Hypoxia (medical)
| Hypoxia | |
|---|---|
|
Cyanosis of the hand in someone with low oxygen saturations | |
| Classification and external resources | |
| Specialty | Pulmonology, toxicology |
| ICD-10 | J96 |
| ICD-9-CM | 799.02 |
| DiseasesDB | 6623 |
| MeSH | D000860 |
Hypoxia (also known as hypoxiation) is a condition in which the body or a region of the body is deprived of adequate oxygen supply. Hypoxia may be classified as either generalized, affecting the whole body, or local, affecting a region of the body. Although hypoxia is often a pathological condition, variations in arterial oxygen concentrations can be part of the normal physiology, for example, during hypoventilation training[1] or strenuous physical exercise.
Hypoxia differs from hypoxemia and anoxemia in that hypoxia refers to a state in which oxygen supply is insufficient, whereas hypoxemia and anoxemia refer specifically to states that have low or zero arterial oxygen supply.[2] Hypoxia in which there is complete deprivation of oxygen supply is referred to as anoxia.
Generalized hypoxia occurs in healthy people when they ascend to high altitude, where it causes altitude sickness leading to potentially fatal complications: high altitude pulmonary edema (HAPE) and high altitude cerebral edema (HACE).[3] Hypoxia also occurs in healthy individuals when breathing mixtures of gasses with a low oxygen content, e.g. while diving underwater especially when using closed-circuit rebreather systems that control the amount of oxygen in the supplied air. A mild and non-damaging intermittent hypoxia is used intentionally during altitude trainings to develop an athletic performance adaptation at both the systemic and cellular level.[4]
Hypoxia is also a serious consequence of preterm birth in the neonate. The main cause for this is that the lungs of the human fetus are among the last organs to develop during pregnancy. To assist the lungs to distribute oxygenated blood throughout the body, infants at risk of hypoxia are often placed inside an incubator capable of providing continuous positive airway pressure (also known as a humidicrib).
Signs and symptoms
Generalised hypoxia
The symptoms of generalized hypoxia depend on its severity and acceleration of onset.
In the case of altitude sickness, where hypoxia develops gradually, the symptoms include light-headedness / fatigue, numbness / tingling of extremities, nausea and anoxia.[5][6] In severe hypoxia, or hypoxia of very rapid onset, ataxia, confusion / disorientation / hallucinations / behavioral change, severe headaches / reduced level of consciousness, papilloedema, breathlessness,[7] pallor,[8] tachycardia and pulmonary hypertension eventually leading to the late signs cyanosis, slow heart rate / cor pulmonale and low blood pressure followed by death.[9][10]
Because hemoglobin is a darker red when it is not bound to oxygen (deoxyhemoglobin), as opposed to the rich red color that it has when bound to oxygen (oxyhemoglobin), when seen through the skin it has an increased tendency to reflect blue light back to the eye.[11] In cases where the oxygen is displaced by another molecule, such as carbon monoxide, the skin may appear 'cherry red' instead of cyanotic.[12]
Local hypoxia
If tissue is not being perfused properly, it may feel cold and appear pale; if severe, hypoxia can result in cyanosis, a blue discoloration of the skin. If hypoxia is very severe, a tissue may eventually become gangrenous.
Extreme pain may also be felt at or around the site.
Cause
Oxygen passively diffuses in the lung alveoli according to a pressure gradient. Oxygen diffuses from the breathed air, mixed with water vapour, to arterial blood, where its partial pressure is around 100 mmHg (13.3 kPa).[13] In the blood, oxygen is bound to hemoglobin, a protein in red blood cells. The binding capacity of hemoglobin is influenced by the partial pressure of oxygen in the environment, as described in the oxygen–hemoglobin dissociation curve. A smaller amount of oxygen is transported in solution in the blood.
In peripheral tissues, oxygen again diffuses down a pressure gradient into cells and their mitochondria, where it is used to produce energy in conjunction with the breakdown of glucose, fats and some amino acids.
