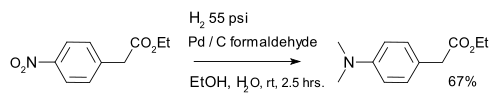Nitro compound

Nitro compounds are organic compounds that contain one or more nitro functional groups (–NO2). They are often highly explosive, especially when the compound contains more than one nitro group and is impure. The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. This property of both nitro and nitrate groups is because their thermal decomposition yields molecular nitrogen N2 gas plus considerable energy, due to the high strength of the bond in molecular nitrogen.
The nitro group is also strongly electron-withdrawing. This makes the C-H bonds alpha to the nitro group acidic. The pKas of nitromethane and isopropyl nitrate, reported by Matthews and Bordwell respectively, are 17.2 and 16.9. [1][2][3]
Aromatic nitro compounds are typically synthesized by the action of a mixture of nitric and sulfuric acids on an organic molecule. The one produced on the largest scale, by far, is nitrobenzene. Many explosives are produced by nitration including trinitrophenol (picric acid), trinitrotoluene (TNT), and trinitroresorcinol (styphnic acid).[4]
Occurrence in nature
Chloramphenicol is a rare example of a naturally occurring nitro compound. At least some naturally occurring nitro groups arise by the oxidation of amino groups.[5] 2-Nitrophenol is an aggregation pheromone of ticks.
Examples of nitro compounds are rare in nature. 3-Nitropropionic acid found in fungi and plants (Indigofera). Nitropentadecene is a defense compound found in termites. Nitrophenylethane is found in Aniba canelilla.[6] Nitrophenylethane is also found in members of the Annonaceae, Lauraceae and Papaveraceae.[7]
Many flavin-dependent enzymes are capable of oxidizing aliphatic nitro compounds to less-toxic aldehydes and ketones. Nitroalkane oxidase and 3-nitropropionate oxidase oxidize aliphatic nitro compounds exclusively, whereas other enzymes such as glucose oxidase have other physiological substrates.[8]
Preparation
In organic synthesis various methods exists to prepare nitro compounds.
Aliphatic nitro compounds
Aliphatic nitro compounds can be synthesized by various methods, notable examples include:
- Free radical nitration of alkanes.[9] The reaction produces fragments from the parent alkane, creating a diverse mixture of products; for instance, Nitromethane, Nitroethane, 1-Nitropropane, and 2-Nitropropane are produced by treating propane with nitric acid in the gas phase (e.g. 350-450 °C between 8-12 atm).
- Nucleophilic substitution reactions between halocarbons[10] or organosulfates[11] with silver or alkali nitrite salts.
- Nitromethane can be produced in the laboratory by treating sodium chloroacetate with sodium nitrite.[12]
- Oxidation of oximes[13] or primary amines.[14]
Aromatic nitro compounds
In a classic electrophilic substitution reaction, nitric acid and sulfuric acid produce the nitronium ion, which reacts with aromatic compounds in aromatic nitration. Another method, starting from halogenated phenols, is the Zinke nitration.
Reactions
Nitro compounds participate in several organic reactions, the most important being their reduction to the corresponding amines:
- RNO2 + 3 H2 → RNH2 + 2 H2O
Virtually all aromatic amines (anilines) are derived from nitroaromatics.
Aliphatic nitro compounds
- Aliphatic nitro compounds are reduced to amines with hydrochloric acid and an iron catalyst
- Nitronates are a tautomeric form of aliphatic nitro compounds.
- Hydrolysis of the salts of nitro compounds yield aldehydes or ketones in the Nef reaction
- Nitromethane adds to aldehydes in 1,2-addition in the nitroaldol reaction
- Nitromethane adds to alpha-beta unsaturated carbonyl compounds as a 1,4-addition in the Michael reaction as a Michael donor
- Nitroalkenes are Michael acceptors in the Michael reaction with enolate compounds[15][16]
- In nucleophilic aliphatic substitution, sodium nitrite (NaNO2) replaces an alkyl halide. In the so-called ter Meer reaction (1876) named after Edmund ter Meer,[17] the reactant is a 1,1-halonitroalkane:
- In one study, a reaction mechanism is proposed in which in the first slow step a proton is abstracted from nitroalkane 1 to a carbanion 2 followed by protonation to a nitronate 3 and finally nucleophilic displacement of chlorine based on an experimentally observed hydrogen kinetic isotope effect of 3.3.[18] When the same reactant is reacted with potassium hydroxide the reaction product is the 1,2-dinitro dimer[19]
Aromatic nitro compounds
- Reduction of aromatic nitro compounds with hydrogen gas over a platinum catalyst gives anilines. A variation is formation of a dimethylaminoarene with palladium on carbon and formaldehyde:[20]
- The Leimgruber-Batcho, Bartoli and Baeyer-Emmerling indole syntheses begin with aromatic nitro compounds.
- Indigo can be synthesized in a condensation reaction from ortho-nitrobenzaldehyde and acetone in strongly basic conditions in a reaction known as the Baeyer-Drewson indigo synthesis
- The presence of nitro groups retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution because they are highly electron-withdrawing.
See also
- Functional group
- Reduction of nitro compounds
- Nitration
- Nitrite Also an NO2 group, but bonds differently.
