Human cytomegalovirus
| Cytomegalovirus | |
|---|---|
| Classification and external resources | |
| Specialty | Infectious disease |
| ICD-10 | B25 |
| ICD-9-CM | 078.5 |
| MeSH | D003586 |
| Cytomegalovirus | |
|---|---|
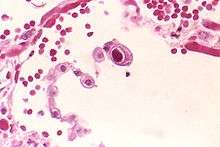 | |
| CMV infection of a lung pneumocyte. | |
| Virus classification | |
| Group: | Group I (dsDNA) |
| Order: | Herpesvirales |
| Family: | Herpesviridae |
| Subfamily: | Betaherpesvirinae |
| Genus: | Cytomegalovirus |
| Species: | Human cytomegalovirus |
| Synonyms | |
| |
Human cytomegalovirus is a species of the Cytomegalovirus genus of viruses, which in turn is a member of the viral family known as Herpesviridae or herpesviruses. It is typically abbreviated as HCMV or, commonly but more ambiguously, as CMV. It is also known as human herpesvirus-5 (HHV-5).[1] Within Herpesviridae, HCMV belongs to the Betaherpesvirinae subfamily, which also includes cytomegaloviruses from other mammals.[2]
Although they may be found throughout the body, HCMV infections are frequently associated with the salivary glands.[2] HCMV infection is typically unnoticed in healthy people, but can be life-threatening for the immunocompromised, such as HIV-infected persons, organ transplant recipients, or newborn infants.[1] Neonatal infection with CMV can lead to significant morbidity and even death. After infection, HCMV remains latent within the body throughout life and can be reactivated at any time. Eventually, it may cause mucoepidermoid carcinoma and possibly other malignancies [3] such as prostate cancer.[4]
HCMV is found throughout all geographic locations and socioeconomic groups, and infects between 60% and 70% of adults in industrialized countries and almost 100% in emerging countries.[5] Of all herpes viruses, HCMV harbors the most genes dedicated to altering (evading) innate and adaptive immunity in the host and represents a life-long burden of antigenic T cell surveillance and immune dysfunction.[6] Commonly it is indicated by the presence of antibodies in the general population.[1] Seroprevalence is age-dependent: 58.9% of individuals aged 6 and older are infected with CMV while 90.8% of individuals aged 80 and older are positive for HCMV.[7] HCMV is also the virus most frequently transmitted to a developing fetus. HCMV infection is more widespread in developing countries and in communities with lower socioeconomic status and represents the most significant viral cause of birth defects in industrialized countries. Congenital HCMV is the leading infectious cause of deafness, learning disabilities, and intellectual disability in children[8] CMV also "seems to have a large impact on immune parameters in later life and may contribute to increased morbidity and eventual mortality."[9]
Signs and symptoms
CMV infections are most significant in the perinatal period and in immunocompromised patients.
Pregnancy and congenital infection
HCMV is one of the vertically transmitted infections that lead to congenital abnormalities. (Others are: Toxoplasmosis, Rubella, and Herpes simplex). Congenital HCMV infection occurs when the mother suffers a primary infection or reactivation during pregnancy.
Up to 5/1000 live births are infected. 5% develop multiple handicaps, and develop cytomegalic inclusion disease with nonspecific signs that resemble rubella. Another 5% later develop cerebral calcification (decreasing IQ levels dramatically and causing sensorineural deafness and psychomotor retardation).
Immunocompromised adults
Primary CMV infection in patients with weakened immune systems can lead to serious disease. However, a more common problem is reactivation of the latent virus.
Infection with CMV is a major cause of disease and death in immunocompromised patients, including organ transplant recipients, patients undergoing hemodialysis, patients with cancer, patients receiving immunosuppressive drugs, and HIV-infected patients. Exposing immunosuppressed patients to outside sources of CMV should be minimized to avoid the risk of serious infection. Susceptible patients without CMV infection can be infected by receiving infected organs and blood products unless care is taken.
