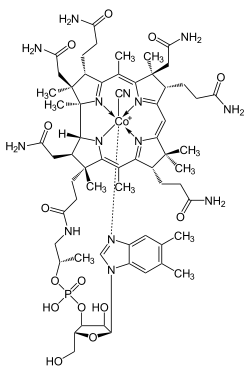
Cyanocobalamin
 | |
 | |
 | |
| Identifiers | |
|---|---|
| 68-19-9 | |
| ChEMBL | ChEMBL1200658 |
| DrugBank | DB00115 |
| EC Number | 200-680-0 V09XX01 (57Co) V09XX02 (58Co) |
| UNII | P6YC3EG204 |
| Properties | |
| C63H88CoN14O14P | |
| Molar mass | 1355.38 g/mol |
| Appearance | Dark red solid |
| Melting point | > 300 °C |
| Boiling point | > 300 °C |
| Soluble | |
| Pharmacology | |
| ATC code | B03 |
| Hazards | |
| Safety data sheet | External MSDS from Fisher Scientific |
| EU classification (DSD) |
Not available |
| S-phrases | S24/25 |
| NFPA 704 | |
| Flash point | N/A |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Cyanocobalamin is the most common and widely produced form of the chemical compounds that have vitamin B12 activity. Vitamin B12 is the "generic descriptor" name for any of such vitamers of vitamin B12. Because the body can convert cyanocobalamin to any one of the active vitamin B12 compounds,[1] by definition this makes cyanocobalamin itself a form (or vitamer) of B12, albeit a largely artificial one.
Cyanocobalamin usually does not occur in living organisms, but animals can convert commercially produced cyanocobalamin into active (cofactor) forms of the vitamin, such as methylcobalamin.[2]
Chemical properties
Cyanocobalamin is one of the most widely manufactured vitamers in the vitamin B12 family (the family of chemicals that function as B12 when put into the body), because cyanocobalamin is the most air-stable of the B12 forms. It is the easiest to crystallize and, therefore, easiest to purify after it is produced by bacterial fermentation, or synthesized in vitro. It can be obtained as dark red crystals or as an amorphous red powder. Cyanocobalamin is very hygroscopic in the anhydrous form, and sparingly soluble in water (1:80). It is stable to autoclaving for short periods at 121 °C (250 °F). The vitamin B12 coenzymes are very unstable in light.
In animals the cyanide ligand is replaced by other groups (adenosyl, methyl), which are the biologically active forms. The remaining portion of the cyanocobalamin remains unchanged.
Chemical reactions
In the cobalamins, cobalt normally exists in the trivalent state, Co(III). However, under reducing conditions, the cobalt center is reduced to Co(II) or even Co(I), which are usually denoted as B12r and B12s, for reduced and super reduced, respectively.
B12r and B12s can be prepared from cyanocobalamin by controlled potential reduction, or chemical reduction using sodium borohydride in alkaline solution, zinc in acetic acid, or by the action of thiols. Both B12r and B12s are stable indefinitely under oxygen-free conditions. B12r appears orange-brown in solution, while B12s appears bluish-green under natural daylight, and purple under artificial light.[3]
B12s is one of the most nucleophilic species known in aqueous solution. It is often referred to as a "supernucleophile". This property allows the convenient preparation of cobalamin analogs with different substituents, via nucleophilic attack on alkyl halides and vinyl halides.[3]
For example, cyanocobalamin can be converted to its analog cobalamins via reduction to B12s, followed by the addition of the corresponding alkyl halides, acyl halides, alkene or alkyne. Steric hindrance is the major limiting factor in the synthesis of the B12 coenzyme analogs. For example, no reaction occurs between neopentyl chloride and B12s, whereas the secondary alkyl halide analogs are too unstable to be isolated.[3] This effect may be due to the strong coordination between benzimidazole and the central cobalt atom, pulling it down into the plane of corrin ring. The trans effect also determines the polarizability of the Co-C bond so formed. However, once the benzimidazole is detached from cobalt by quaternization with methyl iodide, it is replaced by H2O or hydroxyl ions. Various secondary alkyl halides are then readily attacked by the modified B12s to give the corresponding stable cobalamin analogs.[4] The products are usually extracted and purified by phenol-methylene chloride extraction or by column chromatography.[3]
