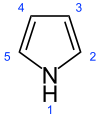
Pyrrole
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1H-Pyrrole | |||
| Other names
Azole | |||
| Identifiers | |||
| 109-97-7 | |||
| 1159 | |||
| ChEBI | CHEBI:19203 | ||
| ChEMBL | ChEMBL16225 | ||
| ChemSpider | 7736 | ||
| EC Number | 203-724-7 | ||
| 1705 | |||
| Jmol interactive 3D | Image Image | ||
| PubChem | 8027 | ||
| RTECS number | UX9275000 | ||
| UNII | 86S1ZD6L2C | ||
| UN number | 1992, 1993 | ||
| |||
| |||
| Properties | |||
| C4H5N | |||
| Molar mass | 67.09 g·mol−1 | ||
| Density | 0.967 g cm−3 | ||
| Melting point | −23 °C (−9 °F; 250 K) | ||
| Boiling point | 129 to 131 °C (264 to 268 °F; 402 to 404 K) | ||
| Vapor pressure | 7 mmHg at 23 °C | ||
| Viscosity | 0.001225 Pa s | ||
| Thermochemistry | |||
| 1.903 J k−1 mol k−1 | |||
| Std enthalpy of formation (ΔfH |
108.2 kJ mol−1 (gas) | ||
| Std enthalpy of combustion (ΔcH |
2242 kJ mol−1 | ||
| Hazards | |||
| NFPA 704 | |||
| Flash point | 33.33 °C (91.99 °F; 306.48 K) | ||
| 550 °C (1,022 °F; 823 K) | |||
| Explosive limits | 3.1–14.8% | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4H4NH.[1] It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., N-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme.[2]
Pyrroles are components of more complex macrocycles, including the porphyrins of heme, the chlorins, bacteriochlorins, chlorophyll, and porphyrinogens.[3]
Properties
Pyrrole is a colorless volatile liquid that darkens readily upon exposure to air, and is usually purified by distillation immediately before use.[4] Pyrrole is a 5-membered aromatic heterocycle, like furan and thiophene. Unlike furan and thiophene, it has a dipole in which the positive end lies on the side of the heteroatom, with a dipole moment of 1.58 D. In CDCl3, it has a chemical shifts at 6.68 (H2, H5) and 6.22 (H3, H4). Pyrrole is weakly basic, with a conjugate acid pKa of -3.8. The most thermodynamically stable pyrrolium cation is formed by protonation at the 2 position. Substitution of pyrrole with alkyl substituents provides a more basic molecule—for example, tetramethylpyrrole has a conjugate acid pKa of +3.7. Pyrrole is also weakly acidic at the N-H position, with a pKa of 17.5.
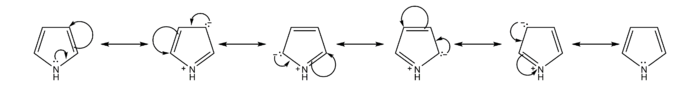 Resonance Contributors of Pyrrole
Resonance Contributors of Pyrrole
History
Pyrrole was first detected by F. F. Runge in 1834, as a constituent of coal tar. In 1857, it was isolated from the pyrolysate of bone. Its name comes from the Greek pyrros (πυρρός, “fiery”), from the reaction used to detect it—the red color that it imparts to wood when moistened with hydrochloric acid.[5]
Occurrence in nature
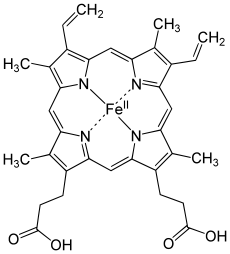
Pyrroles are found in a variety of biological contexts, as parts of cofactors and natural products. Common naturally produced molecules containing pyrroles include vitamin B12, bile pigments like bilirubin and biliverdin, and the porphyrins of heme, chlorophyll, chlorins, bacteriochlorins, and porphyrinogens.[3] Other pyrrole-containing secondary metabolites include PQQ, makaluvamine M, ryanodine, rhazinilam, lamellarin, prodigiosin, myrmicarin, and sceptrin. Pyrroles are also found in several drugs, including atorvastatin, ketorolac, and sunitinib.
One of the first syntheses of pyrrole-containing molecules was that of haemin, synthesized by Emil Fischer in 1929.[6]
Synthesis
Pyrrole is prepared industrially by treatment of furan with ammonia in the presence of solid acid catalysts, like SiO2 and Al2O3.[5]