In patients with a depressed immune system, CMV-related disease may be much more aggressive.
Specific disease entities recognised in those people are
- CMV hepatitis, which may cause fulminant liver failure
- cytomegalovirus retinitis (inflammation of the retina, characterised by a "pizza pie appearance" on ophthalmoscopy)
- cytomegalovirus colitis (inflammation of the large bowel)
- CMV pneumonitis
- CMV esophagitis[10]
- polyradiculopathy, transverse myelitis, and subacute encephalitis
Patients without CMV infection who are given organ transplants from CMV-infected donors require prophylactic treatment with valganciclovir (ideally) or ganciclovir, and regular serological monitoring to detect a rising CMV titre; if treated early establishment of a potentially life-threatening infection can be prevented.
Immunocompetent adults
CMV infections can still be of clinical significance in adult immunocompetent populations. However infection is typically asymptomatic or persists as a mononucleosis syndrome.[11]
- CMV mononucleosis (some sources reserve "mononucleosis" for EBV only). However, the mononucleosis syndrome associated with CMV typically lacks signs of enlarged cervical lymph nodes and splenomegaly.[12]
- Post-transfusion CMV—similar to CMV mononucleosis
- A 2009 study suggests that CMV infection may be linked to the development of arterial hypertension.[13] Mice fed a high cholesterol diet showed significantly more vascular damage and hypertension when they had been infected with CMV. CMV infection stimulated cytokines – IL6, TNF, and MCP1 – in the infected mice, indicating that the infection led to an inflammatory response in vessels and other tissues. Further, renin and angiotensin II release were increased in these animals as additional factors to lead to hypertension. In humans CMV infection has been dem onstrated in the aortic smooth muscle cells from patients with abdominal aortic aneurysms suggesting that CMV infection contributes to vascular disease.[14][15]
- CMV has been also been associated with Guillain–Barré syndrome, type 1 diabetes,[16] and type 2 diabetes.[17]
Pathogenesis
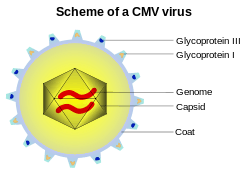
Most healthy people who are infected by HCMV after birth have no symptoms.[1] Some develop a syndrome similar to infectious mononucleosis or glandular fever,[18] with prolonged fever, and a mild hepatitis. A sore throat is common. After infection, the virus remains latent in lymphocytes in the body for the rest of the person's life. Overt disease rarely occurs unless immunity is suppressed either by drugs, infection or old age. Initial HCMV infection, which often is asymptomatic, is followed by a prolonged, inapparent infection during which the virus resides in mononuclear cells without causing detectable damage or clinical illness.
Infectious CMV may be shed in the bodily fluids of any infected person, and can be found in urine, saliva, blood, tears, semen, and breast milk. The shedding of virus can occur intermittently, without any detectable signs or symptoms.

CMV infection can be demonstrated microscopically by the detection of intranuclear inclusion bodies. On H&E staining, the inclusion bodies stain dark pink and are called "owl's eye" inclusion bodies.[19]
HCMV infection is important to certain high-risk groups.[20] Major areas of risk of infection include pre-natal or postnatal infants and immunocompromised individuals, such as organ transplant recipients, persons with leukemia, or those infected with human immunodeficiency virus (HIV). In HIV infected persons, HCMV is considered an AIDS-defining infection, indicating that the T-cell count has dropped to low levels.
Lytically replicating viruses disrupt the cytoskeleton, causing massive cell enlargement, which is the source of the virus' name.
A study published in 2009 links infection with CMV to high blood pressure in mice, and suggests that the result of CMV infection of blood vessel endothelial cells (EC) in humans is a major cause of atherosclerosis.[13] Researchers also found that when the cells were infected with CMV, they created renin, a protein known to contribute to high blood pressure.
