroids, including most synthetic steroid drugs, with natural product classes the glucocorticoids (which regulate many aspects of metabolism and immune function) and the mineralocorticoids (which help maintain blood volume and control renal excretion of electrolytes)
- Anabolic steroids, natural and synthetic, which interact with androgen receptors to increase muscle and bone synthesis. In popular use, the term "steroids" often refers to anabolic steroids.
Plant steroids include steroidal alkaloids found in Solanaceae,[7] the phytosterols, and the brassinosteroids (which include several plant hormones). In prokaryotes, biosynthetic pathways exist for the tetracyclic steroid framework (e.g. in mycobacteria)[8] – where its origin from eukaryotes is conjectured[9] – and the more-common pentacyclic triterpinoid hopanoid framework.[10]
Types
Intact ring system
It is also possible to classify steroids based on their chemical composition. One example of how MeSH performs this classification is available at the Wikipedia MeSH catalog. Examples of this classification include:


| Class | Example | Number of carbon atoms |
|---|---|---|
| Cholestanes | Cholesterol | 27 |
| Cholanes | Cholic acid | 24 |
| Pregnanes | Progesterone | 21 |
| Androstanes | Testosterone | 19 |
| Estranes | Estradiol | 18 |
The gonane (steroid nucleus) is the parent 17-carbon tetracyclic hydrocarbon molecule with no alkyl sidechains.[11]
Cleaved, contracted, and expanded rings
Secosteroids (Latin seco, "to cut") are a subclass of steroidal compounds resulting, biosynthetically or conceptually, from scission (cleavage) of parent steroid rings (generally one of the four). Major secosteroid subclasses are defined by the steroid carbon atoms where this scission has taken place. For instance, the prototypical secosteroid cholecalciferol, vitamin D3 (shown), is in the 9,10-secosteroid subclass and derives from the cleavage of carbon atoms C-9 and C-10 of the steroid B-ring; 5,6-secosteroids and 13,14-steroids are similar.[12]
Norsteroids (nor-, L. norma; "normal" in chemistry, indicating carbon removal)[13] and homosteroids (homo-, Greek homos; "same", indicating carbon addition) are structural subclasses of steroids formed from biosynthetic steps. The former involves enzymic ring expansion-contraction reactions, and the latter is accomplished (biomimetically) or (more frequently) through ring closures of acyclic precursors with more (or fewer) ring atoms than the parent steroid framework.[14]
Combinations of these ring alterations are known in nature. For instance, ewes who graze on corn lily ingest cyclopamine (shown) and veratramine, two of a sub-family of steroids where the C- and D-rings are contracted and expanded respectively via a biosynthetic migration of the original C-13 atom. Ingestion of these C-nor-D-homosteroids results in birth defects in lambs: cyclopia from cyclopamine and leg deformity from veratramine.[15] A further C-nor-D-homosteroid (nakiterpiosin) is excreted by Okinawan cyanobacteriosponges – Terpios hoshinota – leading to coral mortality from black coral disease.[16] Nakiterpiosin-type steroids are active against the signaling pathway involving the smoothened and hedgehog proteins, a pathway which is hyperactive in a number of cancers.
Biological significance
Steroids and their metabolites often function as signalling molecules (the most notable examples are steroid hormones), and steroids and phospholipids are components of cell membranes. Steroids such as cholesterol decrease membrane fluidity.[17] Similar to lipids, steroids are highly concentrated energy stores. However, they are not typically sources of energy; in mammals, they are normally metabolized and excreted.
Pharmacological action
Two classes of drugs target the mevalonate pathway: statins (used to reduce elevated cholesterol levels) and bisphosphonates (used to treat a number of bone-degenerative diseases).
Biosynthesis and metabolism
The hundreds of steroids found in animals, fungi, and plants are made from lanosterol (in animals and fungi; see examples above) or cycloartenol (in plants). Lanosterol and cycloartenol derive from cyclization of the triterpenoid squalene.[6]

Steroid biosynthesis is an anabolic pathway which produces steroids from simple precursors. A unique biosynthetic pathway is followed in animals (compared to many other organisms), making the pathway a common target for antibiotics and other anti-infection drugs. Steroid metabolism in humans is also the target of cholesterol-lowering drugs, such as statins.