- Nitroalkene
- Nitroglycerin
References
- ↑ Reich, Hans. "Bordwell pKa table: "Nitroalkanes"". University of Wisconsin Chemistry Department. Retrieved 17 January 2016.
- ↑ Matthews, Walter; et al. (1975). "Equilibrium acidities of carbon acids. VI. Establishment of an absolute scale of acidities in dimethyl sulfoxide solution". Journal of the American Chemical Society 97 (24): 7006. doi:10.1021/ja00857a010. Retrieved 17 January 2016.
- ↑ Bordwell, F.G. (1975). "Acidities of carbon acids. VII. Conjugation and strain in some cyclopropyl anions.". Journal of the American Chemical Society 97 (24): 7160–7162. doi:10.1021/ja00857a033. Retrieved 17 January 2016.
- ↑ Gerald Booth "Nitro Compounds, Aromatic" Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a17_411
- ↑ Georg Zocher, Robert Winkler, Christian Hertweck, Georg E. Schulz "Structure and Action of the N-oxygenase AurF from Streptomyces thioluteus" J. Molecular Biology (2007) 373, 65–74. doi:10.1016/j.jmb.2007.06.014
- ↑ José Guilherme S. Maia, Eloísa Helena A. Andrade "Database of the Amazon aromatic plants and their essential oils " Quim. Nova, (2009) 32(3), 595-622, 2009 {http://www.scielo.br/pdf/qn/v32n3/a06v32n3.pdf}
- ↑ Klaus Kubitzki, Jens G. Rohwer, Volker Bittrich "Flowering Plants · Dicotyledons: Magnoliid, Hamamelid and Caryophyllid Families" 1993,Springer-Verlag, Berlin
- ↑ Nagpal, Akanksha; Michael P. Valley; Paul F. Fitzpatrick; Allen M. Orville (1/5/2006). "Crystal Structures of Nitroalkane Oxidase: Insights into the Reaction Mechanism from a Covalent Complex of the Flavoenzyme Trapped during Turnover". Biochemistry 45 (4): 1138–50. doi:10.1021/bi051966w. PMC 1855086. PMID 16430210. Check date values in:
|date=(help) - ↑ Markofsky, Sheldon; Grace, W.G. (2000). "Nitro Compounds, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a17_401.
- ↑ Kornblum, N.; Ungnade, H. E. (1963). "1-Nitroöctane". Organic Syntheses 4: 724. doi:10.15227/orgsyn.038.0075.
- ↑ Walden, P. (1907). "Zur Darstellung aliphatischer Sulfocyanide, Cyanide und Nitrokörper". Berichte der deutschen chemischen Gesellschaft 40 (3): 3214–3217. doi:10.1002/cber.19070400383.
- ↑ Whitmore, F. C.; Whitmore, Marion G. (1923). "Nitromethane". Organic Syntheses 1: 401. doi:10.15227/orgsyn.003.0083.
- ↑ Olah, George A.; Ramaiah, Pichika; Chang-Soo, Lee; Prakash, Surya (1992). "Convenient Oxidation of Oximes to Nitro Compounds with Sodium Perborate in Glacial Acetic Acid". Synlett 4: 337–339. doi:10.1055/s-1992-22006.
- ↑ Ehud, Keinan; Yehuda, Mazur (1977). "Dry ozonation of amines. Conversion of primary amines to nitro compounds". The Journal of Organic Chemistry 42 (5): 844–847. doi:10.1021/jo00425a017.
- ↑ Ranganathan, Darshan; Rao, Bhushan; Ranganathan, Subramania; Mehrotra, Ashok; and Iyengar, Radha (1980). "Nitroethylene: a stable, clean, and reactive agent for organic synthesis". The Journal of Organic Chemistry 45 (7): 1185–1189. doi:10.1021/jo01295a003. Retrieved 5 January 2014.
- ↑ Jubert, Carole and Knochel, Paul (1992). "Preparation of polyfunctional nitro olefins and nitroalkanes using the copper-zinc reagents RCu(CN)ZnI". The Journal of Organic Chemistry 57 (20): 5431–5438. doi:10.1021/jo00046a027. Retrieved 5 January 2014.
- ↑ Edmund ter Meer (1876). "Ueber Dinitroverbindungen der Fettreihe". Justus Liebigs Annalen der Chemie 181 (1): 1–22. doi:10.1002/jlac.18761810102.
- ↑ aci-Nitroalkanes. I. The Mechanism of the ter Meer Reaction M. Frederick Hawthorne J. Am. Chem. Soc.; 1956; 78(19) pp 4980 – 4984; doi:10.1021/ja01600a048
- ↑ 3-Hexene, 3,4-dinitro- D. E. Bisgrove, J. F. Brown, Jr., and L. B. Clapp. Organic Syntheses, Coll. Vol. 4, p.372 (1963); Vol. 37, p.23 (1957). (Article)
- ↑ Organic Syntheses, Coll. Vol. 5, p.552 (1973); Vol. 47, p.69 (1967). http://orgsynth.org/orgsyn/pdfs/CV5P0552.pdf
| Wikimedia Commons has media related to Nitro compounds. |
|