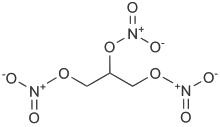
Nitroglycerin
 | |
.png) | |
 | |
| Names | |
|---|---|
| IUPAC name
1,2,3-Trinitroxypropane | |
| Systematic IUPAC name
2,3-Bis(nitrooxy)propyl nitrate | |
| Other names
1,3-Dinitrooxypropan-2-yl nitrate Propane-1,2,3-triyl trinitrate | |
| Identifiers | |
| 55-63-0 | |
| 1802063 | |
| ChEBI | CHEBI:28787 |
| ChEMBL | ChEMBL730 |
| ChemSpider | 4354 |
| DrugBank | DB00727 |
| EC Number | 200-240-8 |
| 165859 | |
| 7053 | |
| Jmol interactive 3D | Image Image |
| KEGG | D00515 |
| MeSH | Nitroglycerin |
| PubChem | 4510 |
| UNII | G59M7S0WS3 |
| UN number | 0143, 0144, 1204, 3064, 3319 |
| |
| |
| Properties | |
| C3H5N3O9 | |
| Molar mass | 227.09 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 1.6 g cm−3 (at 15 °C) |
| Melting point | 14 °C (57 °F; 287 K) |
| Boiling point | 50 °C (122 °F; 323 K) explodes |
| slightly[1] | |
| Solubility | acetone, ether, benzene, alcohol[1] |
| log P | 2.154 |
| Structure | |
| Tetragonal at C1, C2, and C3 Trigonal planar at N7, N8, and N9 | |
| Tetrahedral at C1, C2, and C3 Dihedral at N7, N8, and N9 | |
| Explosive data | |
| Shock sensitivity | High |
| Friction sensitivity | High |
| Detonation velocity | 7700 m s−1 |
| RE factor | 1.50 |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH |
−370 kJ mol−1 |
| Std enthalpy of combustion (ΔcH |
−1.529 MJ mol−1 |
| Pharmacology | |
| ATC code | C01 C05AE01 |
| Legal status |
|
| |
| Intravenous, Oral, Sublingual, Topical, Transdermal | |
| Pharmacokinetics: | |
| <1% | |
| Hepatic | |
| 3 min | |
| Hazards | |
| Main hazards | Explosive |
| EU classification (DSD) |
|
| R-phrases | R3, R12, R26/27/28, R33, R51/53 |
| S-phrases | (S1/2), S33, S35, S36/37, S45, S53, S61 |
| NFPA 704 | |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
C 0.2 ppm (2 mg/m3) [skin][2] |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Nitroglycerin (NG), also known as nitroglycerine, trinitroglycerin (TNG), trinitroglycerine, nitro, glyceryl trinitrate (GTN), or 1,2,3-trinitroxypropane, is a heavy, colorless, oily, explosive liquid most commonly produced by nitrating glycerol with white fuming nitric acid under conditions appropriate to the formation of the nitric acid ester. Chemically, the substance is an organic nitrate compound rather than a nitro compound, yet the traditional name is often retained. Invented in 1847, nitroglycerin has been used as an active ingredient in the manufacture of explosives, mostly dynamite, and as such it is employed in the construction, demolition, and mining industries. Since the 1880s, it has been used by the military as an active ingredient, and a gelatinizer for nitrocellulose, in some solid propellants, such as cordite and ballistite.
Nitroglycerin is also a major component in double-based smokeless gunpowders used by reloaders. Combined with nitrocellulose, there are hundreds of (powder) combinations used by rifle, pistol, and shotgun reloaders.