Cobalamin analogs prepared by this method include the naturally occurring coenzymes methylcobalamin and cobamamide, and also other cobalamins which do not occur naturally, such as vinylcobalamin, carboxymethylcobalamin and cyclohexylcobalamin.[3] This reaction is currently being experimented with for use as a catalyst for chemical dehalogenation, organic reagent and photosensitized catalyst system.[5]

Production
Cyanocobalamin is commercially prepared by bacterial fermentation. Fermentation by a variety of microorganisms yields a mixture of methyl-, hydroxo-, and adenosylcobalamin. These compounds are converted to cyanocobalamin by addition of potassium cyanide in the presence of sodium nitrite and heat. Since a number of species of Propionibacterium produce no exotoxins or endotoxins and have been granted GRAS status (generally regarded as safe) by the Food and Drug Administration of the United States, they are currently the preferred bacterial fermentation organisms for vitamin B12 production.[6]
Historically, a form of vitamin B12 called hydroxocobalamin is often produced by bacteria, and was then changed to cyanocobalamin in the process of being purified in activated charcoal columns after being separated from the bacterial cultures. This change was not immediately realized when vitamin B12 was first being extracted for characterization. Cyanide is naturally present in activated charcoal, and hydroxocobalamin, which has great affinity for cyanide, picks it up, and is changed to cyanocobalamin. Cyanocobalamin is the form in most pharmaceutical preparations because adding cyanide stabilizes the molecule.[2]
France accounts for 80% of world production, and more than 10 tonnes/year of this compound is sold; 55% of sales is destined for animal feed, while the remaining 45% is for human consumption.[7]
Pharmaceutical use
Cyanocobalamin is usually prescribed after surgical removal of part or all of the stomach or intestine to ensure adequate levels of vitamin B12 in the bloodstream. It also is used to treat pernicious anemia, vitamin B12 deficiency (due to low intake from food), thyrotoxicosis, hemorrhage, malignancy, liver disease and kidney disease. Cyanocobalamin injections are often prescribed to gastric bypass patients having had part of their small intestine bypassed, making it difficult for B12 to be absorbed via food or vitamins. Cyanocobamide is also used to perform the Schilling test to check a person's ability to absorb vitamin B12.[8]
End product of cyanide poisoning treatment
In cases of cyanide poisoning the patient is given hydroxocobalamin, which is a precursor to cyanocobalamin. The hydroxocobalamin binds with the cyanide ion and forms cyanocobalamin, which can then be excreted by the kidneys. This has been used for many years in France and was approved by the U.S. FDA in Dec 2006, marked under the name Cyanokit.[9][10]
Possible side effects
The oral use of cyanocobalamin may lead to skin problems or immunological reactions such as itching and anaphylactic shock.[11] Possible side effects of cyanocobalamin injection include several allergic reactions such as hives, difficult breathing; redness of the face; swelling of the arms, hands, feet, ankles or lower legs; extreme thirst; diarrhea. Less-serious side effects may include headache, dizziness, leg pain, itching, or rash.[12]
In the treatment of megaloblastic anemia with concurrent vitamin B12 deficiency using B12 vitamers (including cyanocobalamin), there is a possibility that patients could develop hypokalemia due to increased erythropoiesis (red blood cell production) and consequent cellular uptake of potassium upon anemia resolution.[13] When treated with vitamin B12, patients with Leber's disease may suffer serious optic atrophy, possibly leading to blindness.[14]
Forms of vitamin B12 for injection (such as hydroxocobalamin itself) are commonly available as pharmaceuticals, and are actually the most commonly used injectable forms of vitamin B12 in many countries. Injectable cyanocobalamin remains the most commonly injectable vitamin B12 in the United States.
