Amlodipine/valsartan
 | |
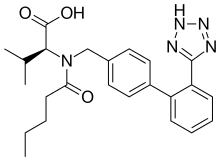 | |
| Combination of | |
|---|---|
| Amlodipine | Calcium channel blocker |
| Valsartan | Angiotensin II receptor antagonist |
| Clinical data | |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration | Oral |
| Identifiers | |
| ATC code | C09DB01 |
| PubChem | CID 11354874 |
| ChemSpider |
13092186 |
| KEGG |
D09745 |
| (verify) | |
Exforge is the brand name of an oral blood pressure lowering medication which combines two medications in a film-coated tablet. It contains amlodipine, a dihydropyridine-type calcium channel blocker, and valsartan, an angiotensin II receptor antagonist (ARB or A2RA). The patent is held by Novartis Pharmaceuticals. Novartis markets valsartan as Diovan. The patent expired in September 2012.[1] Pfizer's patent on amlodipine expired in 2007. Until then, it was marketed as Norvasc.
Exforge is available in different dose preparations: 5 mg/80 mg, 5 mg/160 mg, 10 mg/160 mg, 5 mg/320 mg and 10/320 mg of amlodipine and valsartan, respectively.
Exforge is also available with hydrochlorothiazide under the brand name Exforge HCT, for patients who need a three drug regimen to manage their blood pressure.
Cipla has just launched Amlopres VL in this combination.
References
- ↑ "Generic Exforge HCT". Blood-pressure.emedtv.com. Retrieved 2012-03-05.
External links
| ||||||||||||||||||