Deguelin
 | |
| Names | |
|---|---|
| IUPAC name
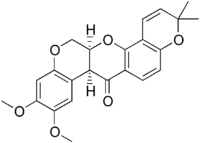
(7aS,13aS)-13,13a-Dihydro-9,10-dimethoxy-3,3-dimethyl-3H-bis[1]benzopyrano [3,4-b:6’,5’-3]pyran-7(7aH)-one | |
| Other names
Deguelin; 3H-Bis(1)benzopyrano (3,4-b:6',5'-e)pyran-7(7aH)-one, 13,13a-dihydro-9,10-dimethoxy-3,3- dimethyl-, (7aS,13aS)-; 3H-Bis(1) benzopyrano(3,4-b:6',5'-e)pyran- 7(7aH)-one, 13,13a-dihydro- 9, 10-dimethoxy-3,3-dimethyl-, (7aS-cis)- | |
| Identifiers | |
| 522-17-8 | |
| ChEMBL | ChEMBL393417 |
| ChemSpider | 97058 |
| Jmol interactive 3D | Image |
| PubChem | 107935 |
| |
| |
| Properties | |
| C23H22O6 | |
| Molar mass | 394.42 g/mol |
| Melting point | 171 °C (340 °F; 444 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Deguelin is a derivative of rotenone. Both are compounds classified as rotenoids of the flavonoid family and are naturally occurring insecticides. They can be produced by extraction from several plant species belonging to three genera of the Fabaceae or Legume family, Lonchocarpus, Derris, or Tephrosia.
Cubé resin, the root extract from Cubé (Lonchocarpus utilis) and from Barbasco (Lonchocarpus urucu), is used as a commercial insecticide and piscicide (fish poison). The major active ingredients are rotenone and deguelin. Although "organic" (produced by nature) cubé resin is no longer considered environmentally safe.
Pharmacokinetics
- Mean residence time (MRT) = 6.98 h[1]
- Terminal half-life (t1/2(gamma)) = 9.26 h
- Area under the curve (AUC) = 57.3 ng h/ml
- Total clearance (Cl) = 4.37 l/h per kg
- Apparent volume of distribution (V) = 3.421 l/kg
- Volume of distribution at steady-state (Vss) = 30.46 l/kg
- Tissue distributions after i.v. (intravenous) administration: heart > fat > mammary gland > colon > liver > kidney > brain > lung.
- Tissue distributions after i.g. (intragastric) administration: perirenal fat > heart > mammary gland > colon > kidney > liver > lung > brain > skin.
- Elimination: Within 5 days of i.g. administration, about 58.1% of the [3H]deguelin was eliminated via the feces and 14.4% via the urine. Approximately 1.7% of unchanged deguelin was found in the feces, and 0.4% in the urine.
Deguelin and anti-cancer activity
Deguelin displays anti-cancer activity by inhibiting the growth of pre-cancerous and cancerous cells[2] - particularly for lung cancer.[3] So far the compound has shown no toxic effects on normal cells. However, high doses of deguelin are suspected of having negative effects on the heart, lungs and nerves. The molecular mechanisms include the induction of apoptosis, mediatated through AKT/PKB signaling pathways in malignant and premaligant human bronchial epithelia (HBE) cells, with only minimal effects on normal HBE cells. Deguelin inhibits AKT by both Phosphoinositol-3-phosphate kinase (PI3K)-dependent and PI3K-independent pathways.
Deguelin and Parkinson's disease
Research has shown a correlation between intravenous deguelin and Parkinson's disease in rats.[4] The study does not suggest that deguelin exposure is responsible for Parkinson's disease in humans, but is consistent with the belief that chronic exposure to environmental toxins can increase the likelihood of the disease.
References
- ↑ Udeani GO, Zhao GM, Shin YG; et al. (March 2001). "Pharmacokinetics of deguelin, a cancer chemopreventive agent in rats". Cancer Chemother. Pharmacol. 47 (3): 263–8. doi:10.1007/s002800000187. PMID 11320671.
- ↑ Lee HY (September 2004). "Molecular mechanisms of deguelin-induced apoptosis in transformed human bronchial epithelial cells". Biochem. Pharmacol. 68 (6): 1119–24. doi:10.1016/j.bcp.2004.05.033. PMID 15313408.
- ↑ Chun KH, Kosmeder JW, Sun S; et al. (February 2003). "Effects of deguelin on the phosphatidylinositol 3-kinase/Akt pathway and apoptosis in premalignant human bronchial epithelial cells". J. Natl. Cancer Inst. 95 (4): 291–302. doi:10.1093/jnci/95.4.291. PMID 12591985.
- ↑ Caboni P, Sherer TB, Zhang N; et al. (November 2004). "Rotenone, deguelin, their metabolites, and the rat model of Parkinson's disease". Chem. Res. Toxicol. 17 (11): 1540–8. doi:10.1021/tx049867r. PMID 15540952.
External links
- Fang N, Casida JE (May 1999). "Cubé resin insecticide: identification and biological activity of 29 rotenoid constituents". J. Agric. Food Chem. 47 (5): 2130–6. doi:10.1021/jf981188x. PMID 10552508.
- Udeani GO, Gerhauser C, Thomas CF; et al. (August 1997). "Cancer chemopreventive activity mediated by deguelin, a naturally occurring rotenoid". Cancer Res. 57 (16): 3424–8. PMID 9270008.
- Lee HY, Suh YA, Kosmeder JW, Pezzuto JM, Hong WK, Kurie JM (February 2004). "Deguelin-induced inhibition of cyclooxygenase-2 expression in human bronchial epithelial cells". Clin. Cancer Res. 10 (3): 1074–9. doi:10.1158/1078-0432.CCR-0833-3. PMID 14871987.
- Fang N, Casida JE (March 1998). "Anticancer action of cubé insecticide: correlation for rotenoid constituents between inhibition of NADH:ubiquinone oxidoreductase and induced ornithine decarboxylase activities". Proc Natl Acad Sci USA. 95 (7): 3380–4. doi:10.1073/pnas.95.7.3380. PMC 19844. PMID 9520374.
| ||||||||||