CMV encodes a protein, UL16, which is involved in the immune evasion of NK cell responses. It binds to ligands ULBP1, ULBP2 and MICB of NK cell activating receptor NKG2D, which prevents their surface expression. These ligands are normally upregulated in times of cellular stress, such as in viral infection, and by preventing their upregulation, CMV can prevent its host cell from dying due to NK cells[21]
Transmission
The mode of HCMV transmission from person to person is entirely unknown but is presumed to occur through bodily fluids.[2] Infection requires close, intimate contact with a person secreting the virus in their saliva, urine, or other bodily fluids. CMV can be transmitted sexually and via breast milk, and also occurs through receiving transplanted organs or blood transfusions.[22] Although HCMV is not highly contagious, it has been shown to spread in households and among young children in day care centers.[1]
Replication
HCMV replicates within infected endothelial cells[23] at a slow rate, taking about 5 days in cell culture.[24] Like other herpesviruses, HCMV expresses genes in a temporally controlled manner.[25][26] Immediate early genes (0–4 hours after infection) are involved in the regulation of transcription, followed by early genes (4–48 hours after infection) which are involved in viral DNA replication and further transcriptional regulation.[25] Late genes are expressed during the remainder of infection up to viral egress and typically code for structural proteins. While HCMV encodes for its own functional DNA polymerase, the virus makes use of the host RNA polymerase for the transcription of all of its genes.[27]
Synthesis of the viral double-stranded DNA genome occurs at the host cell nucleus within specialized viral replication compartments.[28]
Interestingly, nearly 75% of the genes encoded by HCMV strain AD169 can be deleted and still result in the production of infectious virus.[29] This suggests that the virus focuses on avoiding the host immune system for a timely entrance into latency.
Prevention
A phase 2 study of a CMV-vaccine published in 2009 indicated an efficacy of 50%—the protection provided was limited, and a number of subjects contracted CMV infection despite vaccination. In one case also congenital CMV was encountered.[30]
In 2013, Astellas Pharma has started on individuals who received a hematopoietic stem cell transplant a phase 3 trial with its CMV deoxyribonucleic acid DNA cytomegalovirus vaccine ASP0113.[31]
In 2015, Astellas Pharma has commenced on healthy volunteers a phase 1 trial with its cytomegalovirus vaccine ASP0113.[32]
Further cytomegalovirus vaccines candidates are the CMV-MVA Triplex vaccine and the CMVpp65-A*0201 peptide vaccine. Both vaccine candidates are sponsored by the City of Hope National Medical Center. As of 2016, the development is in clinical phase 2 trial stage.[33][34]
Wash hands often with soap and water for 15–20 seconds, especially after: changing diapers, feeding a young child, wiping young child's nose or mouth, and or handling children's toys. Avoid contact with saliva when kissing a child.
Diagnosis


Most infections with CMV are not diagnosed because the virus usually produces few, if any, symptoms and tends to reactivate intermittently without symptoms. However, persons who have been infected with CMV develop antibodies to the virus, and these antibodies persist in the body for the lifetime of that individual. A number of laboratory tests that detect these antibodies to CMV have been developed to determine if infection has occurred and are widely available from commercial laboratories. In addition, the virus can be cultured from specimens obtained from urine, throat swabs, bronchial lavages and tissue samples to detect active infection. Both qualitative and quantitative polymerase chain reaction (PCR) testing for CMV are available as well, allowing physicians to monitor the viral load of CMV-infected patients.
CMV pp65 antigenemia test is an immunofluorescence based assay which utilizes an indirect immunofluorescence technique for identifying the pp65 protein of cytomegalovirus in peripheral blood leukocytes. The CMV pp65 assay is widely used for monitoring CMV infections and its response to antiviral treatment in patients who are under immunosuppressive therapy and have had renal transplantation surgery as the antigenemia results are obtained about 5 days before the onset of symptomatic CMV disease. The advantage of this assay is the rapidity in providing results in a few hours and that the pp65 antigen determination represents a useful parameter for the physician to initiate antiviral therapy. The major disadvantage of the pp65 assay is that only limited number of samples can be processed per test batch.