Pyrrole can also be formed by catalytic dehydrogenation of pyrrolidine.
Laboratory routes
Several syntheses of the pyrrole ring have been described.
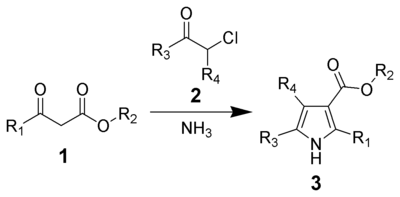
Hantzsch pyrrole synthesis
The Hantzsch pyrrole synthesis is the reaction of β-ketoesters (1) with ammonia (or primary amines) and α-haloketones (2) to give substituted pyrroles (3).[7][8]
Knorr pyrrole synthesis
The Knorr pyrrole synthesis involves the reaction of an α-amino ketone or an α-amino-β-ketoester with an activated methylene compound.[9][10][11] The method involves the reaction of an α-amino-ketone (1) and a compound containing a methylene group α- to (bonded to the next carbon to) a carbonyl group (2).[12]

Paal-Knorr pyrrole synthesis
In the Paal-Knorr pyrrole synthesis, a 1,4-dicarbonyl compound reacts with ammonia or a primary amine to form a substituted pyrrole.[13][14]
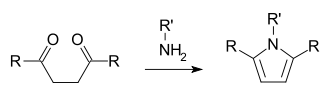
Van Leusen reaction
The Van Leusen reaction can be used to form pyrroles, by reaction of tosylmethyl isocyanide (TosMIC) with an enone in the presence of base, in a Michael addition. A 5-endo cyclization then forms the 5-membered ring, which reacts to eliminate the tosyl group. The last step is tautomerization to the pyrrole.

Barton-Zard synthesis
The Barton-Zard synthesis proceeds in a manner similar to the van Leusen synthesis. An isocyanoacetate reacts with a nitroalkene in a 1,4-addition, followed by 5-endo-dig cyclization, elimination of the nitro group, and tautomerization.[15]

Piloty–Robinson pyrrole synthesis
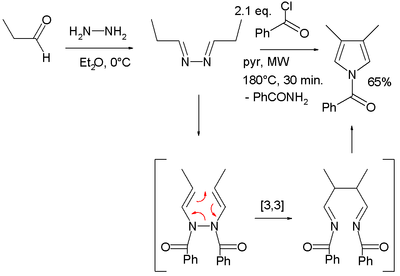
The starting materials in the Piloty–Robinson pyrrole synthesis, named for Gertrude and Robert Robinson and Oskar Piloty, are 2 equivalents of an aldehyde and hydrazine.[16][17] The product is a pyrrole with substituents at the 3 and 4 positions. The aldehyde reacts with the diamine to an intermediate di-imine (R–C=N−N=C–R). In the second step, a [3,3]-sigmatropic rearrangement takes place between. Addition of hydrochloric acid leads to ring-closure and loss of ammonia to form the pyrrole. The mechanism was developed by the Robinsons.
In one modification, propionaldehyde is treated first with hydrazine and then with benzoyl chloride at high temperatures and assisted by microwave irradiation:[18]
Cycloaddition-based routes
Various methods for the synthesis of pyrroles via two-component cycloaddition reactions are known. For example, dipolar cycloaddition of an oxido-oxazolium with an alkyne, followed by a retro-Diels-Alder reaction to expel CO2 provides pyrrole. Similar reactions can be performed using azalactones.

Pyrroles can be prepared by silver-catalyzed cyclization of alkynes with isonitriles, where R2 is an electron-withdrawing group, and R1 is an alkane, aryl group, or ester. Examples of disubstituted alkynes have also been seen to form the desired pyrrole in considerable yield. The reaction is proposed to proceed via an Ag-acetylide intermediate. This method is analogous to the azide-alkyne click chemistry used to form azoles.
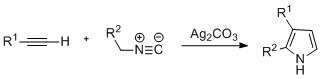
Other methods
One synthetic route to pyrrole involves the decarboxylation of ammonium mucate, the ammonium salt of mucic acid. The salt is typically heated in a distillation setup with glycerol as a solvent.[19]

Biosynthesis of pyrroles
The de novo biosynthesis of pyrrole rings begins with aminolevulinic acid (ALA), which is synthesized from glycine and succinyl-CoA. ALA dehydratase catalyzes the condensation of two ALA molecules via a Knorr-type ring synthesis to form porphobilinogen (PBG). This later reacts to form, for example, the macrocycles heme and chlorophyll.[20]

Reactions and reactivity
Due to its aromatic character, pyrrole is difficult to hydrogenate, does not easily react as a diene in Diels-Alder reactions, and does not undergo usual olefin reactions. Its reactivity is similar to that of benzene and aniline, in that it is easy to alkylate and acylate. Under acidic conditions, pyrroles polymerize easily, and thus many electrophilic reagents that are used in benzene chemistry are not applicable to pyrroles.
Reaction of pyrrole with electrophiles
Pyrroles generally react with electrophiles at the α position (C2 or C5), due to the highest degree of stability of the protonated intermediate.