In humans and other animals the biosynthesis of steroids follows the mevalonate pathway, which uses acetyl-CoA as building blocks for dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP).[18] In subsequent steps DMAPP and IPP join to form geranyl pyrophosphate (GPP), which synthesizes the steroid lanosterol. Modifications of lanosterol into other steroids are classified as steroidogenesis transformations.
Mevalonate pathway

The mevalonate, or HMG-CoA reductase pathway begins with acetyl-CoA and ends with dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP). DMAPP and IPP donate isoprene units, which are assembled and modified to form terpenes and isoprenoids[19] (a large class of lipids, which include the carotenoids and form the largest class of plant natural products.[20] Here, the isoprene units are joined to make squalene and folded into a set of rings to make lanosterol.[21] Lanosterol can then be converted into other steroids, such as cholesterol and ergosterol.[21][22]
Steroidogenesis

Steroidogenesis is the biological process by which steroids are generated from cholesterol and changed into other steroids.[23] The pathways of steroidogenesis differ among species. The major classes of steroid hormones, with prominent members and examples of related functions, are:
- Progestogens:
- Progesterone, which regulates cyclical changes in the endometrium of the uterus and maintains a pregnancy
- Corticosteroids (corticoids):
- Aldosterone, a mineralocorticoid which helps regulate blood pressure
- Cortisol, a glucocorticoid whose functions include immunosuppression
- Androgens:
- Testosterone, which contributes to the development and maintenance of male secondary sex characteristics
- Estrogens:
- Estrogen, which contributes to the development and maintenance of female secondary sex characteristics
Human steroidogenesis occurs in a number of locations:
- Progestogens are the precursors of all other human steroids, and all human tissues which produce steroids must first convert cholesterol to pregnenolone. This conversion is the rate-limiting step of steroid synthesis, which occurs inside the mitochondrion of the respective tissue.[24]
- Corticosteroids are produced in the adrenal cortex.
- Estrogen and progesterone are made primarily in the ovary and the placenta during pregnancy, and testosterone in the testes.
- Testosterone is also converted to estrogen to regulate the supply of each in females and males.
- Some neurons and glia in the central nervous system (CNS) express the enzymes required for the local synthesis of pregnane neurosteroids, de novo or from peripheral sources.
Alternative pathways
In plants and bacteria, the non-mevalonate pathway uses pyruvate and glyceraldehyde 3-phosphate as substrates.[19][25]
Catabolism and excretion
Steroids are primarily oxidized by cytochrome P450 oxidase enzymes, such as CYP3A4. These reactions introduce oxygen into the steroid ring, allowing the cholesterol to be broken up by other enzymes into bile acids.[26] These acids can then be eliminated by secretion from the liver in bile.[27] The expression of the oxidase gene can be upregulated by the steroid sensor PXR when there is a high blood concentration of steroids.[28] Steroid hormones, lacking the side chain of cholesterol and bile acids, are typically hydroxylated at various ring positions or oxidized at the 17 position, conjugted with sulfate or glucuronic acid and excreted in the urine.[29]
Isolation, structure determination, and methods of analysis
Steroid isolation, depending on context, is the isolation of chemical matter required for chemical structure elucidation, derivitzation or degradation chemistry, biological testing, and other research needs (generally milligrams to grams, but often more[30] or the isolation of "analytical quantities" of the substance of interest (where the focus is on identifying and quantifying the substance (for example, in biological tissue or fluid). The amount isolated depends on the analytical method, but is generally less than one microgram.[31] The methods of isolation to achieve the two scales of product are distinct, but include extraction, precipitation, adsorption, chromatography, and crystallization. In both cases, the isolated substance is purified to chemical homogeneity; combined separation and analytical methods, such as LC-MS, are chosen to be "orthogonal"—achieving their separations based on distinct modes of interaction between substance and isolating matrix—to detect a single species in the pure sample. Structure determination refers to the methods to determine the chemical structure of an isolated pure steroid, using an evolving array of chemical and physical methods which have included NMR and small-molecule crystallography.[2]:10–19 Methods of analysis overlap both of the above areas, emphasizing analytical methods to determining if a steroid is present in a mixture and determining its quantity.[31]