CMV should be suspected if a patient has symptoms of infectious mononucleosis but has negative test results for mononucleosis and Epstein-Barr virus, or if they show signs of hepatitis, but has negative test results for hepatitis A, B, and C.
For best diagnostic results, laboratory tests for CMV antibody should be performed by using paired serum samples. One blood sample should be taken upon suspicion of CMV, and another one taken within 2 weeks. A virus culture can be performed at any time the patient is symptomatic. Laboratory testing for antibody to CMV can be performed to determine if a woman has already had CMV infection. However, routine testing of all pregnant women is costly and the need for testing should therefore be evaluated on a case-by-case basis.
Serologic testing
The enzyme-linked immunosorbent assay (or ELISA) is the most commonly available serologic test for measuring antibody to CMV. The result can be used to determine if acute infection, prior infection, or passively acquired maternal antibody in an infant is present. Other tests include various fluorescence assays, indirect hemagglutination, (PCR) and latex agglutination.
An ELISA technique for CMV-specific IgM is available, but may give false-positive results unless steps are taken to remove rheumatoid factor or most of the IgG antibody before the serum sample is tested. Because CMV-specific IgM may be produced in low levels in reactivated CMV infection, its presence is not always indicative of primary infection. Only virus recovered from a target organ, such as the lung, provides unequivocal evidence that the current illness is caused by acquired CMV infection. If serologic tests detect a positive or high titer of IgG, this result should not automatically be interpreted to mean that active CMV infection is present. However, if antibody tests of paired serum samples show a fourfold rise in IgG antibody and a significant level of IgM antibody, meaning equal to at least 30% of the IgG value, or virus is cultured from a urine or throat specimen, the findings indicate that an active CMV infection is present.
Relevance to blood donors
Although the risks discussed above are generally low, CMV assays are part of the standard screening for non-directed blood donation (donations not specified for a particular patient) in the U.S., the UK and many other countries. CMV-negative donations are then earmarked for transfusion to infants or immunocompromised patients. Some blood donation centers maintain lists of donors whose blood is CMV negative due to special demands.[35]
Treatment
Cytomegalovirus Immune Globulin Intravenous (human) (CMV-IGIV) is an immunoglobulin G (IgG) containing a standardized number of antibodies to cytomegalovirus. It may be used for the prophylaxis of cytomegalovirus disease associated with transplantation of kidney, lung, liver, pancreas, and heart. Alone or in combination with an antiviral agent, it has been shown to:
- Reduce the risk of CMV-related disease and death in some of the highest-risk transplant patients
- Provide a measurable long-term survival benefit
- Produce minimal treatment-related side effects and adverse events.[36]
Ganciclovir (Cytovene) treatment is used for patients with depressed immunity who have either sight-related or life-threatening illnesses. Valganciclovir (Valcyte) is an antiviral drug that is also effective and is given orally. The therapeutic effectiveness is frequently compromised by the emergence of drug-resistant virus isolates. A variety of amino acid changes in the UL97 protein kinase and the viral DNA polymerase have been reported to cause drug resistance. Foscarnet or cidofovir are only given to patients with CMV resistant to ganciclovir, because foscarnet has notable nephrotoxicity, resulting in increased or decreased Ca2+ or P, and decreased Mg2+.