Pyrroles react easily with nitrating (e.g. HNO3/Ac2O), sulfonating (Py-SO3), and halogenating (e.g. NCS, NBS, Br2, SO2Cl2, and KI/H2O2) agents. Halogenation generally provides polyhalogenated pyrroles, but monohalogenation can be performed. As is typical for electrophilic additions to pyrroles, halogenation generally occurs at the α position, but can also occur at the β position by silation of the nitrogen. This is a useful method for further functionalization of the generally less reactive β position.
Acylation
Acylation generally occurs at the 2-position, through the use of various methods. Acylation with anhydrides and acid chlorides can occur without a catalyst; alternatively, a Lewis acid may be used. 2-Acylpyrroles are also obtained from reaction with nitriles, by the Houben–Hoesch reaction. Pyrrole aldehydes can be formed by a Vilsmeier–Haack reaction. N-acylation of simple pyrrole does not occur.

Alkylation
Electrophilic alkylation of simple pyrrole is uncommon. Alkylation to form enones at C2 has been seen.
Reaction of deprotonated pyrrole
The NH proton in pyrroles is moderately acidic with a pKa of 16.5. Pyrrole can be deprotonated with strong bases such as butyllithium and sodium hydride. The resulting alkali pyrrolide is nucleophilic. Treating this conjugate base with an electrophile such as methyl iodide gives N-methylpyrrole. N-metallated pyrrole can react with electrophiles at the N or C positions, depending on the coordinating metal. More ionic N-metal bonds (such as with Li, Na, and K) and more solvating solvents lead to N-alkylation. Nitrophilic metals, such as MgX, lead to alkylation at C (mainly C2), due to a higher degree of coordination to the nitrogen atom. In the cases of N-substituted pyrroles, metallation of the carbons is more facile. Alkyl groups can be introduced as electrophiles, or by cross-coupling reactions.
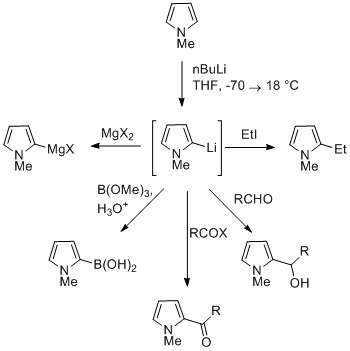
Substitution at C3 can be achieved through the use of N-substituted 3-bromopyrrole, which can be synthesized by bromination of N-silylpyrrole with NBS.
Reductions
Pyrroles can undergo reductions to pyrrolidines and to pyrrolines. For example, Birch reduction of pyrrole esters and amides produced pyrrolines, with the regioselectivity depending on the position of the electron-withdrawing group.
Cyclization reactions
Pyrroles with N-substitution can undergo cycloaddition reactions such as (4+2), (2+2), (2+1) cyclizations. Diels-Alder cyclizations can occur with the pyrrole acting as a diene, especially in the presence of an electron-withdrawing group on the N. Vinylpyrroles can also act as dienes.

Pyrroles can react with carbenes, such as dichlorocarbene, in a [2+1] cycloaddition. With dichlorocarbene, a dichlorocyclopropane intermediate is formed, which breaks down to form 3-chloropyridine (Ciamician-Dennstedt Rearrangement).[21]