For over 130 years, nitroglycerin has been used medically as a potent vasodilator to treat heart conditions, such as angina pectoris and chronic heart failure. Though it was previously known that these beneficial effects are due to nitroglycerin being converted to nitric oxide, a potent vasodilator, it was not until 2002 that the enzyme for this conversion was discovered to be mitochondrial aldehyde dehydrogenase.[3] Nitroglycerin is available in sublingual tablets, sprays, and patches.[4] Other potential suggested uses include adjunct therapy in prostate cancer.[5]
History
Nitroglycerin was the first practical explosive produced that was stronger than black powder. It was first synthesized by the Italian chemist Ascanio Sobrero in 1847, working under Théophile-Jules Pelouze at the University of Turin. Sobrero initially called his discovery pyroglycerine and warned vigorously against its use as an explosive.
Nitroglycerin was later adopted as a commercially useful explosive by Alfred Nobel, who experimented with safer ways to handle the dangerous compound after his younger brother, Emil Oskar Nobel, and several factory workers were killed in an explosion at the Nobels' armaments factory in 1864 in Heleneborg, Sweden.[6]
One year later, Nobel founded Alfred Nobel & Company in Germany and built an isolated factory in the Krümmel hills of Geesthacht near Hamburg. This business exported a liquid combination of nitroglycerin and gunpowder called "Blasting Oil", but this was extremely unstable and difficult to handle, as evidenced in numerous catastrophes. The buildings of the Krümmel factory were destroyed twice.[7]
In April 1866, three crates of nitroglycerin were shipped to California for the Central Pacific Railroad, which planned to experiment with it as a blasting explosive to expedite the construction of the 1,659-foot (506 m)-long Summit Tunnel through the Sierra Nevada Mountains. One of the crates exploded, destroying a Wells Fargo company office in San Francisco and killing 15 people. This led to a complete ban on the transportation of liquid nitroglycerin in California. The on-site manufacture of nitroglycerin was thus required for the remaining hard-rock drilling and blasting required for the completion of the First Transcontinental Railroad in North America.[8]
Liquid nitroglycerin was widely banned elsewhere as well, and these legal restrictions led to Alfred Nobel and his company's developing dynamite in 1867. This was made by mixing nitroglycerin with diatomaceous earth ("kieselgur" in German) found in the Krümmel hills. Similar mixtures, such as "dualine" (1867), "lithofracteur" (1869), and "gelignite" (1875), were formed by mixing nitroglycerin with other inert absorbents, and many combinations were tried by other companies in attempts to get around Nobel's tightly held patents for dynamite.
Dynamite mixtures containing nitrocellulose, which increases the viscosity of the mix, are commonly known as "gelatins".
Following the discovery that amyl nitrite helped alleviate chest pain, Dr. William Murrell experimented with the use of nitroglycerin to alleviate angina pectoris and to reduce the blood pressure. He began treating his patients with small diluted doses of nitroglycerin in 1878, and this treatment was soon adopted into widespread use after Murrell published his results in the journal The Lancet in 1879.[9] A few months before his death in 1896, Alfred Nobel was prescribed nitroglycerine for this heart condition, writing to a friend: "Isn't it the irony of fate that I have been prescribed nitro-glycerin, to be taken internally! They call it Trinitrin, so as not to scare the chemist and the public." [10] The medical establishment also used the name "glyceryl trinitrate" for the same reason.
Wartime production rates
Large quantities of nitroglycerin were manufactured during World War I and World War II for use as military propellants and in military engineering work. During World War I, HM Factory, Gretna, the largest propellant factory in Great Britain, produced about 800 tonne of Cordite RDB per week. This amount took at least 336 tons of nitroglycerin per week (assuming no losses in production). The Royal Navy had its own factory at the Royal Navy Cordite Factory, Holton Heath in Dorset, England. A large cordite factory was also built in Canada during World War I. The Canadian Explosives Limited cordite factory at Nobel, Ontario, was designed to produce 1,500,000 lb (680 t) of cordite per month, requiring about 286 tonnes of nitroglycerin per month.
Instability and desensitization
In its pure form, nitroglycerin is a contact explosive, with physical shock causing it to explode, and it degrades over time to even more unstable forms. This makes nitroglycerin highly dangerous to transport or use. In its undiluted form, it is one of the world's most powerful explosives, comparable to the more recently developed RDX and PETN.
