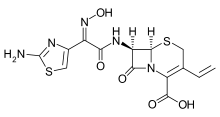
Cefdinir
 | |
| Systematic (IUPAC) name | |
|---|---|
|
8-[2-(2-amino-1,3-thiazol-4-yl)-1-hydroxy-2-nitroso- ethenyl]amino-4-ethenyl-7-oxo-2-thia-6- azabicyclo[4.2.0]oct-4-ene-5-carboxylic acid | |
| Clinical data | |
| Trade names | Cefzon, Omnicef |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a698001 |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration | Oral |
| Pharmacokinetic data | |
| Bioavailability | 16% to 21% (dose-dependent) |
| Protein binding | 60% to 70% |
| Metabolism | Negligible |
| Biological half-life | 1.7 ± 0.6 hours |
| Excretion | Renal |
| Identifiers | |
| CAS Number |
91832-40-5 |
| ATC code | J01DD15 |
| PubChem | CID 6915944 |
| DrugBank |
DB00535 |
| ChemSpider |
5291705 |
| UNII |
CI0FAO63WC |
| KEGG |
D00917 |
| ChEBI |
CHEBI:3485 |
| ChEMBL |
CHEMBL927 |
| Chemical data | |
| Formula | C14H13N5O5S2 |
| Molar mass | 395.416 g/mol |
| |
| |
| (verify) | |
Cefdinir is a third-generation oral cephalosporin antibiotic sold under the brand names Cefzon and Omnicef.
As of 2008, cefdinir, as Omnicef, was the highest-selling cephalosporin antibiotic in the United States, with more than US$585 million in retail sales of its generic versions alone.[1] Cephalosporin antibiotic structurally similar to cefixime.
It was discovered by Fujisawa Pharmaceutical Co., Ltd. (now Astellas) and introduced in 1991 under the brand name Cefzon.[2][3] Warner-Lambert licensed this cephalosporin for marketing in US from Fujisawa.[4] Abbott obtained U.S. marketing rights to Omnicef (cefdinir) in December 1998 through an agreement with Warner-Lambert Company.[5] It was approved by FDA on Dec 4, 1997.[6] It is available in US as Omnicef by Abbott Laboratories and in India as Cednir by Abbott, Kefnir by Glenmark, Cefdair by Xalra Pharma and Cefdiel by Ranbaxy.
Mechanism of action
Indications
Therapeutic uses of cefdinir include otitis media, soft tissue infections, and respiratory tract infections, including sinusitis, strep throat (note: no documented resistance of Group A Streptococcus to penicillin has ever been reported, and penicillin or amoxicillin is the first line treatment), community-acquired pneumonia, and acute exacerbations of bronchitis.
Susceptible organisms
Cefdinir is a bacteriocidal antibiotic. It can be used to treat infections caused by several Gram-negative and Gram-positive bacteria.
Spectrum of bacterial susceptibility and resistance
Cefdinir is a broad-spectrum antibiotic and has been used to treat infections of the respiratory tract including pneumonia, sinusitis, and bronchitis. The following represents MIC susceptibility data for a few medically significant microorganisms.[7]
- Haemophilus influenzae: 0.05 - 4 μg/ml
- Streptococcus pneumoniae: 0.006 - 64 μg/ml
- Streptococcus pyogenes: ≤0.004 - 2 μg/ml
Side effects
Side effects include diarrhea, vaginal infections or inflammation, nausea, headache, and abdominal pain."[8]
Available forms
Cefdinir is administered orally. It is available as capsules and a suspension. Dosage, schedule, and duration of therapy varies according to the type of infection.
"Blood" in the stool
The pediatric version of cefdinir can bind to iron in the digestive tract; in rare cases, this causes a rust or red discoloration of the stool. Blood typically appears dark brown or black in stool, and testing may confirm which is present. If the reddish stool is accompanied by abdominal pain, weight loss, diarrhea, etc., a Clostridium difficile infection caused by the antibiotic could be signified. Special caution required for pediatric dry powder; it may take longer for proper mixing.
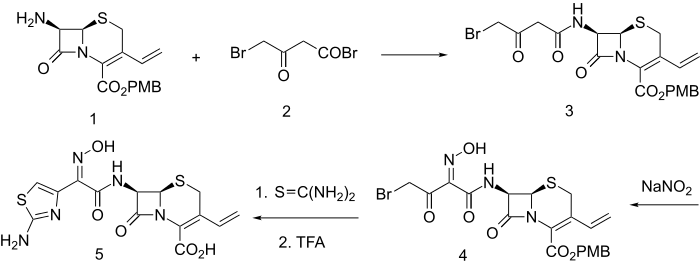
Synthesis
Acylation of the primary amine 1 with 4-bromo-3-oxobutanoyl bromide (2) leads to the amide (3). The active methylene group in that product is then nitrosated with sodium nitrite; the initial product spontaneously tautomerizes to afford the oxime (4). The bromoketone array in that intermediate constitutes a classical starting function for construction of thiazoles. Reaction of 4 with thiourea thus leads to formation of an aminothiazole moiety. Thus there is obtained the antibiotic cefdinir (5).
References
- ↑ "2008 Top 200 generic drugs by retail dollars" PDF (399.4 KB). Drug Topics (May 26, 2009). Retrieved on July 24, 2009.
- ↑ "2005 News Releases - Astellas Pharma Inc.". astellas.com.
- ↑ Cabri, Walter; Ghetti, Paolo; Pozzi, Giovanni; Alpegiani, Marco (2007). "Polymorphisms and Patent, Market, and Legal Battles: Cefdinir Case Study". Organic Process Research & Development 11: 64. doi:10.1021/op0601060.
- ↑ "Document". elsevierbi.com.
- ↑ "Medicis.com - Medicis Pharmaceutical Corporation". globenewswire.com.
- ↑ "Cefdinir medical facts from Drugs.com". drugs.com.
- ↑ "Susceptibility and Minimum Inhibitory Concentration (MIC) Data" (PDF). October 14, 2015. Retrieved October 2015.
- ↑ "Omnicef capsules Patient Information" (PDF). Abbott Laboratories. February 2004. Archived from the original (PDF) on 2006-11-18. Retrieved 2006-11-24.
- ↑ T. Takaya et al., BE 897864; eidem, U.S. Patent 4,559,334 (1984, 1985 both to Fujisawa).
- ↑ Inamoto, Y; Chiba, T; Kamimura, T; Takaya, T (1988). "FK 482, a new orally active cephalosporin synthesis and biological properties". The Journal of antibiotics 41 (6): 828–30. PMID 3255303.
- ↑ Kamachi, H; Narita, Y; Okita, T; Abe, Y; Iimura, S; Tomatsu, K; Yamasaki, T; Okumura, J; Naito, T; Oki, T (1988). "Synthesis and biological activity of a new cephalosporin, BMY-28232 and its prodrug-type esters for oral use". The Journal of antibiotics 41 (11): 1602–16. PMID 3198494.
- ↑ González, M; Rodríguez, Z; Tolón, B; Rodríguez, J. C.; Velez, H; Valdés, B; López, M. A.; Fini, A (2003). "An alternative procedure for preparation of cefdinir". Il Farmaco 58 (6): 409–18. doi:10.1016/S0014-827X(03)00063-6. PMID 12767379.
External links
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||