Diisobutylaluminium hydride
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Diisobutylaluminum hydride | |
| Other names
DIBAH; DIBAL; DiBAlH; DIBAL-H; DIBALH | |
| Identifiers | |
| 1191-15-7 | |
| ChemSpider | 10430352 |
| Jmol interactive 3D | Image |
| |
| |
| Properties | |
| C16H38Al2 (dimer) | |
| Molar mass | 142.22 (monomer) |
| Appearance | colorless liquid |
| Density | 0.798 g/cm3 |
| Melting point | −80 °C (−112 °F; 193 K) |
| Boiling point | 116 to 118 °C (241 to 244 °F; 389 to 391 K) at 1 mmHg |
| hydrocarbon solvents | |
| Hazards | |
| Main hazards | ignites in air |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Diisobutylaluminium hydride (DIBALH, DIBAL, DIBAL-H or DIBAH, DYE-bal) is a reducing agent with the formula (i-Bu2AlH)2, where i-Bu represents isobutyl (-CH2CH(CH3)2). This organoaluminium compound was investigated originally as a co-catalyst for the polymerization of alkenes.[1]
Properties
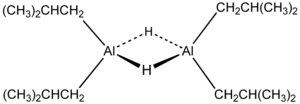
Like most organoaluminum compounds, the compound’s structure is probably more than that suggested by its empirical formula. A variety of techniques, not including X-ray crystallography, suggest that the compound exists as a dimer and a trimer, consisting of tetrahedral aluminium centers sharing bridging hydride ligands.[2] Hydrides are small and, for aluminium derivatives, are highly basic, thus they bridge in preference to the alkyl groups.
DIBALH can be prepared by heating triisobutylaluminium (itself a dimer) to induce beta-hydride elimination:[3]
- (i-Bu3Al)2 → (i-Bu2AlH)2 + 2 (CH3)2C=CH2
Although DIBALH can be purchased commercially as a colorless liquid, it is more commonly purchased and dispensed as a solution in an organic solvent such as toluene or hexane.
Use in organic synthesis
DIBALH is useful in organic synthesis for a variety of reductions, including converting esters and nitriles to aldehydes. DIBALH efficiently reduces α-β unsaturated esters to the corresponding allylic alcohol.[4] By contrast, LiAlH4 reduces esters and acyl chlorides to primary alcohols, and nitriles to primary amines [use Feiser work-up procedure]. DIBALH reacts slowly with electron-poor compounds, and more quickly with electron-rich compounds. Thus, it is an electrophilic reducing agent whereas LiAlH4 can be thought of as a nucleophilic reducing agent.
Safety
DIBALH, like most alkylaluminium compounds, reacts violently with air and water, potentially leading to fires.
References
- ↑ Ziegler, K.; Martin, H.; Krupp, F. (1960). "Metallorganische Verbindungen, XXVII Aluminiumtrialkyle und Dialkyl-Aluminiumhydride aus Aluminiumisobutyl-Verbindungen". Justus Liebigs Annalen der Chemie 629 (1): 14–19. doi:10.1002/jlac.19606290103.
- ↑ Self, M. F.; Pennington, W. T.; Robinson, G. H. (1990). "Reaction of Diisobutylaluminum Hydride with a Macrocyclic Tetradentate Secondary Amine. Synthesis and Molecular Structure of [Al(iso-Bu)]2[C10H20N4][Al(iso-Bu)3]2: Evidence of an Unusual Disproportionation of (iso-Bu)2AlH". Inorganica Chimica Acta 175 (2): 151–153. doi:10.1016/S0020-1693(00)84819-7.
- ↑ Eisch, J. J. (1981). Organometallic Syntheses 2. New York: Academic Press. ISBN 0-12-234950-4.
- ↑ Galatsis, P. (2001). "Diisobutylaluminum Hydride". Encyclopedia of Reagents for Organic Synthesis. New York: John Wiley & Sons. doi:10.1002/047084289X.rd245. ISBN 0471936235.
External links
- Stockman, R. (2001). "Dibal reduction of an amino acid derived methyl ester; Garner's Aldehyde". ChemSpider Synthetic Pages. doi:10.1039/SP161. SyntheticPage 161.
- "Oxidation And Reduction Reactions in Organic Chemistry". University of Southern Maine, Department of Chemistry.
- "Diisobutyl Aluminum hydride (DIBAL-H) and Other Isobutyl Aluminum Alkyls (DIBAL-BOT, TIBAL) as Specialty Organic Synthesis Reagents" (PDF). Akzo-Nobel.
| ||||||||||||||||