Drug resistance
All three currently licensed anti-HCMV drugs target the viral DNA polymerase, pUL54. Ganciclovir (GCV) acts as nucleoside analogue. Its antiviral activity requires phosphorylation by the HCMV protein kinase, pUL97.[37] The second drug, Cidofovir (CDV), is a nucleotide analogue, which is already phosphorylated and thus active. Finally, Foscarnet (FOS) has a different mode of action. It directly inhibits polymerase function by blocking the pyrophosphate binding site of pUL54 (note: investigational drug letermovir acts through a mechanism that involves viral terminase).[38] Two HCMV proteins are implicated in antiviral resistance against these three drugs: pUL97 and pUL54. Specific mutations in pUL97 can cause reduced phosphorylation activity of this viral protein kinase. Thus, fewer monophosphorylated – and thus active – GCV can be synthesized,[39] leading to antiviral resistance against GCV. About 90% of all GCV resistances are caused by such mutations in UL97.[40] Mutations in pUL54 may have different effects leading to antiviral drug resistance: A. They can lead to decreased affinity to antiviral compounds. This resistance mechanism concerns GCV, CDV and FOS and may lead to multidrug resistance.[41] B. Some mutations in pUL54 can increase the polymerase’s exonuclease activity. This causes enhanced recognition of incorporated GCV and CDV. As a result, these dNTP analogues are excised more efficiently. Major risk factors for HCMV drug resistance are the residual capacity of the host’s immune system to control viral replication and the overall amount and duration of viral replication.[42] HCMV antiviral drug resistance can be detected by phenotypic or by genotypic drug resistance testing. Phenotypic resistance testing involves cultivation of the virus in cell culture and testing its susceptibility using different antiviral drug concentrations in order to determine EC50 values. In contrast, genotypic resistance testing means the detection of resistance associated mutations in UL97 and UL54 by sequencing. Genotypic resistance testing is becoming the method of choice because it is faster, but requires previous phenotypic characterisation of each newly found mutation. This can be performed via a web-based search tool that links a patient’s HCMV sequence to a database containing all published UL97 and UL54 mutations and corresponding antiviral drug susceptibility phenotypes.[43]
References
- 1 2 3 4 5 Ryan KJ, Ray CG (editors) (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 556; 566–9. ISBN 0-8385-8529-9.
- 1 2 3 Koichi Yamanishi; Arvin, Ann M.; Gabriella Campadelli-Fiume; Edward Mocarski; Moore, Patrick; Roizman, Bernard; Whitley, Richard (2007). Human herpesviruses: biology, therapy, and immunoprophylaxis. Cambridge, UK: Cambridge University Press. ISBN 0-521-82714-0.
- ↑ Melnick M, Sedghizadeh PP, Allen CM, Jaskoll T; Sedghizadeh; Allen; Jaskoll (10 November 2011). "Melnick M., Sedghizadeh P. S., Allen C. M., Jaskoll T". Experimental and Molecular Pathology 92 (1): 118–25. doi:10.1016/j.yexmp.2011.10.011. PMID 22101257.
- ↑ http://europepmc.org/abstract/med/68820
- ↑ T. Fülöp, A. Larbi and G. Pawelec (2013) (September 2013). "Human T cell aging and the impact of persistent viral infections". Frontiers in Immunology 4: 1–9. doi:10.3389/fimmu.2013.00271.
|article=ignored (help) - ↑ S. Varani and M. P. Landini (2011) (2011). "Cytomegalovirus-induced immunopathology and its clinical consequences". Herpesviridae 2 (6): 6. doi:10.1186/2042-4280-2-6.
- ↑ Staras SA, Dollard SC, Radford KW, Flanders WD, Pass RF, Cannon MJ; Dollard; Radford; Flanders; Pass; Cannon (November 2006). "Seroprevalence of cytomegalovirus infection in the United States, 1988–1994". Clin. Infect. Dis. 43 (9): 1143–51. doi:10.1086/508173. PMID 17029132.
- ↑ Elizabeth G. Damato and Caitlin W. Winnen (2006). "Cytomegalovirus infection: perinatal implications". J Obstet Gynecol Neonatal Nurs 31 (1): 86–92. doi:10.1111/j.1552-6909.2002.tb00026.x. PMID 11843023.