Early in its history, it was discovered that liquid nitroglycerin can be "desensitized" by cooling it to about 5 to 10 °C (40 to 50 °F). At this temperature nitroglycerin freezes, contracting upon solidification. Thawing it out can be extremely sensitizing, especially if impurities are present or the warming is too rapid.[11] It is possible to chemically "desensitize" nitroglycerin to a point where it can be considered approximately as "safe" as modern high explosives, such as by the addition of approximately 10% to 30% ethanol, acetone,[12] or dinitrotoluene. (The percentage varies with the desensitizing agent used.) Desensitization requires extra effort to reconstitute the "pure" product. Failing this, it must be assumed that desensitized nitroglycerin is substantially more difficult to detonate, possibly rendering it useless as an explosive for practical application.
A serious problem in the use of nitroglycerin results from its high freezing point 13 °C (55 °F). Solid nitroglycerin is much less sensitive to shock than the liquid, a feature that is common in explosives. In the past, nitroglycerin was often shipped in the frozen state, but this resulted in a high number of accidents during the thawing process just before its use. This disadvantage is overcome by using mixtures of nitroglycerin with other polynitrates. For example, a mixture of nitroglycerin and ethylene glycol dinitrate freezes at −29 °C (−20 °F).[13]
Detonation
Nitroglycerin and any diluents can certainly deflagrate, i.e., burn. The explosive power of nitroglycerin derives from detonation: energy from the initial decomposition causes a strong pressure wave that detonates the surrounding fuel. This is a self-sustained shock wave that propagates through the explosive medium at some 30 times the speed of sound as a near-instantaneous pressure-induced decomposition of the fuel into a white-hot gas. Detonation of nitroglycerin generates gases that would occupy more than 1,200 times the original volume at ordinary room temperature and pressure. The heat liberated raises the temperature to about 5,000 °C (9,000 °F).[14] This is entirely different from deflagration, which depends solely upon available fuel regardless of pressure or shock. The decomposition results in much higher ratio of energy to gas moles released compared to other explosives, making it one of the hottest detonating high explosives.
Manufacturing
Nitroglycerin can be produced by acid catalyzed nitration of glycerol (glycerine).
The industrial manufacturing process often reacts glycerol with a nearly 1:1 mixture of concentrated sulfuric acid and concentrated nitric acid. This can be produced by mixing white fuming nitric acid—a quite expensive pure nitric acid in which the oxides of nitrogen have been removed, as opposed to red fuming nitric acid, which contains nitrogen oxides—and concentrated sulfuric acid. More often, this mixture is attained by the cheaper method of mixing fuming sulfuric acid, also known as oleum—sulfuric acid containing excess sulfur trioxide—and azeotropic nitric acid (consisting of about 70 percent nitric acid, with the rest being water).
The sulfuric acid produces protonated nitric acid species, which are attacked by glycerol's nucleophilic oxygen atoms. The nitro group is thus added as an ester C-O-NO2 and water is produced. This is different from an electrophilic aromatic substitution reaction in which nitronium ions are the electrophile.
The addition of glycerol results in an exothermic reaction (i.e., heat is produced), as usual for mixed-acid nitrations. However, if the mixture becomes too hot, it results in "runaway", a state of accelerated nitration accompanied by the destructive oxidation of organic materials by the hot nitric acid and the release of poisonous nitrogen dioxide gas at high risk of an explosion. Thus, the glycerin mixture is added slowly to the reaction vessel containing the mixed acid (not acid to glycerin). The nitrator is cooled with cold water or some other coolant mixture and maintained throughout the glycerin addition at about 22 °C (72 °F), much below which the esterification occurs too slowly to be useful. The nitrator vessel, often constructed of iron or lead and generally stirred with compressed air, has an emergency trap door at its base, which hangs over a large pool of very cold water and into which the whole reaction mixture (called the charge) can be dumped to prevent an explosion, a process referred to as drowning. If the temperature of the charge exceeds about 30 °C (86 °F) (actual value varying by country) or brown fumes are seen in the nitrator's vent, then it is immediately drowned.
