DDEF2
| ArfGAP with SH3 domain, ankyrin repeat and PH domain 2 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
PDB rendering based on 1dcq. | |||||||||||||
| |||||||||||||
| Identifiers | |||||||||||||
| Symbols | ASAP2 ; AMAP2; CENTB3; DDEF2; PAG3; PAP; Pap-alpha; SHAG1 | ||||||||||||
| External IDs | OMIM: 603817 MGI: 2685438 HomoloGene: 2888 GeneCards: ASAP2 Gene | ||||||||||||
| |||||||||||||
| RNA expression pattern | |||||||||||||
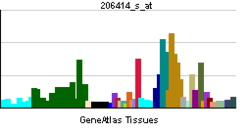 | |||||||||||||
| More reference expression data | |||||||||||||
| Orthologs | |||||||||||||
| Species | Human | Mouse | |||||||||||
| Entrez | 8853 | 211914 | |||||||||||
| Ensembl | ENSG00000151693 | ENSMUSG00000052632 | |||||||||||
| UniProt | O43150 | Q7SIG6 | |||||||||||
| RefSeq (mRNA) | NM_001135191 | NM_001004364 | |||||||||||
| RefSeq (protein) | NP_001128663 | NP_001004364 | |||||||||||
| Location (UCSC) |
Chr 2: 9.21 – 9.41 Mb |
Chr 12: 21.11 – 21.27 Mb | |||||||||||
| PubMed search | |||||||||||||
Arf-GAP with SH3 domain, ANK repeat and PH domain-containing protein 2 is a protein that in humans is encoded by the ASAP2 gene.[1][2][3]
This gene encodes a multidomain protein containing an N-terminal alpha-helical region with a coiled-coil motif, followed by a pleckstrin homology (PH) domain, an Arf-GAP domain, an ankyrin homology region, a proline-rich region, and a C-terminal Src homology 3 (SH3) domain. The protein localizes in the Golgi apparatus and at the plasma membrane, where it colocalizes with protein tyrosine kinase 2-beta (PYK2). The encoded protein forms a stable complex with PYK2 in vivo. This interaction appears to be mediated by binding of its SH3 domain to the C-terminal proline-rich domain of PYK2. The encoded protein is tyrosine phosphorylated by activated PYK2. In vitro it shows strong GTPase-activating protein (GAP) activity towards the small GTPases ADP-ribosylation factor (ARF) 1 and ARF5 and weak activity towards ARF6. The encoded protein is believed to function as an ARF GAP that controls ARF-mediated vesicle budding when recruited to Golgi membranes. In addition, it functions as a substrate and downstream target for PYK2 and SRC, a pathway that may be involved in the regulation of vesicular transport.[3]
Interactions
DDEF2 has been shown to interact with PTK2B.[4]
References
- ↑ Andreev J, Simon JP, Sabatini DD, Kam J, Plowman G, Randazzo PA, Schlessinger J (Mar 1999). "Identification of a New Pyk2 Target Protein with Arf-GAP Activity". Mol Cell Biol 19 (3): 2338–50. PMC 84026. PMID 10022920.
- ↑ Ishikawa K, Nagase T, Nakajima D, Seki N, Ohira M, Miyajima N, Tanaka A, Kotani H, Nomura N, Ohara O (Feb 1998). "Prediction of the coding sequences of unidentified human genes. VIII. 78 new cDNA clones from brain which code for large proteins in vitro". DNA Res 4 (5): 307–13. doi:10.1093/dnares/4.5.307. PMID 9455477.
- 1 2 "Entrez Gene: DDEF2 development and differentiation enhancing factor 2".
- ↑ Andreev, J; Simon J P; Sabatini D D; Kam J; Plowman G; Randazzo P A; Schlessinger J (Mar 1999). "Identification of a New Pyk2 Target Protein with Arf-GAP Activity". Mol. Cell. Biol. (UNITED STATES) 19 (3): 2338–50. ISSN 0270-7306. PMC 84026. PMID 10022920.