Hypoxia can result from a failure at any stage in the delivery of oxygen to cells. This can include decreased partial pressures of oxygen, problems with diffusion of oxygen in the lungs, insufficient available hemoglobin, problems with blood flow to the end tissue, and problems with breathing rhythm.
Experimentally, oxygen diffusion becomes rate limiting (and lethal) when arterial oxygen partial pressure falls to 60 mmHg (5.3 kPa) or below.
Almost all the oxygen in the blood is bound to hemoglobin, so interfering with this carrier molecule limits oxygen delivery to the periphery. Hemoglobin increases the oxygen-carrying capacity of blood by about 40-fold,[14] with the ability of hemoglobin to carry oxygen influenced by the partial pressure of oxygen in the environment, a relationship described in the oxygen–hemoglobin dissociation curve. When the ability of hemoglobin to carry oxygen is interfered with, a hypoxic state can result.[15]:997–999
Ischemia
Ischemia, meaning insufficient blood flow to a tissue, can also result in hypoxia. This is called 'ischemic hypoxia'. This can include an embolic event, a heart attack that decreases overall blood flow, or trauma to a tissue that results in damage. An example of insufficient blood flow causing local hypoxia is gangrene that occurs in diabetes.
Diseases such as peripheral vascular disease can also result in local hypoxia. For this reason, symptoms are worse when a limb is used. Pain may also be felt as a result of increased hydrogen ions leading to a decrease in blood pH (acidity) created as a result of anaerobic metabolism.
Hypoxemic hypoxia
This refers specifically to hypoxic states where the arterial content of oxygen is insufficient.[16] This can be caused by alterations in respiratory drive, such as in respiratory alkalosis, physiological or pathological shunting of blood, diseases interfering in lung function resulting in a ventilation-perfusion mismatch, such as a pulmonary embolus, or alterations in the partial pressure of oxygen in the environment or lung alveoli, such as may occur at altitude or when diving.
Anemia
Hemoglobin plays a substantial role in carrying oxygen throughout the body,[14] and when it is deficient, anemia can result, causing 'anaemic hypoxia' if tissue perfusion is decreased. Iron deficiency is the most common cause of anemia. As iron is used in the synthesis of hemoglobin, less hemoglobin will be synthesised when there is less iron, due to insufficient intake, or poor absorption.[15]:997–999
Anemia is typically a chronic process that is compensated over time by increased levels of red blood cells via upregulated erythropoetin. A chronic hypoxic state can result from a poorly compensated anaemia.[15]:997–999
Carbon monoxide poisoning
Carbon monoxide competes with oxygen for binding sites on hemoglobin molecules. As carbon monoxide binds with hemoglobin hundreds of times tighter than oxygen, it can prevent the carriage of oxygen.[17] Carbon monoxide poisoning can occur acutely, as with smoke intoxication, or over a period of time, as with cigarette smoking. Due to physiological processes, carbon monoxide is maintained at a resting level of 4-6 ppm. This is increased in urban areas (7 - 13 ppm) and in smokers (20 - 40 ppm).[18] A carbon monoxide level of 40 ppm is equivalent to a reduction in hemoglobin levels of 10 g/L.[18][19]
CO has a second toxic effect, namely removing the allosteric shift of the oxygen dissociation curve and shifting the foot of the curve to the left. In so doing, the hemoglobin is less likely to release its oxygens at the peripheral tissues.[20] Certain abnormal hemoglobin variants also have higher than normal affinity for oxygen, and so are also poor at delivering oxygen to the periphery.
Hypoxic breathing gases
The breathing gas in scuba diving may contain an insufficient partial pressure of oxygen, particularly in malfunction of rebreathers. Such situations may lead to unconsciousness without symptoms since carbon dioxide levels are normal and the human body senses pure hypoxia poorly.
A similar problem exists when inhaling certain odorless asphyxiant gases. These produce a hypoxic breathing gas which can produce unconsciousness and death without symptoms. This may cause inert gas asphyxiation. Such asphyxia may be deliberate with use of a suicide bag. Accidental death has occurred in cases where concentrations of nitrogen in controlled atmospheres, or methane in mines, has not been detected or appreciated.
