Ceftibuten
 | |
| Systematic (IUPAC) name | |
|---|---|
|
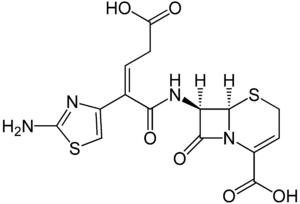
(6R,7R)-7-([(Z)-2-(2-amino-1,3-thiazol-4-yl)-5-hydroxy-5-oxopent-2-enoyl]amino) -8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid | |
| Clinical data | |
| Trade names | Cedax |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a698023 |
| Identifiers | |
| CAS Number |
97519-39-6 |
| ATC code | J01DD14 |
| PubChem | CID 5282242 |
| DrugBank |
DB01415 |
| ChemSpider |
4445419 |
| UNII |
IW71N46B4Y |
| KEGG |
D00922 |
| ChEBI |
CHEBI:3510 |
| ChEMBL |
CHEMBL1605 |
| Chemical data | |
| Formula | C15H14N4O6S2 |
| Molar mass | 410.427 g.mol−1 |
| |
| |
| (verify) | |
Ceftibuten is a third-generation cephalosporin antibiotic. It is an orally administered agent, with two dosage forms, capsule or oral suspension. It is marketed by Pernix Therapeutics under the trade name Cedax.
Clinical use
Indications
Ceftibuten is used to treat acute bacterial exacerbations of chronic bronchitis (ABECB), acute bacterial otitis media, pharyngitis, and tonsilitis. It is also indicated for pneumonia, infections of the urinary tract, enteritis, and gastroenteritis.
Adverse reactions
In 3,000 patients, ceftibuten was well tolerated. The most frequent reactions were gastrointestinal and nauseas.
Formulations
Ceftibuten is available as capsules containing 400 mg, and a powder for oral suspension containing 180 mg per 5 ml.
Spectrum of susceptibility
Ceftibuten is active against Haemophilus influenzae, Moraxella catarrhalis, Escherichia coli, Klebsiella pneumoniae, K. oxytoca, Proteus vulgaris, P. mirabilis, P. providence, Salmonella sp., Shigella sp., Enterobacter sp., and Streptococcus sp.
The following represents minimum inhibitory concentration (MIC) susceptibility data for a few clinically significant microorganisms:
- Haemophilus influenzae: 0.015–1.0 µg/ml
- Moraxella catarrhalis: 0.5–4.0 µg/ml
- Streptococcus pneumoniae: 0.5–256 µg/ml [1]
References
External links
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||