- ↑ Caruso C, Buffa S, Candore G; et al. (2009). "Mechanisms of immunosenescence". Immun Ageing 6: 10. doi:10.1186/1742-4933-6-10. PMC 2723084. PMID 19624841.
- ↑ Meinhard Classen; Guido N. J. Tytgat; M.D. Ph.D.; Charles J. Lightdale (2010). Gastroenterological Endoscopy. Thieme. pp. 490–. ISBN 978-3-13-125852-6. Retrieved 26 June 2010.
- ↑ Cohen JI, Corey GR; Corey (March 1985). "Cytomegalovirus infection in the normal host". Medicine (Baltimore) 64 (2): 100–14. doi:10.1097/00005792-198503000-00003. PMID 2983175.
- ↑ Klemola E, Von Essen R, Henle G, Henle W; von Essen; Henle; Henle (June 1970). "Infectious-mononucleosis-like disease with negative heterophil agglutination test. Clinical features in relation to Epstein-Barr virus and cytomegalovirus antibodies". J. Infect. Dis. 121 (6): 608–14. doi:10.1093/infdis/121.6.608. PMID 4316146.
- 1 2 Cheng J, Ke Q, Jin Z; et al. (May 2009). Früh, Klaus, ed. "Cytomegalovirus infection causes an increase of arterial blood pressure". PLoS Pathog. 5 (5): e1000427. doi:10.1371/journal.ppat.1000427. PMC 2673691. PMID 19436702.
- ↑ Gredmark-Russ S, Dzabic M, Rahbar A, Wanhainen A, Björck M, Larsson E, Michel JB, Söderberg-Nauclér C.; Dzabic; Rahbar; Wanhainen; Björck; Larsson; Michel; Söderberg-Nauclér (2009). "Active cytomegalovirus infection in aortic smooth muscle cells from patients with abdominal aortic aneurysm". J Mol Med. 87 (4): 347–56. doi:10.1007/s00109-008-0413-4. PMID 19083194.
- ↑ Yonemitsu Y, Nakagawa K, Tanaka S, Mori R, Sugimachi K, Sueishi K.; Nakagawa; Tanaka; Mori; Sugimachi; Sueishi (1996). "In situ detection of frequent and active infection of human cytomegalovirus in inflammatory abdominal aortic aneurysms: possible pathogenic role in sustained chronic inflammatory reaction". Lab Invest. 74 (4): 723–36. PMID 8606483.
- ↑ "ASSOCIATION OF CYTOMEGALOVIRUS INFECTION WITH AUTOIMMUNE TYPE 1 DIABETES". The Lancet 332: 1–4. doi:10.1016/S0140-6736(88)92941-8.
- ↑ "Detection of cytomegalovirus nucleic acid sequences in pancreas in type 2 diabetes". The Lancet 336: 644–648. doi:10.1016/0140-6736(90)92145-8.
- ↑ Bottieau E, Clerinx J, Van den Enden E; et al. (2006). "Infectious mononucleosis-like syndromes in febrile travelers returning from the tropics". J Travel Med 13 (4): 191–7. doi:10.1111/j.1708-8305.2006.00049.x. PMID 16884400.
- ↑ Mattes FM, McLaughlin JE, Emery VC, Clark DA, Griffiths PD; McLaughlin; Emery; Clark; Griffiths (August 2000). "Histopathological detection of owl's eye inclusions is still specific for cytomegalovirus in the era of human herpesviruses 6 and 7". J. Clin. Pathol. 53 (8): 612–4. doi:10.1136/jcp.53.8.612. PMC 1762915. PMID 11002765.
- ↑ Bennekov T, Spector D, Langhoff E; Spector; Langhoff (March 2004). "Induction of immunity against human cytomegalovirus". Mt. Sinai J. Med. 71 (2): 86–93. PMID 15029400.
