D-chiro-Inositol
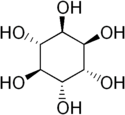 | |
| Names | |
|---|---|
| IUPAC names
cis-1,2,4-trans-3,5,6 -Cyclohexanehexol | |
| Other names
DCI 1,2,4/3,5,6-Hexahydroxycyclohexane | |
| Identifiers | |
| 643-12-9 | |
| ChEBI | CHEBI:27372 |
| ChemSpider | 10254647 |
| 4645 | |
| Jmol interactive 3D | Image |
| |
| |
| Properties | |
| C6H12O6 | |
| Molar mass | 180.16 g·mol−1 |
| Melting point | 230 °C (446 °F; 503 K) |
| Chiral rotation ([α]D) |
[α]23/D +55°, c = 1.2 in H2O |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
D-chiro-Inositol (commonly abbreviated DCI) is a member of a family of related substances often referred to collectively as "inositol," although that term encompasses several isomers of questionable biological relevance. It is known to be an important secondary messenger in insulin signal transduction.[2]
DCI is not abundant in most diets. It can be found in small quantities in buckwheat farinetta.[3] It is likely that in higher vertebrates DCI is made from myo-inositol via the action of an epimerase.[4] Contrary to common claims of the non-availability of DCI, it is in fact commercially available to the public as a nutritional supplement in several countries.
DCI appears to have substantial beneficial effects for PCOS, an observation rationalized by the apparent role of DCI in the etiology of PCOS.[5][6][7] In double-blind studies, women with PCOS who received DCI experienced the following statistically significant benefits when compared with a control group: lowered free and total testosterone, lowered blood pressure, increased insulin sensitivity and a corresponding improvement in glucose disposal, and increased frequency of ovulation.[5][6]
Dietary sources
Dietary sources include buckwheat[3] and Cucurbita ficifolia (fig leaf melon).[8]
See also
References
- ↑ Merck Index, 11th Edition, 4883
- ↑ Larner J (2002). "D-chiro-inositol--its functional role in insulin action and its deficit in insulin resistance". Int. J. Exp. Diabetes Res. 3 (1): 47–60. doi:10.1080/15604280212528. PMC 2478565. PMID 11900279.
- 1 2 Steadman KJ, Burgoon MS, Schuster RL, Lewis BA, Edwardson SE, Obendorf RL. J (2000). "Fagopyritols, D-chiro-inositol, and other soluble carbohydrates in buckwheat seed milling fractions". J Agric Food Chem. 48 (7): 2843–7. doi:10.1021/jf990709t. PMID 10898633.
- ↑ Sun TH, Heimark DB, Nguygen T, Nadler JL, Larner J (2002). "Both myo-inositol to chiro-inositol epimerase activities and chiro-inositol to myo-inositol ratios are decreased in tissues of GK type 2 diabetic rats compared to Wistar controls". Biochem. Biophys. Res. Commun. 293 (3): 1092–8. doi:10.1016/S0006-291X(02)00313-3. PMID 12051772.
- 1 2 Nestler JE, Jakubowicz DJ, Reamer P, Gunn RD, Allan G (1999). "Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome". N. Engl. J. Med. 340 (17): 1314–20. doi:10.1056/NEJM199904293401703. PMID 10219066.
- 1 2 Iuorno MJ, Jakubowicz DJ, Baillargeon JP; et al. (2002). "Effects of d-chiro-inositol in lean women with the polycystic ovary syndrome". Endocrine practice 8 (6): 417–23. doi:10.4158/EP.8.6.417. PMID 15251831.
- ↑ Nestler JE, Jakubowicz DJ, Iuorno MJ (2000). "Role of inositolphosphoglycan mediators of insulin action in the polycystic ovary syndrome". J. Pediatr. Endocrinol. Metab. 13. Suppl 5: 1295–8. PMID 11117673.
- ↑ Xia, T; Wang Q (2000). "D-chiro-inositol found in Cucurbita ficifolia (Cucurbitaceae) fruit extracts plays the hypoglycaemic role in streptozocin-diabetic rats". J Pharm Pharmacol 58 (11): 1527–32. doi:10.1211/jpp.58.10.0014. PMID 17132216.