Chemical synthesis
Microbial catabolism of phytosterol sidechains yields C-19 steroids, a precursor to most steroid hormones, or C-22 steroids (a precursor to adrenocortical hormones).[32][33]
The chemical conversion of sapogenins to steroids—e.g., via the Marker degradation—is a method of partial synthesis that is a long-established alternative to microbial transformation of phytosterols to steroids, and underpinned Syntex efforts using the Mexican barbasco trade (harvesting and marketing large tubers of wild-growing plants, e.g., yams) to produce early synthetic steroids.[30]
Research awards
A number of Nobel Prizes have been awarded for steroid research, including:
- 1927 (Chemistry) Heinrich Otto Wieland – Constitution of bile acids and sterols and their connection to vitamins[34]
- 1928 (Chemistry) Adolf Otto Reinhold Windaus – Constitution of sterols and their connection to vitamins[35]
- 1939 (Chemistry) Adolf Butenandt and Leopold Ruzicka – Isolation and structural studies of steroid sex hormones, and related studies on higher terpenes[36]
- 1950 (Physiology or Medicine) Edward Calvin Kendall, Tadeus Reichstein, and Philip Hench – Structure and biological effects of adrenal hormones[37]
- 1965 (Chemistry) Robert Burns Woodward – In part, for the synthesis of cholesterol, cortisone, and lanosterol[38]
- 1969 (Chemistry) Derek Barton and Odd Hassel – Development of the concept of conformation in chemistry, emphasizing the steroid nucleus[39]
- 1975 (Chemistry) Vladimir Prelog – In part, for developing methods to determine the stereochemical course of cholesterol biosynthesis from mevalonic acid via squalene[40]
See also
References
- 1 2 3 4 G.P. Moss and the Working Party of the IUPAC-IUB Joint Commission on Biochemical Nomenclature (1989). "Nomenclature of steroids, recommendations 1989" (PDF). Pure & Appl. Chem. 61 (10): 1783–1822. doi:10.1351/pac198961101783. Also available with the same authors (and year) at "IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). The nomenclature of steroids. Recommendations 1989". European Journal of Biochemistry / FEBS 186 (3): 429–58. Dec 1989. doi:10.1111/j.1432-1033.1989.tb15228.x. PMID 2606099. Note, the article co-authors, the Working Party of the IUPAC-IUB JCBN, were P. Karlson (chairman), J.R. Bull, K. Engel, J. Fried†, H.W. Kircher, K.L. Loening, G.P. Moss, G. Popják and M.R. Uskokovic. Also available online at "The Nomenclature of Steroids". London, GBR: Queen Mary University of London. p. 3S-1.4. Retrieved 10 May 2014. (See also note 4 therein.)
- 1 2 3 Lednicer D (2011). Steroid Chemistry at a Glance. Hoboken: Wiley. ISBN 978-0-470-66084-3.
- ↑ Rhen T, Cidlowski JA (Oct 2005). "Antiinflammatory action of glucocorticoids--new mechanisms for old drugs" (PDF). The New England Journal of Medicine 353 (16): 1711–23. doi:10.1056/NEJMra050541. PMID 16236742.
- ↑ Moss GP (1989). "Nomenclature of Steroids (Recommendations 1989)". Pure & Appl. Chem. 61 (10): 1783–1822. doi:10.1351/pac198961101783. PDF "IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). The nomenclature of steroids. Recommendations 1989". European Journal of Biochemistry / FEBS 186 (3): 429–58. Dec 1989. doi:10.1111/j.1432-1033.1989.tb15228.x. PMID 2606099.
- ↑ Also available in print at Hill RA, Makin HL, Kirk DN, Murphy GM (1991). Dictionary of Steroids. London, GBR: Chapman and Hall. pp. xxx–lix. ISBN 0412270609. Retrieved 20 June 2015.
- 1 2 "Lanosterol biosynthesis". Recommendations on Biochemical & Organic Nomenclature, Symbols & Terminology. International Union Of Biochemistry And Molecular Biology.
- ↑ Wink M (Sep 2003). "Evolution of secondary metabolites from an ecological and molecular phylogenetic perspective". Phytochemistry 64 (1): 3–19. doi:10.1016/S0031-9422(03)00300-5. PMID 12946402.
- ↑ Bode HB, Zeggel B, Silakowski B, Wenzel SC, Reichenbach H, Müller R (Jan 2003). "Steroid biosynthesis in prokaryotes: identification of myxobacterial steroids and cloning of the first bacterial 2,3(S)-oxidosqualene cyclase from the myxobacterium Stigmatella aurantiaca". Molecular Microbiology 47 (2): 471–81. doi:10.1046/j.1365-2958.2003.03309.x. PMID 12519197.