Further reading
- Brown MT, Andrade J, Radhakrishna H; et al. (1998). "ASAP1, a Phospholipid-Dependent Arf GTPase-Activating Protein That Associates with and Is Phosphorylated by Src". Mol. Cell. Biol. 18 (12): 7038–51. PMC 109286. PMID 9819391.
- Kondo A, Hashimoto S, Yano H; et al. (2000). "A New Paxillin-binding Protein, PAG3/Papα/KIAA0400, Bearing an ADP-Ribosylation Factor GTPase-activating Protein Activity, Is Involved in Paxillin Recruitment to Focal Adhesions and Cell Migration". Mol. Biol. Cell 11 (4): 1315–27. doi:10.1091/mbc.11.4.1315. PMC 14849. PMID 10749932.
- Uchida H, Kondo A, Yoshimura Y; et al. (2001). "Pag3/Papα/Kiaa0400, a Gtpase-Activating Protein for Adp-Ribosylation Factor (Arf), Regulates Arf6 in Fcγ Receptor–Mediated Phagocytosis of Macrophages". J. Exp. Med. 193 (8): 955–66. doi:10.1084/jem.193.8.955. PMC 2193405. PMID 11304556.
- Kim H, Lee Y (2002). "Interaction of poly(A) polymerase with the 25-kDa subunit of cleavage factor I". Biochem. Biophys. Res. Commun. 289 (2): 513–8. doi:10.1006/bbrc.2001.5992. PMID 11716503.
- Oshiro T, Koyama S, Sugiyama S; et al. (2002). "Interaction of POB1, a downstream molecule of small G protein Ral, with PAG2, a paxillin-binding protein, is involved in cell migration". J. Biol. Chem. 277 (41): 38618–26. doi:10.1074/jbc.M203453200. PMID 12149250.
- Strausberg RL, Feingold EA, Grouse LH; et al. (2003). "Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences". Proc. Natl. Acad. Sci. U.S.A. 99 (26): 16899–903. doi:10.1073/pnas.242603899. PMC 139241. PMID 12477932.
- Ota T, Suzuki Y, Nishikawa T; et al. (2004). "Complete sequencing and characterization of 21,243 full-length human cDNAs". Nat. Genet. 36 (1): 40–5. doi:10.1038/ng1285. PMID 14702039.
- Hashimoto S, Hashimoto A, Yamada A; et al. (2004). "A novel mode of action of an ArfGAP, AMAP2/PAG3/Papa lpha, in Arf6 function". J. Biol. Chem. 279 (36): 37677–84. doi:10.1074/jbc.M404196200. PMID 15231847.
- Kojima C, Hashimoto A, Yabuta I; et al. (2005). "Regulation of Bin1 SH3 domain binding by phosphoinositides". EMBO J. 23 (22): 4413–22. doi:10.1038/sj.emboj.7600442. PMC 526460. PMID 15483625.
- Gerhard DS, Wagner L, Feingold EA; et al. (2004). "The Status, Quality, and Expansion of the NIH Full-Length cDNA Project: The Mammalian Gene Collection (MGC)". Genome Res. 14 (10B): 2121–7. doi:10.1101/gr.2596504. PMC 528928. PMID 15489334.
- Onodera Y, Hashimoto S, Hashimoto A; et al. (2005). "Expression of AMAP1, an ArfGAP, provides novel targets to inhibit breast cancer invasive activities". EMBO J. 24 (5): 963–73. doi:10.1038/sj.emboj.7600588. PMC 554134. PMID 15719014.
- Olsen JV, Blagoev B, Gnad F; et al. (2006). "Global, in vivo, and site-specific phosphorylation dynamics in signaling networks". Cell 127 (3): 635–48. doi:10.1016/j.cell.2006.09.026. PMID 17081983.
- Ewing RM, Chu P, Elisma F; et al. (2007). "Large-scale mapping of human protein–protein interactions by mass spectrometry". Mol. Syst. Biol. 3 (1): 89. doi:10.1038/msb4100134. PMC 1847948. PMID 17353931.
| |||||||||