Commercial uses
Pyrrole is essential to the production of many different chemicals. N-methylpyrrole is a precursor to N-methylpyrrolecarboxylic acid, a building-block in pharmaceutical chemistry.[5] Although there is a claim that pyrrole is used as an additive to cigarettes, it is typically listed as a constituent of tobacco smoke and not as an ingredient.[22]
Analogs and derivatives
Structural analogs of pyrrole include:
- Pyrroline, a partially saturated analog with one double bond
- Pyrrolidine, the saturated hydrogenated analog
Derivatives of pyrrole include indole, a derivative with a fused benzene ring.
See also
References
- ↑ Loudon, Marc G. (2002). "Chemistry of Naphthalene and the Aromatic Heterocycles.". Organic Chemistry (Fourth ed.). New York: Oxford University Press. pp. 1135–1136. ISBN 0-19-511999-1.
- ↑ Cox, Michael; Lehninger, Albert L; Nelson, David R. (2000). Lehninger principles of biochemistry. New York: Worth Publishers. ISBN 1-57259-153-6.
- 1 2 Jonas Jusélius and Dage Sundholm (2000). "The aromatic pathways of porphins, chlorins and bacteriochlorins". Phys. Chem. Chem. Phys. (Open access) 2 (10): 2145–2151. doi:10.1039/b000260g.
- ↑ Armarego, Wilfred, L.F.; Chai, Christina, L.L. (2003). Purification of Laboratory Chemicals (5th ed.). Elsevier. p. 346.
- 1 2 3 Albrecht Ludwig Harreus "Pyrrole" in Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a22_453
- ↑ Emil, Fischer. "Nobel Prize Lecture" (PDF).
- ↑ Hantzsch, A (1890). Ber. 23: 1474. Missing or empty
|title=(help) - ↑ Feist, F (1902). Ber. 35: 1538. Missing or empty
|title=(help) - ↑ Knorr, L. (1884). "Synthese von Pyrrolderivaten". Berichte der deutschen chemischen Gesellschaft 17 (2): 1635. doi:10.1002/cber.18840170220.
- ↑ Knorr, L. (1886). Ann. 236: 290. Missing or empty
|title=(help) - ↑ Knorr, L.; Lange, H. (1902). "Ueber die Bildung von Pyrrolderivaten aus Isonitrosoketonen". Berichte der deutschen chemischen Gesellschaft 35 (3): 2998. doi:10.1002/cber.19020350392.
- ↑ Corwin, A. H. (1950). Heterocyclic Compounds 1: 287. Missing or empty
|title=(help) - ↑ Paal, C. (1884), Berichte der deutschen chemischen Gesellschaft 17: 2756, doi:10.1002/cber.188401702228 Missing or empty
|title=(help) - ↑ Knorr, L. (1884), Berichte der deutschen chemischen Gesellschaft 17: 2863, doi:10.1002/cber.188401702254 Missing or empty
|title=(help) - ↑ Jie Jack Li (2013). Heterocyclic Chemistry in Drug Discovery. New York: Wiley. ISBN 9781118354421.
- ↑ Piloty, O. (1910). "Synthese von Pyrrolderivaten: Pyrrole aus Succinylobernsteinsäureester, Pyrrole aus Azinen". Chem. Ber. 43: 489. doi:10.1002/cber.19100430182.
- ↑ Robinson, Gertrude Maud; Robinson, Robert (1918). "LIV.—A new synthesis of tetraphenylpyrrole". J. Chem. Soc. 113: 639. doi:10.1039/CT9181300639.
- 1 2 Benjamin C. Milgram, Katrine Eskildsen, Steven M. Richter, W. Robert Scheidt, and Karl A. Scheidt (2007). "Microwave-Assisted Piloty–Robinson Synthesis of 3,4-Disubstituted Pyrroles" (Note). J. Org. Chem. 72 (10): 3941–3944. doi:10.1021/jo070389. PMC 1939979. PMID 17432915.
- ↑ Practical Organic Chemistry, Vogel, 1956, Page 837, Link (12 MB)
- ↑ Walsh, Christopher T.; Garneau-Tsodikova, Sylvie; Howard-Jones, Annaleise R. "Biological formation of pyrroles: Nature's logic and enzymatic machinery". Natural Product Reports 23 (4): 517. doi:10.1039/b605245m.
- ↑ Ciamician, G. L. and Dennstedt, M. Ber. 14, 1153 (1881); Corwin, A. H. Heterocyclic Compounds 1, 309 (1950); Mosher, H. S. Heterocyclic Compounds 475
- ↑ Fowles, Jefferson; Michael Bates; Dominique Noiton (March 2000). "The Chemical Constituents in Cigarettes and Cigarette Smoke: Priorities for Harm Reduction" (PDF). Porirua, New Zealand: New Zealand Ministry of Health. pp. 20, 49–65. Retrieved 2012-09-23.
Further reading
- Jones, R. Jones, ed. (1990). Pyrroles. Part I. The Synthesis and the physical and Chemical aspects of the Pyrrole Ring. The Chemistry of heterocyclic compounds 48. Chichester: John Wiley & Sons. doi:10.1002/recl.19911100712. ISBN 0-471-62753-4.
External links
- Synthesis of pyrroles (overview of recent methods)
- Substitution reaction mechanisms of nitrogen-containing heteroaromatics
- Jolicoeur, Benoit; Chapman, Erin E.; Thompson, Alison; Lubell, William D. (2006). "Pyrrole protection". Tetrahedron 62 (50): 11531. doi:10.1016/j.tet.2006.08.071.
|