Cyanide poisoning
Histotoxic hypoxia results when the quantity of oxygen reaching the cells is normal, but the cells are unable to use the oxygen effectively, due to disabled oxidative phosphorylation enzymes. This may occur in Cyanide poisoning.
Other
Hemoglobin's function can also be lost by chemically oxidizing its iron atom to its ferric form. This form of inactive hemoglobin is called methemoglobin and can be made by ingesting sodium nitrite[21] as well as certain drugs and other chemicals.
Physiological compensation
Acute
If oxygen delivery to cells is insufficient for the demand (hypoxia), hydrogen will be shifted to pyruvic acid converting it to lactic acid. This temporary measure (anaerobic metabolism) allows small amounts of energy to be released. Lactic acid build up (in tissues and blood) is a sign of inadequate mitochondrial oxygenation, which may be due to hypoxemia, poor blood flow (e.g., shock) or a combination of both.[22] If severe or prolonged it could lead to cell death.
In humans, hypoxia is detected by chemoreceptors in the carotid body. This response does not control ventilation rate at normal pO
2, but below normal the activity of neurons innervating these receptors increases dramatically, so much so to override the signals from central chemoreceptors in the hypothalamus, increasing pO
2 despite a falling pCO2
It is seen in a few humans (encountered with hypoxia), there is word loss in their speech due their state of confusion and cell damages in the brain.
In most tissues of the body, the response to hypoxia is vasodilation. By widening the blood vessels, the tissue allows greater perfusion.
By contrast, in the lungs, the response to hypoxia is vasoconstriction. This is known as "Hypoxic pulmonary vasoconstriction", or "HPV".
Chronic
When the pulmonary capillary pressure remains elevated chronically (for at least 2 weeks), the lungs become even more resistant to pulmonary edema because the lymph vessels expand greatly, increasing their capability of carrying fluid away from the interstitial spaces perhaps as much as 10-fold. Therefore, in patients with chronic mitral stenosis, pulmonary capillary pressures of 40 to 45 mm Hg have been measured without the development of lethal pulmonary edema.[Guytun and Hall physiology]
Hypoxia exists when there is a reduced amount of oxygen in the tissues of the body. Hypoxemia refers to a reduction in PO2 below the normal range, regardless of whether gas exchange is impaired in the lung, CaO2 is adequate, or tissue hypoxia exists. There are several potential physiologic mechanisms for hypoxemia, but in patients with COPD the predominant one is V/Q mismatching, with or without alveolar hypoventilation, as indicated by PaCO2. Hypoxemia caused by V/Q mismatching as seen in COPD is relatively easy to correct, so that only comparatively small amounts of supplemental oxygen (less than 3 L/min for the majority of patients) are required for LTOT. Although hypoxemia normally stimulates ventilation and produces dyspnea, these phenomena and the other symptoms and signs of hypoxia are sufficiently variable in patients with COPD as to be of limited value in patient assessment. Chronic alveolar hypoxia is the main factor leading to development of cor pulmonale—right ventricular hypertrophy with or without overt right ventricular failure—in patients with COPD. Pulmonary hypertension adversely affects survival in COPD, to an extent that parallels the degree to which resting mean pulmonary artery pressure is elevated. Although the severity of airflow obstruction as measured by FEV1 is the best correlate with overall prognosis in patients with COPD, chronic hypoxemia increases mortality and morbidity for any severity of disease. Large-scale studies of LTOT in patients with COPD have demonstrated a dose-response relationship between daily hours of oxygen use and survival. There is reason to believe that continuous, 24-hours-per-day oxygen use in appropriately selected patients would produce a survival benefit even greater than that shown in the NOTT and MRC studies.[23]
Treatment
To counter the effects of high-altitude diseases, the body must return arterial pO
2 toward normal. Acclimatization, the means by which the body adapts to higher altitudes, only partially restores pO
2 to standard levels. Hyperventilation, the body’s most common response to high-altitude conditions, increases alveolar pO
2 by raising the depth and rate of breathing. However, while pO
2 does improve with hyperventilation, it does not return to normal. Studies of miners and astronomers working at 3000 meters and above show improved alveolar pO
2 with full acclimatization, yet the pO
2 level remains equal to or even below the threshold for continuous oxygen therapy for patients with chronic obstructive pulmonary disease (COPD).[24] In addition, there are complications involved with acclimatization. Polycythemia, in which the body increases the number of red blood cells in circulation, thickens the blood, raising the danger that the heart can’t pump it.