- ↑ Desmond E, Gribaldo S (2009). "Phylogenomics of sterol synthesis: insights into the origin, evolution, and diversity of a key eukaryotic feature". Genome Biology and Evolution 1: 364–81. doi:10.1093/gbe/evp036. PMC 2817430. PMID 20333205.
- ↑ Siedenburg G, Jendrossek D (Jun 2011). "Squalene-hopene cyclases". Applied and Environmental Microbiology 77 (12): 3905–15. doi:10.1128/AEM.00300-11. PMC 3131620. PMID 21531832.
- ↑ Edgren RA, Stanczyk FZ (Dec 1999). "Nomenclature of the gonane progestins". Contraception 60 (6): 313. doi:10.1016/S0010-7824(99)00101-8. PMID 10715364.
- ↑ Hanson JR (Jun 2010). "Steroids: partial synthesis in medicinal chemistry". Natural Product Reports 27 (6): 887–99. doi:10.1039/c001262a. PMID 20424788.
- ↑ "IUPAC Recommendations: Skeletal Modification in Revised Section F: Natural Products and Related Compounds (IUPAC Recommendations 1999)". International Union of Pure and Applied Chemistry (IUPAC). 1999.
- ↑ Wolfing J (2007). "Recent developments in the isolation and synthesis of D-homosteroids and related compounds". ARKIVOC: 210–230.
- ↑ Gao G, Chen C (2012). "Nakiterpiosin". In Corey EJ, Li JJ. Total synthesis of natural products: at the frontiers of organic chemistry. Berlin: Springer. doi:10.1007/978-3-642-34065-9. ISBN 978-3-642-34064-2.
- ↑ Uemura E, Kita M, Arimoto H, Kitamura M (2009). "Recent aspects of chemical ecology: Natural toxins, coral communities, and symbiotic relationships". Pure Appl. Chem. 81 (6): 1093–1111. doi:10.1351/PAC-CON-08-08-12.
- ↑ Sadava D, Hillis DM, Heller HC, Berenbaum MR (2011). Life: The Science of Biology 9th Edition. San Francisco: Freeman. pp. 105–114. ISBN 1-4292-4646-4.
- ↑ Grochowski LL, Xu H, White RH (May 2006). "Methanocaldococcus jannaschii uses a modified mevalonate pathway for biosynthesis of isopentenyl diphosphate". Journal of Bacteriology 188 (9): 3192–8. doi:10.1128/JB.188.9.3192-3198.2006. PMC 1447442. PMID 16621811.
- 1 2 Kuzuyama T, Seto H (Apr 2003). "Diversity of the biosynthesis of the isoprene units". Natural Product Reports 20 (2): 171–83. doi:10.1039/b109860h. PMID 12735695.
- ↑ Dubey VS, Bhalla R, Luthra R (Sep 2003). "An overview of the non-mevalonate pathway for terpenoid biosynthesis in plants" (PDF). Journal of Biosciences 28 (5): 637–46. doi:10.1007/BF02703339. PMID 14517367.
- 1 2 Schroepfer GJ (1981). "Sterol biosynthesis". Annual Review of Biochemistry 50: 585–621. doi:10.1146/annurev.bi.50.070181.003101. PMID 7023367.
- ↑ Lees ND, Skaggs B, Kirsch DR, Bard M (Mar 1995). "Cloning of the late genes in the ergosterol biosynthetic pathway of Saccharomyces cerevisiae--a review". Lipids 30 (3): 221–6. doi:10.1007/BF02537824. PMID 7791529.
- ↑ Hanukoglu I (Dec 1992). "Steroidogenic enzymes: structure, function, and role in regulation of steroid hormone biosynthesis". The Journal of Steroid Biochemistry and Molecular Biology 43 (8): 779–804. doi:10.1016/0960-0760(92)90307-5. PMID 22217824.
- ↑ Rossier MF (Aug 2006). "T channels and steroid biosynthesis: in search of a link with mitochondria". Cell Calcium 40 (2): 155–64. doi:10.1016/j.ceca.2006.04.020. PMID 16759697.
- ↑ Lichtenthaler HK (Jun 1999). "The 1-deoxy-d-xylulose-5-phosphate pathway of isoprenoid biosynthesis in plants". Annual Review of Plant Physiology and Plant Molecular Biology 50: 47–65. doi:10.1146/annurev.arplant.50.1.47. PMID 15012203.
