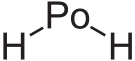Polonium hydride
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Polonium hydride | |||
| Systematic IUPAC name
Polane | |||
| Identifiers | |||
| 31060-73-8 | |||
| ChEBI | CHEBI:30444 | ||
| ChemSpider | 22383 | ||
| 25163, 169602 | |||
| PubChem | 23941 | ||
| Properties | |||
| PoH2 | |||
| Molar mass | 210.998 g/mol | ||
| Melting point | −35.3 °C (−31.5 °F; 237.8 K)[1] | ||
| Boiling point | 36.1 °C (97.0 °F; 309.2 K)[1] | ||
| Related compounds | |||
| Other anions |
H2O H2S H2Se H2Te | ||
| Other cations |
BiH3 HAt | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Polonium hydride (also known as polonium dihydride, hydrogen polonide, or polane) is a chemical compound with the formula PoH2. It is a liquid at room temperature. It is very unstable chemically and tends to decompose into elemental polonium and hydrogen; like all polonium compounds, it is highly radioactive. It is a volatile and very labile compound, from which many polonides can be derived.[2]
Preparation
Polonium hydride cannot be produced by direct reaction from the elements upon heating. Other unsuccessful routes to synthesis include the reaction of polonium tetrachloride (PoCl4) with lithium aluminium hydride (LiAlH4), which only produces elemental polonium, and the reaction of hydrochloric acid with magnesium polonide (MgPo). The fact that these synthesis routes do not work may be caused by the radiolysis of polonium hydride upon formation.[3]
Trace quantities of polonium hydride may be prepared by reacting hydrochloric acid with polonium-plated magnesium foil. In addition, the diffusion of trace quantities of polonium in palladium or platinum that is saturated with hydrogen (see palladium hydride) may be due to the formation and migration of polonium hydride.[3]
Experiments conducted on the trace scale show indications that the reaction between polonium metal and nascent hydrogen may produce polonium hydride.[3]
Properties
Polonium hydride is a more covalent compound than most metal hydrides because polonium straddles the border between metals and metalloids and has some nonmetallic properties. It is intermediate between a hydrogen halide like hydrogen chloride and a metal hydride like stannane.
It should have properties similar to that of hydrogen selenide and hydrogen telluride, other borderline hydrides. It is very unstable at room temperature and must be stored at freezer temperatures to prevent reversion to elemental polonium and hydrogen; this is because it is an endothermic compound, like the lighter hydrogen telluride and hydrogen selenide, and decomposes into its constituent elements, releasing heat in the process. The amount of heat given off in the decomposition of polonium hydride is over 100 kJ/mol, the largest of all the hydrogen chalcogenides.
Polonium hydride is a liquid due to van der Waals forces, and not due to the same reason as why water (hydrogen oxide) is liquid (hydrogen bonding).
It is predicted that, like the other hydrogen chalcogenides, polonium may form two types of salts: polonide (containing the Po2− anion) and one from polonium hydride (containing –PoH, which would be the polonium analogue of thiol, selenol and tellurol). However, no salts from polonium hydride are known. An example of a polonide is lead polonide (PbPo), which occurs naturally as lead is formed in the alpha decay of polonium.[4]
Polonium hydride is difficult to work with due to the extreme radioactivity of polonium and its compounds and has only been prepared in very dilute tracer quantities. As a result its physical properties are not definitely known.[3] It is also unknown if polonium hydride forms an acidic solution in water like its lighter homologues, or if it behaves more like a metal hydride (see also hydrogen astatide).
References
- 1 2 Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). Lehrbuch der Anorganischen Chemie (in German) (102 ed.). Walter de Gruyter. p. 627. ISBN 978-3-11-017770-1.
- ↑ Holleman, A. F.; Wiberg, E. (2001), Inorganic Chemistry, San Diego: Academic Press, p. 594, ISBN 0-12-352651-5
- 1 2 3 4 Bagnall, K. W. (1962). "The Chemistry of Polonium". Advances in Inorganic Chemistry and Radiochemistry. New York: Academic Press. pp. 197–230. ISBN 9780120236046. Retrieved June 7, 2012.
- ↑ Weigel, F. (1959). "Chemie des Poloniums". Angewandte Chemie 71: 289–316. doi:10.1002/ange.19590710902.
| ||||||||||||||||||