In high-altitude conditions, only oxygen enrichment can counteract the effects of hypoxia. By increasing the concentration of oxygen in the air, the effects of lower barometric pressure are countered and the level of arterial pO
2 is restored toward normal capacity. A small amount of supplemental oxygen reduces the equivalent altitude in climate-controlled rooms. At 4000 m, raising the oxygen concentration level by 5 percent via an oxygen concentrator and an existing ventilation system provides an altitude equivalent of 3000 m, which is much more tolerable for the increasing number of low-landers who work in high altitude.[25] In a study of astronomers working in Chile at 5050 m, oxygen concentrators increased the level of oxygen concentration by almost 30 percent (that is, from 21 percent to 27 percent). This resulted in increased worker productivity, less fatigue, and improved sleep.[24]
Oxygen concentrators are uniquely suited for this purpose. They require little maintenance and electricity, provide a constant source of oxygen, and eliminate the expensive, and often dangerous, task of transporting oxygen cylinders to remote areas. Offices and housing already have climate-controlled rooms, in which temperature and humidity are kept at a constant level. Oxygen can be added to this system easily and relatively cheaply.
A prescription renewal for home oxygen following hospitalization requires an assessment of the patient for ongoing hypoxemia.[26]
See also
- Anoxia (disambiguation)
- Asphyxia
- G-LOC cerebral hypoxia induced by excessive g-forces
- Hypoxia (disambiguation) subsidiary articles e.g. Intrauterine hypoxia.
- Hypoxic tumor
- Hypoxicator a device intended for hypoxia acclimatisation in a controlled manner
- Hyperoxia
- Sleep apnea
- Intermittent hypoxic training
- Hypoventilation training
- Time of useful consciousness
References
- ↑ Woorons, Xavier, Hypoventilation training, push your limits! Arpeh, 2014, 164p. (ISBN 978-2-9546040-1-5)
- ↑ West, John B. (1977). Pulmonary Pathophysiology: The Essentials. Williams & Wilkins. p. 22. ISBN 0-683-08936-6.
- ↑ Cymerman, A; Rock, PB. "Medical Problems in High Mountain Environments. A Handbook for Medical Officers". USARIEM-TN94-2. US Army Research Inst. of Environmental Medicine Thermal and Mountain Medicine Division Technical Report. Retrieved 2009-03-05.
- ↑ Gore CJ, Clark SA, Saunders PU; Clark; Saunders (September 2007). "Nonhematological mechanisms of improved sea-level performance after hypoxic exposure". Med Sci Sports Exerc 39 (9): 1600–9. doi:10.1249/mss.0b013e3180de49d3. PMID 17805094.
- ↑ Robinson first1=Grace; Strading first2=John; West first3=Sophie (2009). Oxford Handbook of Respiratory Medicine. Oxford University Press. p. 880. ISBN 0199545162.
- ↑ Bergqvist, Pia (15 April 2015). "Preventing Hypoxia: What To Do Now". Flying (magazine). Retrieved 22 April 2015.
- ↑ Robinson, Grace; Strading, John; West, Sophie (2009). Oxford Handbook of Respiratory Medicine. Oxford University Press. p. 880. ISBN 0199545162.
- ↑ Illingworth, Robin; Graham, Colin; Hogg, Kerstin (2012). Oxford Handbook of Emergency Medicine. Oxford University Press. p. 768. ISBN 0199589569.
- ↑ Hillman, Ken; Bishop, Gillian (2004). Clinical Intensive Care and Acute Medicine. Cambridge University Press. p. 685. ISBN 1139449362.
- ↑ Longmore, J.; Longmore, Murray; Wilkinson, Ian; Rajagopalan, Supraj (2006). Mini Oxford Handbook of Clinical Medicine. Oxford University Press. p. 874. ISBN 0198570716.