Vitamin B12 metabolism
The two bioactive forms of vitamin B12 are methylcobalamin in the cytosol and adenosylcobalamin in mitochondria. For humans vitamin B12 is only present in animal products (ox liver, mackerel and ox meat) - Reference Intake RI - but also injections of hydroxocobalamin can be used as a source of vitamin B12. Multivitamins often contain cyanocobalamin, which is presumably converted to bioactive forms in the body. Both methylcobalamin and adenosylcobalamin are commercially available as supplement pills. The MMACHC gene product catalyzes the decyanation of cyanocobalamin as well as the dealkylation of alkylcobalamins including methylcobalamin and adenosylcobalamin.[15] This function has also been attributed to cobalamin reductases.[16] The MMACHC gene product and cobalamin reductases enable the interconversion of cyano- and alkylcobalamins.[17]
B12 cyanocobalamin is added to fortify[18] nutrition, like baby milk powder, breakfast cereals, energy drinks, Alpro Soya, vegetarian meat replacements, poultry[19] and swine[20] diets and fish feed.[21] Vitamin B12 becomes inactive due to Hydrogen cyanide HCN and Nitric oxide NO in cigarette smoke. Vitamin B12 becomes inactive due to Nitrous oxide N2O commonly known as laughing gas, used for anaesthesia and as party drug. Vitamin B12 becomes inactive due to microwave or other forms of heating[22] - This all leads to vitamin B12 deficiency and serious diseases, especially for babies and elderly people.
Vitamin B12 is the catalyst of Carbon biochemistry (Methionine cycle, SAMe, DNA, proteins and PEMT) – the Cobalt ion coordinates C as in MUT enzyme molecule (PDB 4REQ).
In the Cytosol
B12 methylcobalamin and 5-methyltetrahydrofolate are needed by Methionine synthase in the Methionine cycle to transfer a methyl group from 5-methyltetrahydrofolate to homocysteine, thereby generating tetrahydrofolate (THF) and methionine, which is used to make SAMe.
SAMe is the universal methyl donor and is used for DNA methylation and to make phospholipid membranes, choline, sphingomyelin, and acetylcholine and other neurotransmitters. Methionine is also the start codon (AUG) in the genetic code, and so protein biosynthesis begins with methionine.
B12 is needed to prevent cancer and autoimmune diseases, since DNA methylation regulates genes and cellular differentiation.
B12 is needed for the prevention of Alzheimer, since synthesis of choline, sphingomyelin membranes, and acetylcholine and other neurotransmitters depend on B12.
THF is needed for DNA synthesis so reduced availability of THF results in ineffective production of cells with rapid turnover, in particular red blood cells.
B12 is needed to prevent megaloblastic anemia and pernicious anemia, since making THF needed for DNA synthesis depends on B12, but sufficient supply of folate in the diet can hide this effect of B12 deficiency, since folate can be converted to THF.
This part of biochemistry in the cytosol needs adequate supply of vitamin B12 methylcobalamin, folate, choline and Magnesium - ATP.
In the Transsulfuration pathway, homocysteine is converted first into cystathionine and then into cysteine and propionyl-CoA.
The metabolism of propionyl-CoA occurs in the mitochondria and requires B12 adenosylcobalamin to make succinyl-CoA. When the conversion of propionyl-CoA to succinyl-CoA in the mitochondria fails due to vitamin B12 deficiency, then elevated blood levels of methylmalonic acid MMA occur. So elevated blood levels of homocysteine and methylmalonic acid MMA are both indicators of vitamin B12 deficiency.

LDL cholesterol is a lipoprotein used by cells to produce in mitochondria ATP energy molecules. Statins can produce muscle pains. LDL is not a type of cholesterol, but rather can be viewed as a container that transports fats, cholesterol, vitamin D, and fat-soluble anti-oxidants to all the tissues of the body.
In Mitochondria
B12 adenosylcobalamin is needed as cofactor in Methylmalonyl-CoA mutase - MUT enzyme.