Use as an explosive and a propellant

The main use of nitroglycerin, by tonnage, is in explosives such as dynamite and in propellants.
Nitroglycerin is an oily liquid that may explode when subjected to heat, shock or flame. It is dangerously sensitive and dropping or bumping a container may cause it to explode.[18]
Alfred Nobel developed the use of nitroglycerin as a blasting explosive by mixing the nitroglycerin with inert absorbents, particularly "kieselguhr," or diatomaceous earth. He named this explosive dynamite and patented it in 1867.[19] It was supplied ready for use in the form of sticks, individually wrapped in greased waterproof paper. Dynamite and similar explosives were widely adopted for civil engineering tasks, such as in drilling highway and railroad tunnels, for mining, for clearing farmland of stumps, in quarrying, and in demolition work. Likewise, military engineers have used dynamite for construction and demolition work.
Nitroglycerin was also used as an ingredient in military propellants for use in firearms.
Nitroglycerin has been used in conjunction with hydraulic fracturing, a process used to recover oil and gas from shale formations. The technique involves displacing and detonating nitroglycerin in natural or hydraulically induced fracture systems, or displacing and detonating nitroglycerin in hydraulically induced fractures followed by wellbore shots using pelletized TNT.[20]
Nitroglycerin has an advantage over some other high explosives, that on detonation it produces practically no visible smoke. Therefore, it is useful as an ingredient in the formulation of various kinds of "smokeless powder".[21]
Its sensitivity has limited the usefulness of nitroglycerin as a military explosive, and less sensitive explosives such as TNT, RDX, and HMX have largely replaced it in munitions. It remains important in military engineering, and combat engineers still use dynamite.
Alfred Nobel then developed ballistite, by combining nitroglycerin and guncotton. He patented it in 1887. Ballistite was adopted by a number of European governments, as a military propellant. Italy was the first to adopt it. The British Government and the Commonwealth governments adopted cordite instead, which had been developed by Sir Frederick Abel and Sir James Dewar of the United Kingdom in 1889. The original Cordite Mk I consisted of 58% nitroglycerin, 37% guncotton, and 5.0% petroleum jelly. Ballistite and cordite were both manufactured in the forms of cords.
Smokeless powders were originally developed using nitrocellulose as the sole explosive ingredient. Therefore, they were known as single-base propellants. A range of smokeless powders that contain both nitrocellulose and nitroglycerin, known as double-base propellants, were also developed. Smokeless powders were originally supplied only for military use, but they were also soon developed for civilian use and were quickly adopted for sports. Some are known as sporting powders. Triple-base propellants contain nitrocellulose, nitroglycerin, and nitroguanidine, but are reserved mainly for extremely high-caliber ammunition rounds such as those used in tank cannons and naval artillery.
Blasting gelatin, also known as gelignite, was invented by Nobel in 1875, using nitroglycerin, wood pulp, and sodium or potassium nitrates. This was an early low-cost, flexible explosive.
Medical use
Nitroglycerin belongs to a group of drugs called nitrates, which includes many other nitrates like isosorbide dinitrate (Isordil) and isosorbide mononitrate (Imdur, Ismo, Monoket).[22] These agents all exert their effect by being converted to nitric oxide in the body by mitochondrial aldehyde dehydrogenase,[3] and nitric oxide is a potent natural vasodilator.