- ↑ Welte, Stefan A.; Sinzger, Christian; Lutz, Stefan Z.; Singh-Jasuja, Harpreet; Sampaio, Kerstin Laib; Eknigk, Ute; Rammensee, Hans-Georg; Steinle, Alexander (2003). "Selective intracellular retention of virally induced NKG2D ligands by the human cytomegalovirus UL16 glycoprotein". European Journal of Immunology 33 (1): 194–203. doi:10.1002/immu.200390022. PMID 12594848.
- ↑ Taylor GH (February 2003). "Cytomegalovirus". Am Fam Physician 67 (3): 519–24. PMID 12588074.
- ↑ Kahl, M.; Siegel-Axel, D.; Stenglein, S. (2000-08-01). "Efficient Lytic Infection of Human Arterial Endothelial Cells by Human Cytomegalovirus Strains". Journal of Virology 74 (16): 7628–7635. ISSN 0022-538X. PMC 112284. PMID 10906217.
- ↑ Emery, Vincent C.; Cope, Alethea V.; Bowen, E. Frances; Gor, Dehila; Griffiths, Paul D. (1999-07-19). "The Dynamics of Human Cytomegalovirus Replication in Vivo". The Journal of Experimental Medicine 190 (2): 177–182. doi:10.1084/jem.190.2.177. ISSN 0022-1007. PMID 10432281.
- 1 2 Wathen, M. W.; Stinski, M. F. (1982-02-01). "Temporal patterns of human cytomegalovirus transcription: mapping the viral RNAs synthesized at immediate early, early, and late times after infection". Journal of Virology 41 (2): 462–477. ISSN 0022-538X. PMC 256775. PMID 6281461.
- ↑ Stern-Ginossar, Noam; Weisburd, Ben; Michalski, Annette; Le, Vu Thuy Khanh; Hein, Marco Y.; Huang, Sheng-Xiong; Ma, Ming; Shen, Ben; Qian, Shu-Bing (2012-11-23). "Decoding Human Cytomegalovirus". Science 338 (6110): 1088–1093. doi:10.1126/science.1227919. ISSN 0036-8075. PMC 3817102. PMID 23180859.
- ↑ Snaar, S. P.; Vincent, M.; Dirks, R. W. (1999-02-01). "RNA polymerase II localizes at sites of human cytomegalovirus immediate-early RNA synthesis and processing". The Journal of Histochemistry and Cytochemistry: Official Journal of the Histochemistry Society 47 (2): 245–254. ISSN 0022-1554. PMID 9889260.
- ↑ Penfold, M. E.; Mocarski, E. S. (1997-12-08). "Formation of cytomegalovirus DNA replication compartments defined by localization of viral proteins and DNA synthesis". Virology 239 (1): 46–61. doi:10.1006/viro.1997.8848. ISSN 0042-6822. PMID 9426445.
- ↑ Dunn, Walter; Chou, Cassie; Li, Hong; Hai, Rong; Patterson, David; Stolc, Viktor; Zhu, Hua; Liu, Fenyong (2003-11-25). "Functional profiling of a human cytomegalovirus genome". Proceedings of the National Academy of Sciences 100 (24): 14223–14228. doi:10.1073/pnas.2334032100. ISSN 0027-8424. PMC 283573. PMID 14623981.
- ↑ Pass RF, Zhang C, Evans A; et al. (March 2009). "Vaccine prevention of maternal cytomegalovirus infection". N. Engl. J. Med. 360 (12): 1191–9. doi:10.1056/NEJMoa0804749. PMC 2753425. PMID 19297572.
- ↑ "A Study to Evaluate a Therapeutic Vaccine, ASP0113, in Cytomegalovirus (CMV)-Seropositive Recipients Undergoing Allogeneic, Hematopoietic Cell Transplant (HCT) (HELIOS)". ClinicalTrials.gov. 2013-06-12. Retrieved 2015-10-26.