Processing of cholesterol and protein gives propionyl-CoA that is converted to methylmalonyl-CoA , which is used by MUT enzyme to make succinyl-CoA. Succinyl-CoA is needed to make heme for hemoglobin and it is used in the citric acid cycle to make ATP chemical energy molecules. Magnesium - ATP is used in the cytosol to power the sodium-potassium pump used to maintain the membrane potential. Failure in maintaining the membrane resting potential leads to apoptosis and the cell will die. Magnesium - ATP is also used for muscle contraction and to power the calcium pump. The sarcoplasmic reticulum contains large stores of calcium, which it sequesters and then releases when the muscle cell is stimulated. When the Calcium pump fails to pump calcium back into the sarcoplasmic reticulum, then overload of calcium ions in the cytosol will give crystallization (calcium carbonate and phosphate) and the cell will die.
B12 is needed to prevent cardiovascular disease, since processing cholesterol depends on B12 and making ATP needed for the sodium and calcium ion pumps depends partly on B12.
B12 is needed to prevent anemia, since making porphyrin and heme in mitochondria for producing hemoglobin in red blood cells depends on succinyl-CoA made by B12 adenosylcobalamin.
This part of biochemistry in the mitochondria needs adequate supply of vitamin B12 adenosylcobalamin, biotin, iron and magnesium ions. Egg yolk, legumes, and liver are the best sources for biotin. Raw egg white contains avidin, which binds biotin very strongly and that occurs when mixing egg yolk with egg white. When boiling eggs then avidin denatures and becomes inactive so that biotin remains available from the egg yolk in the diet.
Bad absorption of B12 is possibly related with coeliac disease. Intestinal absorption of vitamin B12 requires successively 3 different protein molecules: Haptocorrin, Intrinsic Factor and Transcobalamin II. The structure of these proteins is available in the Protein Data Bank PDB. You can use the Simple Viewer and then Shift+Mouse = Zoom and Ctrl+Mouse = Move to study these protein structures. Haptocorrin (PDB 4KKI) - Intrinsic Factor (PDB 3KQ4) - Transcobalamin II (PDB 2BB5).
The enzymes that use B12 as a built-in cofactor are Methylmalonyl-CoA mutase (PDB 4REQ) and Methionine synthase (PDB 1Q8J).
Criticisms of Cyanocobalamin as a B12 supplement
In cyanocobalamin the cobalt catalyst is strongly bound to cyanide which makes B12 inactive.
It is comparable with carbon monoxide poisoning iron Fe2+ of Heme B in hemoglobin (PDB 1GZX), which normally binds Oxygen O2 at that place.
It is comparable with cyanide poisoning iron Fe2+ of Heme A in Cytochrome c oxidase - Complex IV (PDB 1OCC) of OXPHOS in the mitochondria.
Cyanide and carbon monoxide are isoelectronic and it is well known that these compounds are highly toxic,
since as ligands they have a very strong interaction in coordination complexes of transition metal ions like iron and cobalt.
It can be a disadvantage to use cyanocobalamin in multivitamins and to use cyanocobalamin to fortify[18] human and animal[20] nutrition.
It is likely that proteins cannot discriminate between bioactive and inactive B12, e.g. cyanide is visible as CN ligand of B12 in Haptocorrin (PDB 4KKI) protein molecule.
Cyanocobalamin is inactive and might occupy the place of vitamin B12 in enzymes.
Results of a large number of animal experiments are about equally divided between those reporting a positive response to dietary cyanocobalamin and those reporting little or no response. Variable responses may be due to several factors, including initial body stores, environmental sources of the vitamin (such as molds, soil and animal excreta).
See also
References
- ↑ Quadros, EV. "Advances in the Understanding of Cobalamin Assimilation and Metabolism". Br J Haematol. 2010 Jan; 148(2): 195–204.
- 1 2 Herbert, V (1988). "Vitamin B-12: Plant sources, requirements, and assay". The American Journal of Clinical Nutrition 48 (3 Suppl): 852–8. PMID 3046314.
- 1 2 3 4 5 David Dophin. Preparation of the Reduced Forms of Vitamin B12 and of Some Analogs of the Vitamin B12 Coenzyme Containing a Cobalt-Carbon Bond. D.B. McCormick and L.D. Wright, Eds. 1971;Vol. XVIII:34-54.