In medicine, nitroglycerin is used as a medicine for angina pectoris, a painful symptom of ischemic heart disease caused by inadequate flow of blood and oxygen to the heart. Nitroglycerin corrects the imbalance between the flow of oxygen and blood to the heart.[23] The principal action of nitroglycerin is vasodilation (widening of the blood vessels). At low doses, nitroglycerin will dilate veins more than arteries, thereby reducing preload; this is thought to be its primary mechanism of action. But at higher doses, it also dilates arteries, thereby reducing afterload. It is also a potent antihypertensive agent. In cardiac treatment, the lowering of pressure in the arteries reduces the pressure against which the heart must pump, thereby decreasing afterload.[22] Dilating the veins decreases cardiac preload and lowers the oxygen requirement of the heart whilst at the same time reducing ventricular transmural pressure thereby improving coronary blood flow. Improved myocardial oxygen demand vs oxygen delivery ratio leads to the following therapeutic effects during episodes of angina pectoris: subsiding of chest pain, decrease of blood pressure, increase of heart rate, and orthostatic hypotension. Patients experiencing angina when doing certain physical activities can often prevent symptoms by taking nitroglycerin 5 to 10 minutes before the activity.
Nitroglycerin is available in tablets, ointment, solution for intravenous use, transdermal patches, or sprays administered sublingually. Some forms of nitroglycerin last much longer in the body than others. It has been shown that continuous exposure to nitrates can cause the body to stop responding normally to this medicine. Experts recommend that the patches be removed at night, allowing the body a few hours to restore its responsiveness to nitrates. Shorter-acting preparations can be used several times a day with less risk of the body's getting used to this drug.[24] Nitroglycerin was first used by William Murrell to treat angina attacks in 1878, with the discovery published that same year.[9][25]
Industrial exposure
Infrequent exposure to high doses of nitroglycerin can cause severe headaches known as "NG head" or "bang head". These headaches can be severe enough to incapacitate some people; however, humans develop a tolerance to and dependence on nitroglycerin after long-term exposure. Withdrawal can (rarely) be fatal;[26] withdrawal symptoms include chest pain and heart problems and if unacceptable may be treated with re-exposure to nitroglycerin or other suitable organic nitrates.[27]
For workers in nitroglycerin (NTG) manufacturing facilities, the effects of withdrawal sometimes include "Sunday Heart Attacks" in those experiencing regular nitroglycerin exposure in the workplace, leading to the development of tolerance for the vasodilating effects. Over the weekend, the workers lose the tolerance and, when they are re-exposed on Monday, the drastic vasodilation produces a fast heart rate, dizziness, and a headache, this is referred to as "Monday Disease."[28][29]
People can be exposed to nitroglycerin in the workplace by breathing it in, skin absorption, swallowing it, or eye contact. The Occupational Safety and Health Administration (OSHA) has set the legal limit (permissible exposure limit) for nitroglycerin exposure in the workplace as 0.2 ppm (2 mg/m3) skin exposure over an 8-hour workday. The National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) of 0.1 mg/m2 skin exposure over an 8-hour workday. At levels of 75 mg/m3, nitroglycerin is immediately dangerous to life and health.[30]
See also
- Erythritol tetranitrate
- Ethylene glycol dinitrate
- Mannitol hexanitrate
- Methyl nitrate
- Tetranitratoxycarbon
- Xylitol pentanitrate
- RE factor
References
- 1 2 https://www.osha.gov/SLTC/healthguidelines/nitroglycerin/recognition.html
- ↑ "NIOSH Pocket Guide to Chemical Hazards #0456". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 Chen, Z; Foster, MW; Zhang, J; Mao, L; Rockman, HA; Kawamoto, T; Kitagawa, K; Nakayama, KI; et al. (2005). "An essential role for mitochondrial aldehyde dehydrogenase in nitroglycerin bioactivation". Proc. Natl. Acad. Sci. USA 102 (34): 12159–12164. Bibcode:2005PNAS..10212159C. doi:10.1073/pnas.0503723102. PMC 1189320. PMID 16103363.
- ↑ "Bookmarkable URL intermediate page".
- ↑ Daily Mail: "How dynamite could help destroy prostate cancer" Retrieved 2010-02-23
- ↑ NobelPrize.org: Emil Nobel.
- ↑ NobelPrize.org: Krümmel.
