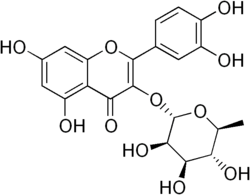
Quercitrin
Not to be confused with quercetin.
 | |
| Names | |
|---|---|
| IUPAC name
2-(3,4-Dihydroxyphenyl)-5,7- dihydroxy-3-[ [(2S,3R,4R,5R,6S)- 3,4,5-trihydroxy-6-methyl-2- tetrahydropyranyl]oxy]-4-chromenone | |
| Other names
Quercetin 3-O-a-L-rhamnoside Thujin Quercetin 3-rhamnoside Quercetin-3-rhamnoside Quercetin-3-L-rhamnoside | |
| Identifiers | |
| 522-12-3 | |
| ChEBI | CHEBI:17558 |
| ChEMBL | ChEMBL82242 |
| ChemSpider | 4444112 |
| Jmol interactive 3D | Image |
| PubChem | 5280459 |
| UNII | 2Y8906LC5P |
| |
| |
| Properties | |
| C21H20O11 | |
| Molar mass | 448.38 g/mol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Quercitrin is a glycoside formed from the flavonoid quercetin and the deoxy sugar rhamnose.
Austrian chemist Heinrich Hlasiwetz (1825-1875) is remembered for his chemical analysis of quercitrin.
Occurrence
Quercitrin is a constituent of the dye quercitron. It can be found in Tartary buckwheat (Fagopyrum tataricum)[1] and in oaks species like the North American white oak (Quercus alba) and English oak (Quercus robur).[2] It is also found in Nymphaea odorata or Taxillus kaempferi.[3]
Metabolism
The enzyme quercitrinase catalyzes the chemical reaction between quercitrin and H2O to yield L-rhamnose and quercetin.
References
- ↑ Tartary Buckwheat (Fagopyrum tataricum Gaertn.) as a Source of Dietary Rutin and Quercitrin. Nina Fabjan, Janko Rode, Iztok Jože Košir, Zhuanhua Wang, Zheng Zhang and Ivan Kreft, J. Agric. Food Chem., 2003, 51 (22), pp. 6452–6455, doi:10.1021/jf034543e
- ↑ Analysis of oak tannins by liquid chromatography-electrospray ionisation mass spectrometry. Pirjo Mämmelä, Heikki Savolainenb, Lasse Lindroosa, Juhani Kangasd and Terttu Vartiainen, Journal of Chromatography A, Volume 891, Issue 1, 1 September 2000, Pages 75-83, doi:10.1016/S0021-9673(00)00624-5
- ↑ The constituents of Taxillus kaempferi and the host, Pinus thunbergii. I. Catechins and flavones of Taxillus kaempferi. Konishi T, Nishio T, Kiyosawa S, Fujiwara Y and Konoshima T, Yakugaku Zasshi., February 1996, volume 116, issue 2, pages 148-157 (article in Japanese)
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
This article is issued from Wikipedia - version of the Saturday, January 30, 2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.