- ↑ "An Evaluation of a Cytomegalovirus (CMV) Vaccine (ASP0113) in CMV-Seropositive and CMV-Seronegative Healthy Subjects and CMV-Seronegative Dialysis Patients". ClinicalTrials.gov. 2015-07-08. Retrieved 2015-10-22.
- ↑ "Multi-antigen CMV-MVA Triplex Vaccine in Reducing CMV Complications in Patients Previously Infected With CMV and Undergoing Donor Hematopoietic Cell Transplant". ClinicalTrials.gov. 2015-07-21. Retrieved 2016-01-23.
- ↑ "Vaccine Therapy in Reducing the Frequency of Cytomegalovirus Events in Patients With Hematologic Malignancies Undergoing Donor Stem Cell Transplant". ClinicalTrials.gov. 2015-03-12. Retrieved 2016-01-23.
- ↑ "United Blood Services FAQs". Archived from the original on 2007-05-19. Retrieved 23 May 2007.
- ↑ Cytogam Prescribing Info CSL Behring AG
- ↑ Sullivan V, Talarico CL, Stanat SCC, Davis M, Coen DM, Biron KK; Talarico; Stanat; Davis; Coen; Biron (1992). "A protein kinase homologue controls phosphorylation of ganciclovir in human cytomegalovirus-infected cells". Nature 358 (6382): 162–164. doi:10.1038/358162a0. PMID 1319560.
- ↑ Chrisp P and Clissold SP (1991). "Foscarnet. A review of its antiviral activity, pharmacokinetic properties and therapeutic use in immunocompromised patients with cytomegalovirus retinitis". Drugs 41 (1): 104–129. doi:10.2165/00003495-199141010-00009. PMID 1706982.
- ↑ Biron KK, Fyfe JA, Stanat SC, Leslie LK, Sorrell JB, Lambe CU, Coen DM; Fyfe; Stanat; Leslie; Sorrell; Lambe; Coen (1986). "A human cytomegalovirus mutant resistant to the nucleoside analog 9-([2-hydroxy-1-(hydroxymethyl)ethoxy]methyl)guanine (BW B759U) induces reduced levels of BW B759U triphosphate". Proc. Natl. Acad. Sci. U.S.A. 83 (22): 8769–8773. doi:10.1073/pnas.83.22.8769. PMC 387013. PMID 3022304.
- ↑ Chou S (1999). "Antiviral drug resistance in human cytomegalovirus". Transpl. Infect. Dis. 1 (2): 105–114. doi:10.1034/j.1399-3062.1999.010204.x. PMID 11428978.
- ↑ Gilbert and Boivin (2005). "Human cytomegalovirus resistance to antiviral drugs". Antimicrob. Agents Chemother. 49 (3): 873–883. doi:10.1128/AAC.49.3.873-883.2005. PMC 549271. PMID 15728878.
|first2=missing|last2=in Authors list (help) - ↑ Drew WL (2000). "Ganciclovir resistance: a matter of time and titre". Lancet 356 (9230): 609–610. doi:10.1016/S0140-6736(00)02597-6. PMID 10968428.
- ↑ Chevillotte M, von Einem J, Meier BM, Lin FM, Kestler HA, Mertens T; von Einem; Meier; Lin; Kestler; Mertens (2010). "A new tool linking human cytomegalovirus drug resistance mutations to resistance phenotypes". Antiviral Research 85 (2): 318–27. doi:10.1016/j.antiviral.2009.10.004. PMID 19853628.
External links
- Human cytomegalovirus at DMOZ
- Cytomegalovirus (CMV) US Centers for Disease Control and Prevention (CDC). May 22, 2009
- Cytomegalovirus (CMV) Clinical DNA Information Resource (CDIR)
- National Institute of Standards and Technology (NIST)
- HCMV drug resistance mutations tool
- Sexually Transmitted Diseases (STDs): Cytomegalovirus (CMV) at Planned Parenthood
- "Human herpesvirus 5". NCBI Taxonomy Browser. 10359.
| ||||||||||||||||||