- ↑ Brodie, J. D. (1969). "On the mechanism of catalysis by vitamin B12". Proceedings of the National Academy of Sciences of the United States of America 62 (2): 461–7. Bibcode:1969PNAS...62..461B. doi:10.1073/pnas.62.2.461. PMC 277821. PMID 5256224.
- ↑ Shimakoshi, Hisashi; Yoshio Hisaeda. "Environmental-friendly catalysts learned from Vitamin B12-dependent enzymes" (PDF). TCIMAIL 128: 2.
- ↑ Riaz, Muhammad; Fouzia Iqbal; Muhammad Akram (2007). "Microbial production of vitamin B12 by methanol utilizing strain of Pseudomonas specie". Pak J. Biochem. Mol. Biol. 1 40: 5–10.
- ↑ Kaesler, Bruno (2005), "Vitamins: 9. Vitamin B12 (Cyanocobalamins)", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a27_443
- ↑ Cyanocobalamin. University of Maryland Medical Center
- ↑ Bledsoe, Bryan (2009). Paramedic Care: Principles & Practice, Medical Emergencies. Upper Saddle River, New Jersey: Pearson Education, Inc. p. 471. ISBN 978-0-13-513702-4.
- ↑ Hamel, J. (2011). "A Review of Acute Cyanide Poisoning with a Treatment Update". Critical Care Nurse 31: 72–82. doi:10.4037/ccn2011799. PMID 21285466.
- ↑ "Cyanocobalamin (Cyanocobalamin 50microgram tablets). Side effects". NHS Choices. Archived from the original on 4 July 2015. Retrieved 4 July 2015.
- ↑ "Cyanocobalamin Injection". MedlinePlus. Archived from the original on 19 April 2015. Retrieved 4 July 2015.
- ↑ "Clinical Vitamin B12 Deficiency. Managing Patients". Centers for Disease Control and Prevention. Archived from the original on 26 April 2015. Retrieved 4 July 2015.
- ↑ "Vitamin B12". MedlinePlus. Archived from the original on 5 April 2015. Retrieved 4 July 2015.
- ↑ Luciana Hannibal, Jihoe Kim, Nicola E. Brasch, Sihe Wang, David S. Rosenblatt, Ruma Banerjee, and Donald W. Jacobsen (August 2009). "Processing of alkylcobalamins in mammalian cells: a role for the MMACHC (cblC) gene product". Mol Genet Metab. 2009 Aug; 97(4): 260–266.
- ↑ Watanabe F, Nakano Y. "Purification and characterization of aquacobalamin reductases from mammals". Methods Enzymol. 1997;281;295-305.
- ↑ Quadros EV, Jackson B, Hoffbrand AV, Linnell JC. "Interconversion of cobalamins in human lymphocytes in vitro and the influence of nitrous oxide on the synthesis of cobalamin coenzymes". Vitamin B12, Proceedings of the Third European Symposium on Vitamin B12 and Intrinsic Factor. 1979;1045-1054.
- 1 2 "DSM in Food, Beverages & Dietary Supplements". http://www.dsm.com/. DSM. Retrieved 2 March 2015. External link in
|website=(help) - ↑ "DSM in Animal Nutrition & Health - Poultry". http://www.dsm.com/markets/anh/en_US/Compendium.html. DSM. Retrieved 2 March 2015. External link in
|website=(help) - 1 2 "DSM in Animal Nutrition & Health - Swine". http://www.dsm.com/markets/anh/en_US/Compendium.html. DSM. Retrieved 2 March 2015. External link in
|website=(help) - ↑ "DSM in Animal Nutrition & Health - Vitamin basics". http://www.dsm.com/markets/anh/en_US/Compendium.html. DSM. Retrieved 2 March 2015. External link in
|website=(help) - ↑ Watanabe, F; Abe, K; Fujita, T; Goto, M; Hiemori, M; Nakano, Y (Jan 19, 1998). "Effects of Microwave Heating on the Loss of Vitamin B(12) in Foods". J Agric Food Chem 46 (1): 206–210. doi:10.1021/jf970670x. PMID 10554220. Retrieved 3 March 2015.
| ||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