- ↑ "Transcontinental Railroad – People & Events: Nitroglycerin", American Experience, PBS.
- 1 2 Sneader, Walter. Drug Discovery: A History. John Wiley and Sons, 2005 ISBN 0-471-89980-1.
- ↑ History of TNG
- ↑ "Tales of Destruction-Thawing can be Hell".
- ↑ "Tales of Destruction – Is Nitroglicerine in This?".
- ↑ "nitroglycerin". Britannica. Retrieved 2005-03-23.
- ↑ Encyclopaedia Britannica.
- ↑ "Zusammensetzung der Zuckerasche". Annalen der Chemie und Pharmacie 64 (3): 398–399. 1848. doi:10.1002/jlac.18480640364.
- ↑ "Ueber Nitroglycerin". Annalen der Chemie und Pharmacie 92 (3): 305–306. 1854. doi:10.1002/jlac.18540920309.
- ↑ Di Carlo, F. J. (1975). "Nitroglycerin Revisited: Chemistry, Biochemistry, Interactions". Drug Metabolism Reviews 4 (1): 1–38. doi:10.3109/03602537508993747. PMID 812687.
- ↑ "Bookmarkable URL intermediate page".
- ↑ Mary Bellis. "Alfred Nobel and the History of Dynamite". About.com Money.
- ↑ Miller, J. S.; Johansen, R. T. (1976). "Fracturing Oil Shale with Explosives for In Situ Recovery." (PDF). Shale Oil, Tar Sand and Related Fuel Sources (American Chemical Society): 151. Retrieved 27 March 2015.
- ↑ "Nitroglycerin".
- 1 2 Omudhome Ogbru, PharmD. "nitroglycerin, Nitro-Bid: Drug Facts, Side Effects and Dosing". MedicineNet.
- ↑ Omudhome Ogbru, PharmD. "nitroglycerin, Nitro-Bid: Drug Facts, Side Effects and Dosing". MedicineNet.
- ↑ Nitroglycerin for angina, February 1997, Vol. 7.
- ↑ Smith, E; Hart, F. D. (1971). "William Murrell, physician and practical therapist". British Medical Journal 3 (5775): 632–633. doi:10.1136/bmj.3.5775.632. PMC 1798737. PMID 4998847.
- ↑ Amdur, Mary O.; Doull, John. Casarett and Doull's Toxicology. 4th edition Pub: Elsevier 1991 ISBN 0071052399
- ↑ John B. Sullivan, Jr.; Gary R. Krieger (2001). Clinical Environmental Health and Toxic Exposures: Latex. Lippincott Williams & Wilkins. pp. 264–. ISBN 978-0-683-08027-8. Retrieved 23 April 2013.
- ↑ Marsh N, Marsh A (2000). "A short history of nitroglycerine and nitric oxide in pharmacology and physiology". Clin. Exp. Pharmacol. Physiol. 27 (4): 313–9. doi:10.1046/j.1440-1681.2000.03240.x. PMID 10779131. Retrieved 2015-10-16.
- ↑ Assembly of Life Sciences (U.S.). Advisory Center on Toxicology. Toxicological Reports. National Academies. pp. 115–. NAP:11288. Retrieved 23 April 2013.
- ↑ "CDC - NIOSH Pocket Guide to Chemical Hazards - Nitroglycerine". www.cdc.gov. Retrieved 2015-11-21.
External links
- "Nitroglycerine! Terrible Explosion and Loss of Lives in San Francisco". Central Pacific Railroad Photographic History Museum. Retrieved 2005-03-23. – 1866 Newspaper article
- WebBook page for C3H5N3O9
- CDC - NIOSH Pocket Guide to Chemical Hazards
- The Tallini Tales of Destruction Detailed and horrific stories of the historical use of nitroglycerin-filled torpedoes to restart petroleum wells.
- Dynamite and TNT at The Periodic Table of Videos (University of Nottingham)
|

