Ehrlich's reagent
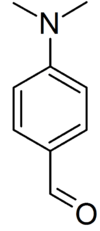
Ehrlich's reagent, also known as the "DMAB test", is a chemical test to presumptively identify indoles. It is primarily used as a simple spot test to identify possible psychoactive compounds such as tryptamines (e.g. DMT) and ergoloids (e.g. LSD). The reagent will also give a positive result for opium, despite the opiates not containing the indole functional group, because of the presence tryptophan in natural opium.[1] It is named after Nobel Prize winner Paul Ehrlich who used it to distinguish typhoid from simple diarrhoea.
The reagent is prepared by dissolving 0.5[2]–2.0 g of p–dimethylaminobenzaldehyde (DMAB) in 50 mL of 95% ethanol and 50 mL of concentrated hydrochloric acid[3][4] and is best used when fresh. Other alcohols, such as 1-propanol, can also be used as well.[5]
The Ehrlich reagent is similar to a number of other indole tests:
- The van Urk reagent, which uses DMAB, sulfuric acid and an oxidant.[6]
- The Renz and Loew reagent, which uses p-dimethylaminocinnamaldehyde and may also be used for the detection of flavonoids.
- The "improved hallucinogen reagent", which uses 5 g DMAB in 100 mL concentrated phosphoric acid (specific gravity 1.75) and 100 mL of methanol.[1][7]
The Ehrlich reagent works by binding to the C2 position of two indole moieties to form a resonance stabilised carbenium ion compound.[8]
See also
- Pill testing
- Other alkaloid spot tests:
References
- 1 2 de Faubert Maunder, MJ (1975). "Field and laboratory test for raw and prepared opium.". Bulletin on narcotics 27 (1): 71–6. PMID 1039285.
- ↑ Spratley, Trinette (2004). "Analytical Profiles for Five "Designer" Tryptamines" (PDF). Microgram Journal 3 (1-2): 55. Retrieved 2013-10-09.
- ↑ O’Neal, Carol L; Crouch, Dennis J; Fatah, Alim A (April 2000). "Validation of twelve chemical spot tests for the detection of drugs of abuse". Forensic Science International 109 (3): 189–201. doi:10.1016/S0379-0738(99)00235-2. PMID 10725655.
- ↑ "Color Test Reagents/Kits for Preliminary Identification of Drugs of Abuse" (PDF). Law Enforcement and Corrections Standards and Testing Program. July 2000. Retrieved 2011-07-24.
- ↑ 02 July 2014. "Ehrlich's Reagent Safety Data Sheet" (PDF). Labchem. Retrieved 11 January 2015.
- ↑ Ehmann, A. (1977). "The van URK-Salkowski reagent — a sensitive and specific chromogenic reagent for silica gel thin-layer chromatographic detection and identification of indole derivatives" (PDF). Journal of Chromatography A 132 (2): 267–201. doi:10.1016/S0021-9673(00)89300-0.
- ↑ Maunder, M. J. de Faubert (August 1974). "A field test for hallucinogens: further improvements". Journal of Pharmacy and Pharmacology 26 (8): 637–638. doi:10.1111/j.2042-7158.1974.tb10677.x.
- ↑ Kovar, Karl-Artur and Laudszun, Martina (February 1989). "Chemistry and Reaction Mechanisms of Rapid Tests for Drugs of Abuse and Precursors Chemicals" (PDF). UNODC. p. 15. Retrieved 3 January 2016.