Shannon Criteria
The Shannon criteria constitute an empirical rule in neural engineering that is used for evaluation of possibility of damage from electrical stimulation to nervous tissue.[1]
The Shannon criteria relate two parameters for pulsed electrical stimulation: charge density per phase, D (μCoulombs/(phase•cm²)) and charge per phase, Q (μCoulombs/phase) with a dimensionless parameter k:

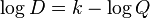
or, alternatively:

According to these criteria, stimulation parameters that yield k > 1.75 could cause damage to the adjacent nervous tissue. Currently, this empirical law is applied in neuromodulation for development of implants for cortical, cochlear, retinal,[2][3] spinal cord[4] and deep brain stimulation.[5][6] Shannon categorizes the relationship between stimulating electrode and target neural tissue as either Near Field, Mid Field, or Far Field, and discusses how equation parameters may be chosen in each case.
Limitations
The data on which the Shannon model is built are restricted to experiments performed in cat cerebral cortex with 8 hours of stimulation at 50 Hz using surface disc electrodes of 1 mm² or larger.
References
- ↑ Shannon, R.V. (April 1992). "A model of safe levels for electrical stimulation". IEEE Transactions on Biomedical Engineering 39 (4): 424–426. doi:10.1109/10.126616.
- ↑ Eiber, Calvin D; Lovell, Nigel H; Suaning, Gregg J (1 February 2013). "Attaining higher resolution visual prosthetics: a review of the factors and limitations". Journal of Neural Engineering 10 (1): 011002. doi:10.1088/1741-2560/10/1/011002.
- ↑ Winter, Jessica O.; Cogan, Stuart F.; Rizzo, Joseph F. (January 2007). "Retinal prostheses: current challenges and future outlook". Journal of Biomaterials Science, Polymer Edition 18 (8): 1031–1055. doi:10.1163/156856207781494403.
- ↑ Wesselink, WA; Holsheimer, J; Boom, HB (June 1998). "Analysis of current density and related parameters in spinal cord stimulation.". IEEE transactions on rehabilitation engineering : a publication of the IEEE Engineering in Medicine and Biology Society 6 (2): 200–7. doi:10.1109/86.681186. PMID 9631328.
- ↑ Testerman, Roy L; Rise, Mark T; Stypulkowski, Paul H (Sep–Oct 2006). "Electrical stimulation as therapy for neurological disorder". IEEE Engineering in Medicine and Biology Magazine 25: 74–8. doi:10.1109/memb.2006.1705750. PMID 17020202.
- ↑ Grill, Warren M (July 2005). "Safety considerations for deep brain stimulation: review and analysis". Expert Review of Medical Devices 2 (4): 409–420. doi:10.1586/17434440.2.4.409.
Further reading
- Merrill, Daniel R.; Bikson, Marom; Jefferys, John G.R. (February 2005). "Electrical stimulation of excitable tissue: design of efficacious and safe protocols". Journal of Neuroscience Methods 141 (2): 171–198. doi:10.1016/j.jneumeth.2004.10.020.
- McCreery, Douglas (2004). "Tissue Reaction to Electrodes:The Problem of Safe and Effective Stimulation of Neural Tissue". In Horch, Kenneth W; Dhillon, Gurpreet S. Neuroprosthetics 2. pp. 592–611. doi:10.1142/9789812561763_0018.