- ↑ Pikuleva IA (Dec 2006). "Cytochrome P450s and cholesterol homeostasis". Pharmacology & Therapeutics 112 (3): 761–73. doi:10.1016/j.pharmthera.2006.05.014. PMID 16872679.
- ↑ Zollner G, Marschall HU, Wagner M, Trauner M (2006). "Role of nuclear receptors in the adaptive response to bile acids and cholestasis: pathogenetic and therapeutic considerations". Molecular Pharmaceutics 3 (3): 231–51. doi:10.1021/mp060010s. PMID 16749856.
- ↑ Kliewer SA, Goodwin B, Willson TM (Oct 2002). "The nuclear pregnane X receptor: a key regulator of xenobiotic metabolism". Endocrine Reviews 23 (5): 687–702. doi:10.1210/er.2001-0038. PMID 12372848.
- ↑ Steimer T. "Steroid Hormone Metabolism". WHO Collaborating Centre in Education and Research in Human Reproduction. Geneva Foundation for Medical Education and Research.
- 1 2 "Russell Marker Creation of the Mexican Steroid Hormone Industry". International Historic Chemical Landmark. American Chemical Society.
- 1 2 Makin HL, Honor JW, Shackleton CH, Griffiths WJ (2010). "General methods for the extraction, purification, and measurement of steroids by chromatography and mass spectrometry". In Makin HL, Gower DB. Steroid analysis. Dordrecht; New York: Springer. pp. 163–282. ISBN 978-1-4020-9774-4.
- ↑ Conner AH, Nagaoka M, Rowe JW, Perlman D (Aug 1976). "Microbial conversion of tall oil sterols to C19 steroids" (PDF). Applied and Environmental Microbiology 32 (2): 310–1. PMC 170056. PMID 987752.
- ↑ Wang F-Q, Yao K, Wei D-Z. "From Soybean Phytosterols to Steroid Hormones, Soybean and Health". In El-Shemy H. Soybean and Health. InTech. doi:10.5772/18808. ISBN 978-953-307-535-8.
- ↑ "The Nobel Prize in Chemistry 1927". The Nobel Foundation.
- ↑ "The Nobel Prize in Chemistry 1928". The Nobel Foundation.
- ↑ "The Nobel Prize in Chemistry 1939". The Nobel Foundation.
- ↑ "The Nobel Prize in Physiology or Medicine 1950". The Nobel Foundation.
- ↑ "The Nobel Prize in Chemistry 1965". The Nobel Foundation.
- ↑ "The Nobel Prize in Chemistry 1969". The Nobel Foundation.
- ↑ "The Nobel Prize in Chemistry 1975". The Nobel Foundation.
Further reading
- Russel CA (2005). "Organic Chemistry: Natural products, Steroids". In Russell CA, Roberts GK. Chemical History: Reviews of the Recent Literature. Cambridge: RSC Publ. ISBN 978-0-85404-464-1.
- "Russell Marker Creation of the Mexican Steroid Hormone Industry - Landmark -". American Chemical Society. 1999.
- Lednicer D (2011). Steroid Chemistry at a Glance. Hoboken: Wiley. doi:10.1002/9780470973639. ISBN 978-0-470-66085-0. A concise history of the study of steroids.
- Yoder RA, Johnston JN (Dec 2005). "A case study in biomimetic total synthesis: polyolefin carbocyclizations to terpenes and steroids". Chemical Reviews 105 (12): 4730–56. doi:10.1021/cr040623. PMC 2575671. PMID 16351060. A review of the history of steroid synthesis, especially biomimetic.
- Han TS, Walker BR, Arlt W, Ross RJ (Feb 2014). "Treatment and health outcomes in adults with congenital adrenal hyperplasia". Nature Reviews. Endocrinology 10 (2): 115–24. doi:10.1038/nrendo.2013.239. PMID 24342885. Adrenal steroidogenesis pathway.
External links
- G.P. Moss and the Working Party of the IUPAC-IUB JCBN. "The Nomenclature of Steroids". London, GBR: Queen Mary University of London.. Alternatively, see or , all web sources accessed 20 June 2015. [The content co-authors, the Working Party of the IUPAC-IUB JCBN, were P. Karlson (chairman), J.R. Bull, K. Engel, J. Fried†, H.W. Kircher, K.L. Loening, G.P. Moss, G. Popják and M.R. Uskokovic.]
- Bowen RA (October 20, 2001). "Steroidogenesis". Pathophysiology of the Endocrine System. Colorado State University.
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|