- ↑ Ahrens, Thomas; Rutherford Basham, Kimberley (1993). Essentials of Oxygenation: Implication for Clinical Practice. Jones & Bartlett Learning. p. 194. ISBN 0867203323.
- ↑ Ramrakha, Punit; Moore, Kevin (2004). Oxford Handbook of Acute Medicine. Oxford University Press. p. 990. ISBN 0198520727.
- ↑ Kenneth Baillie and Alistair Simpson. "Altitude oxygen calculator". Apex (Altitude Physiology Expeditions). Retrieved 2006-08-10. – Online interactive oxygen delivery calculator.
- 1 2 Martin, Lawrence (1999). All you really need to know to interpret arterial blood gases (2nd ed.). Philadelphia: Lippincott Williams & Wilkins. ISBN 0-683-30604-9.
- 1 2 3 Nicki R. Colledge, Brian R. Walker, Stuart H. Ralston, ed. (2010). Davidson's principles and practice of medicine. illustrated by Robert Britton (21st ed.). Edinburgh: Churchill Livingstone/Elsevier. ISBN 978-0-7020-3085-7.
- ↑ West, John B. (2008). Respiratory Physiology: The Essentials (8th ed.). La Jolla: Wolters Kluwer Lippincott Williams & Wilkins. pp. 88–89.
- ↑ Douglas, CG; Haldane, JS, Haldane, JB (Jun 12, 1912). "The laws of combination of hemoglobin with carbon monoxide and oxygen". The Journal of Physiology 44 (4): 275–304. doi:10.1113/jphysiol.1912.sp001517. PMC 1512793. PMID 16993128.
- 1 2 Wald, NJ; Idle, M; Boreham, J; Bailey, A (May 1981). "Carbon monoxide in breath in relation to smoking and carboxyhaemoglobin levels.". Thorax 36 (5): 366–9. doi:10.1136/thx.36.5.366. PMC 471511. PMID 7314006.
- ↑ The formula
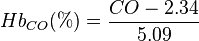 can be used to calculate the amount of carbon monoxide-bound hemoglobin. For example, at carbon monoxide level of 5 ppm,
can be used to calculate the amount of carbon monoxide-bound hemoglobin. For example, at carbon monoxide level of 5 ppm, 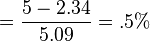 , or a loss of half a percent of their blood's hemoglobin. (Wald et al., 1981)
, or a loss of half a percent of their blood's hemoglobin. (Wald et al., 1981) - ↑ Martin, Lawrence. (1992) All you really need to know to interpret arterial blood gases. 2nd edition ISBN 0-683-30604-9
- ↑ Roueché, Berton (1953) Eleven blue men, and other narratives of medical detection. Boston: Little, Brown.
- ↑ Hobler, K.E.; L.C. Carey (1973). "Effect of acute progressive hypoxemia on cardiac output and plasma excess lactate". Ann Surg 177 (2): 199–202. doi:10.1097/00000658-197302000-00013. PMC 1355564. PMID 4572785.
- ↑ "Pathophysiology and clinical effects of chronic hypoxia". Respir Care 45: 39–51; discussion 51–3. 2000. PMID 10771781.
- 1 2 West, John B.; American College Of Physicians; American Physiological Society (2004). "The Physiologic Basis of High-Altitude Diseases". Annals of Internal Medicine 141 (10): 789–800. doi:10.7326/0003-4819-141-10-200411160-00010. PMID 15545679.
- ↑ West, John B. (1995). "Oxygen Enrichment of Room Air to Relieve the Hypoxia of High Altitude". Respiration Physiology 99 (2): 225–32. doi:10.1016/0034-5687(94)00094-G. PMID 7777705.
- ↑ -->American College of Chest Physicians; American Thoracic Society (September 2013), "Five Things Physicians and Patients Should Question", Choosing Wisely: an initiative of the ABIM Foundation (American College of Chest Physicians and American Thoracic Society), retrieved 6 January 2013
| ||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
