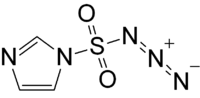
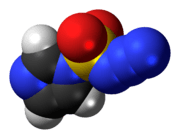
Imidazole-1-sulfonyl azide
 | |
 | |
| Identifiers | |
|---|---|
| 952234-37-6 | |
| ChemSpider | 21374920 |
| Jmol interactive 3D | Image |
| |
| |
| Properties | |
| C3H3N5O2S | |
| Molar mass | 173.15 g·mol−1 |
| Appearance | Colourless Liquid |
| Hazards | |
| Main hazards | Potentially explosive, Harmful |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Imidazole-1-sulfonyl azide is an organic azide compound that can be used as an alternative to trifluoromethanesulfonyl azide. It is a colorless liquid; it may also be used as a better-handled hydrogen sulfate salt.
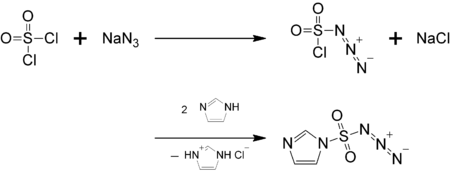
Preparation
This compound is also available commercially [1]
Reactions
Like trifluoromethanesulfonyl azide, this compound generally converts primary amines or ammonium salts to azides when catalyzed by copper(II), nickel(II), zinc(II), and cobalt(II) salts.[2] This reaction is effectively the reverse of the Staudinger reaction. Similarly, it is able to transfer the diazo group (=N2) under basic conditions.[2]
Safety
As with all organic azides, this compound is potentially explosive. The hydrochloride salt was initially reported to be insensitive to impact, vigorous grinding, and prolonged heating at 80 °C, although heating above 150 °C resulted in violent decomposition. Subsequent reports noted that upon storage, the hydrochloride salt hydrolyzed to produce hydrazoic acid, which made the material sensitive.[2][3] Note: We synthesized this and had 20 g explode when unattended in a vacuum oven when it was in a dry state, please be careful if you plan to use/make this chemical.
References
- ↑ van Delft, Floris. "SX-B1001". Synaffix. Retrieved 30 January 2014.
- 1 2 3 E. D. Goddard-Borger and R. V. Stick (2007). "An Efficient, Inexpensive, and Shelf-Stable Diazotransfer Reagent: Imidazole-1-sulfonyl Azide Hydrochloride". Organic Letters 9 (19): 3797–3800. doi:10.1021/ol701581g. PMID 17713918.
E. D. Goddard-Borger and R. V. Stick (2011). "An Efficient, Inexpensive, and Shelf-Stable Diazotransfer Reagent: Imidazole-1-sulfonyl Azide Hydrochloride". Organic Letters (Addition/Correction) 13 (9): 2514. doi:10.1021/ol2007555. - ↑ Fischer, Niko; Goddard-Borger, Ethan D.; Greiner, Robert; Klapötke, Thomas M.; Skelton, Brian W.; Stierstorfer, Jörg (2012). "Sensitivities of Some Imidazole-1-sulfonyl Azide Salts". J. Org. Chem. 77 (4): 1760. doi:10.1021/jo202264r.